Abstract
Objective
To investigate the ability of kobusone to reduce high glucose levels and promote β-cell proliferation.
Methods
Four-week-old female db/db mice were assigned to the kobusone (25 mg/kg body weight, intraperitoneally twice a day) or control group (same volume of PBS). Glucose levels and body weight were measured twice a week. After 6 weeks, intraperitoneal glucose tolerance tests and immunohistochemical studies were performed, and insulin levels were determined. The expression of mRNAs involved in cell proliferation, such as PI3K, Akt, cyclin D3 and p57Kip 2 , was measured by quantitative reverse transcription polymerase chain reaction (RT-qPCR).
Results
Kobusone reduced blood glucose levels after 3 weeks and more strongly increased serum insulin levels than the vehicle. Immunohistochemistry illustrated that kobusone increased 5-bromo-2′-deoxyuridine incorporation into islet β-cells, suggesting that it can stimulate islet β-cell replication in vivo. RT-qPCR indicated that kobusone upregulated the mRNA expression of PI3K, Akt, and cyclin D3 and downregulated that of p57Kip2.
Conclusion
Our findings suggest that kobusone is a potent pancreatic islet β-cell inducer that has the potential to be developed as an anti-diabetic agent.
Introduction
Diabetes is a chronic metabolic disease characterized by hyperglycemia attributable to absolute or relative deficits in insulin secretion by islet β-cells. The loss of β-cell mass in pancreatic islets is a pathological hallmark of the development of both type 1 and type 2 diabetes. 1 Thus, restoring the reduced β-cell mass by regenerating or preserving functional β-cells is a fundamental therapeutic goal for treating or curing diabetes. Unfortunately, currently existing therapies fail to prevent diabetic complications, including cardiovascular disease, neuropathy, nephropathy, and retinopathy. 2 Therefore, there is an urgent need to develop a novel therapy to treat or cure diabetes.
Pancreatic β-cells are dynamic cells that modulate their mass in response to a variety of physiological (pregnancy) 3 , 4 and pathophysiological cues (obesity or insulin resistance). 5 Pre-existing β-cells rather than specialized progenitor cells are the primary sources of new β-cells. 6 , 7 Reports have indicated that preserving β-cell function is a fundamental therapeutic goal for treating or curing diabetes.8–10
A recent study 11 reported that 17 natural products, including kobusone, could stimulate β-cell replication in primary rat islet cells. Kobusone is a natural product produced by Wuhan ChemFaces Biochemical Co., Ltd. (Wuhan, China) and has anti-bacterial, anti-oxidant, anti-inflammatory, analgesic, anti-allergic, anti-helmintic, anti-platelet, anti-diarrheal, and anti-hyperglycemic activities.12–16 Thus, kobusone is widely used in traditional medicine globally for the treatment of many diseases.
This study examined the efficacy of kobusone in stimulating the regeneration of pancreatic islet β-cells in db/db mice and characterized the molecular mechanism of this effect.
Methods
Materials
Kobusone was purchased from Wuhan ChemFaces Biochemical Co., Ltd. A 5-bromo-2′-deoxyuridine (BrdU) Labeling Kit was purchased from BD Biosciences (Franklin Lakes, NJ, USA), and an Ultra Sensitive Mouse Insulin ELISA Kit was purchased from Crystal Chem Inc. (Downers Grove, IL, USA). Primers and probes for PI3K, Akt, cyclin D3, and p57Kip 2 for quantitative reverse transcription polymerase chain reaction (RT-qPCR) were designed by Integrated DNA Technologies (Coralville, IA, USA).
Animals and treatment
Four-week-old female db/db mice (BKS.Cg-m+/+Leprdb) were purchased from The Jackson Laboratory (Bar Harbor, ME, USA). Mice were housed in an animal facility at The Catholic University of Korea, College of Medicine under controlled light (12-hour light/12-hour dark) and temperature conditions with free access to standard mouse food and water. The procedures used and the care of animals were approved by the Institutional Animal Care and Use Committee of The Catholic University of Korea, Seoul, Korea (Approval No.: IACUC 20-10). All studies involving animals were reported in accordance with the ARRIVE guidelines. After 2 weeks of acclimation, the animals were randomly divided into control (n = 5) and treatment groups (n = 5). Mice in the treatment group received kobusone at a dosage of 25 mg/kg body weight (100 µL/g, intraperitoneally twice a day). Control animals were treated with the same volume of PBS. Glucose levels and body weight were randomly measured twice a week. Six weeks after treatment, all animals were subjected to metabolic analysis. Then, all mice were sacrificed using CO2 gas for islet isolation. Half of the islets were used for mRNA measurements, and the remaining portion was used for immunohistochemical analysis.
Intraperitoneal glucose tolerance test (IPGTT)
For the IPGTT, the mice were fasted for 16 hours and intraperitoneally injected with 10% glucose (1 mg/g body weight). Glucose levels were then measured after 0, 30, 60, 90, and 120 minutes using a Glucometer Elite (Bayer, Leverkusen, Germany). 17
Determination of serum insulin levels
Before animals were sacrificed, blood samples (50 µL) were collected from the tails of fed mice into heparinized microhematocrit tubes for the determination of insulin concentrations using an Ultra Sensitive Mouse Insulin ELISA Kit. 18
Immunohistochemical analysis
Pancreata were removed from the db/db mice, fixed overnight in 4% formaldehyde solution, and embedded in paraffin. Paraffin sections (4 µm) were rehydrated, and antigen retrieval in 10 mM sodium citrate solution was performed using a microwave, followed by blocking endogenous peroxidase in 3% H2O2. The following primary antibodies were used: guinea pig anti-swine insulin (1:300; Agilent Technologies, Santa Clara, CA, USA), mouse anti-cyclin D3 (1:40; Vector Laboratory, Burlingame, CA, USA), and mouse anti-BrdU (1:10; BD Biosciences). All images were captured using a Zeiss Axioplan 2 microscope (Carl Zeiss Inc., Oberkochen, Germany). 19 , 20
RT-qPCR analysis
Islets were isolated from pancreases using a method published previously. 18 Total RNA was extracted from islets using an RNeasy Mini Kit (Qiagen, Hilden, Germany). The targeting cDNA sequences were obtained online (www.ncbi.nlm.nih.gov), and PCR primers and probes were designed using PrimerQuest™ (Integrated DNA Technologies). RT-qPCR was performed using an EXPRESS One-Step SuperScript Quantitative RT-PCR Kit (Invitrogen, Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s instructions with a 7900HT Real-Time PCR System (Applied Biosystems, Thermo Fisher Scientific). mRNA levels were calculated using the comparative CT method (XTest/XGAPDH = 2ΔΔCT) with GAPDH as the endogenous reference gene. Primers and probes used in RT-qPCR were designed using PrimerQuest as listed in Table 1.
Primers and probes for quantitative reverse transcription PCR.

Statistical analysis
Data are expressed as the mean ± standard error (SE). Significant differences among groups were evaluated by one-way analysis of variance and Tukey’s multiple comparisons test or by an unpaired two-tailed Student’s t-test using GraphPad PRISM (GraphPad, San Diego, CA, USA). Significance levels are described in individual figure legends.
Results
Kobusone decreased glucose levels in db/db mice
To examine the effects of kobusone, 6-week-old db/db mice were treated with kobusone (25 mg/kg) twice daily. The high glucose levels in db/db mice began to decrease after 3 weeks of treatment. The glucose-lowering effect continued until the end of the 6-week treatment without (Figure 1a). There was no significant change in body weight throughout the whole experiment (Figure 1b). These findings suggest that kobusone, a natural product, lowered blood glucose levels in db/db mice.

Treatment of db/db mice with kobusone reduced blood glucose levels in as little as 3 weeks (a) without causing a dramatic change in body weight (b). Data are presented as the mean ± SE. *, P < 0.01 vs. control.
Kobusone improved glucose tolerance and increased insulin secretion in db/db mice
To determine whether kobusone improves glucose tolerance in diabetic animals, an IPGTT was performed via the IP administration of 10% glucose (1 mg/g body weight) after the mice were fasted for 16 hours. The area under the curve was significantly smaller at 30 (773 vs. 507), 60 (1476 vs. 950), 90 (2017 vs. 1145), and 120 minutes (2411 vs. 1399) in the treated group than that in the control group (all P < 0.001), suggesting the glucose tolerance was improved by kobusone treatment relative to vehicle (Figure 2a). To evaluate islet β-cell insulin secretion, serum insulin levels were determined using a mouse insulin ELISA kit. Again, kobusone enhanced islet β-cell function, thereby boosting insulin secretion into the circulation (Figure 2b).
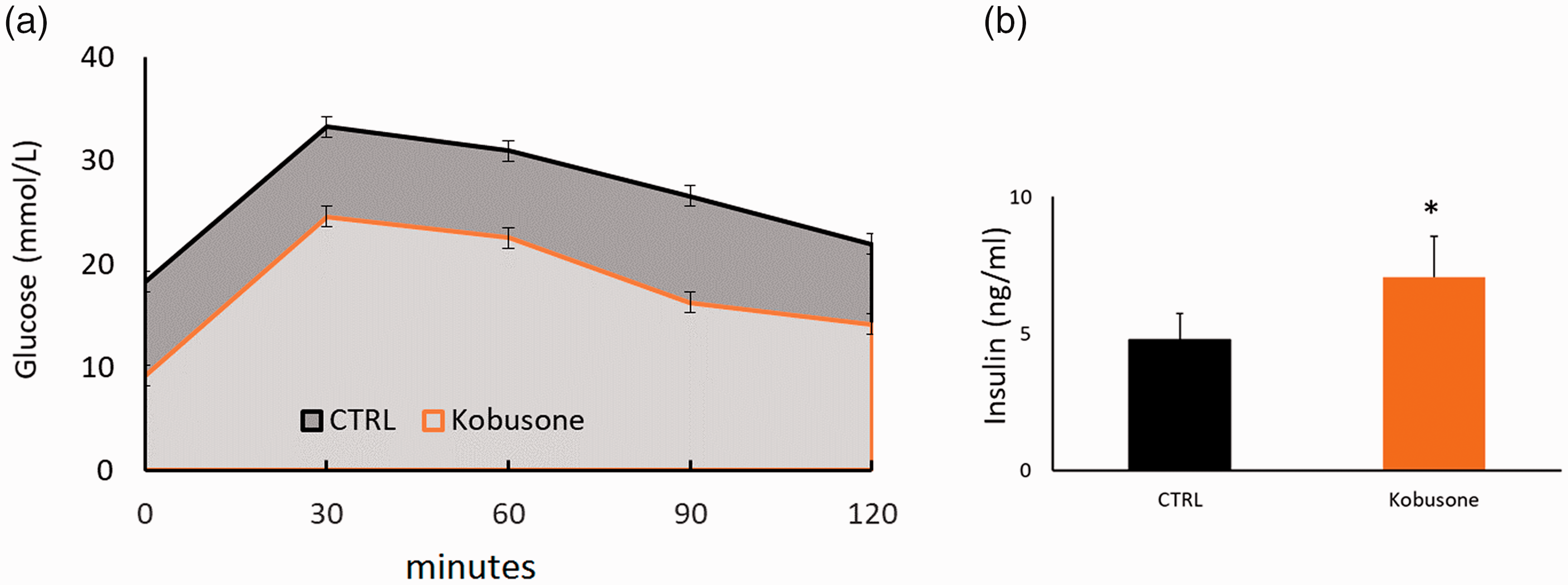
An intraperitoneal glucose tolerance test was performed after 6 weeks of treatment. Kobusone significantly improved glucose tolerance in db/db mice (a) by increasing insulin secretion into blood collected from fed mice (b). Data are presented as the mean ± SE. *, P < 0.01 vs. control.
Kobusone preserved islet mass and stimulated islet β-cell proliferation in db/db mice
After 6 weeks of treatment, pancreata were collected for histological or immunohistochemical studies. Kobusone treatment preserved the normal islet structure, as evidenced by the clear boundary of the islets with the surrounding acinar cells, whereas the structure of control islets was destroyed (Figure 3a and b). Insulin immunostaining revealed that kobusone treatment resulted in strong insulin staining throughout the entire islets (Figure 3d), whereas the control group contained fewer insulin-positive cells in a small part of the islets (Figure 3c). Treated islets contained BrdU-positive cells (red arrows, Figure 3f), whereas no BrdU staining was found in untreated islets (Figure 3e). These results indicate that kobusone preserved the functional islet mass by stimulating β-cell proliferation in db/db mice.

Kobusone preserved the islet mass and structure and induced islet β-cell proliferation in db/db mice. Magnification, ×20.
Kobusone upregulated cyclin D3 and downregulated p57Kip 2 expression in islets through the PI3K/Akt signaling pathway in db/db mice
Cell proliferation is controlled by cyclin-dependent kinases (CDKs), which are positively regulated by cyclins and negatively regulated by CDK inhibitors (CKIs). In addition, the PI3K/Akt signaling pathway plays an important role in regulating multiple biological processes, including cell survival, proliferation, and growth. 21 To examine whether kobusone exerted its role by modulating cyclin D3/p57Kip 2 and PI3K/Akt expression, their mRNA levels were determined by RT-qPCR. Kobusone upregulated cyclin D3, PI3K, and Akt expression but downregulated p57Kip2 expression in pancreatic islets in db/db mice (Figure 4). This result suggests that kobusone stimulated islet β-cell proliferation by modulating CDKs/CDKIs (cyclin D3/p57Kip 2 ) through the PI3K/Akt signaling pathway in db/db mice.

Kobusone upregulated cyclin D3 expression and downregulated p57Kip2 expression in pancreatic islets through the PI3K/Akt signaling pathway in db/db mice in vivo. Total RNA was extracted from islets isolated from control and kobusone-treated db/db mice at the age of 12 weeks, and the mRNA levels of PI3Kr1–3, Akt1–2, cyclin D3, and p57Kip 2 were determined by quantitative reverse transcription PCR. Data are presented as the mean ± SE. *, P < 0.01 vs. control.
Discussion
In our study, we demonstrated that kobusone treatment decreased blood glucose levels and increased serum insulin levels in db/db mice by inducing islet β-cell proliferation via activation of the PI3K/Akt signaling pathway, leading to cyclin D3 upregulation and p57Kip 2 downregulation.
Kobusone is a natural product and a low-molecular-weight fatty acid that can be extracted from the herbaceous plant Cyperus rotundus L. (also known as nutgrass or purple nutsedge), which has anti-bacterial, anti-oxidant, anti-inflammatory, analgesic, anti-allergic, anti-helmintic, anti-platelet, anti-diarrheal, and anti-hyperglycemic activities.12–16 Thus, C. rotundus L. is a widely used plant in traditional medicine globally for the treatment of various diseases.
The PI3K/Akt signaling pathway is an intracellular signal transduction pathway that promotes metabolism, proliferation, cell survival, and growth in response to extracellular signals.21–25 Cell proliferation is controlled by cyclins, CDKs, and CKIs. CDKs are regulated positively by cyclins and negatively by CKIs. 26 , 27 Cyclin D family members play important roles in cell division. The family includes three members (cyclins D1, D2, and D3) that are essential for regulating cell cycle progression via complex formation with CDKs. Human islet β-cells contain abundant cyclin D3, variable amounts of cyclin D1, and little cyclin D2. 28 In particular, cyclin D3 (but not cyclin D1 and D2) in combination with CDK6 stimulates the largest increase in human β-cell proliferation. 28 CDKs play critical roles in cell proliferation by controlling the cell cycle. Cyclin D-dependent kinases initiate Rb phosphorylation in mid-G1 phase, suppressing the ability of Rb to repress E2F family members and activating genes needed to enter S phase. This transition from G1 to S phase in the cell cycle leads to cell proliferation. Conversely, p57Kip2, a member of the CIP/KIP family of CKIs that is highly expressed in human pancreatic β-cells, 29 causes cell cycle arrest in G1 phase. Interestingly, the expression of p57Kip2 is induced by the transcription factor E47, which is involved in the transition from proliferation to cell cycle exit. 30
In our study, kobusone exhibited anti-diabetic activity, lowering blood glucose levels and increasing blood insulin levels in db/db mice. Immunostaining of the pancreata illustrated that kobusone treatment increased BrdU incorporation in islet β-cells, suggesting that it can stimulate islet β-cell regeneration. A molecular mechanism study indicated that the mRNA expression of PI3K, Akt, and cyclin D3 was upregulated by kobusone treatment, whereas p57Kip2 was downregulated. Therefore, our results demonstrated that kobusone can lower blood glucose levels by stimulating pancreatic islet β-cell regeneration in db/db mice. The possible mechanism is that kobusone activates the PI3K/Akt signaling pathway, alleviating the p57Kip2-mediated inhibition of cyclin D family members, which in turn initiate the cell division cycle.
In this study, we tested the potential of kobusone to treat diabetes in mice. Two variables must be considered before preclinical application. We examined the anti-diabetic effect of kobusone in a small number of animals (five per group) over a short period (6 weeks). These two variables greatly limit the generalizability of the study results. Therefore, large-scale animal studies should be performed to confirm the efficacy of kobusone in stimulating β-cell proliferation over a longer period in diabetic animal models. In addition, the side effects of kobusone on carcinogenesis or tumorigenesis should also be examined.
In conclusion, kobusone, a natural product isolated from C. rotundus L, exerted a strong glucose-lowering effect in db/db mice by stimulating β-cell regeneration in vivo. Our data suggest that kobusone can induce cell proliferation by activating the PI3K/Akt signaling pathway, leading to p57Kip2 downregulation and cyclin D3 upregulation. Thus, kobusone is a promising small molecule for development as a pharmacological agent to treat diabetes in humans.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211032849 - Supplemental material for The small molecule kobusone can stimulate islet β-cell replication in vivo
Supplemental material, sj-pdf-1-imr-10.1177_03000605211032849 for The small molecule kobusone can stimulate islet β-cell replication in vivo by Jin woo Choi, Jin-deok Joo, Jang hyeok In, Daewoo Kim, Yongshin Kim, Seung Tae Choi, Jung Han Kim and Hong Soo Jung in Journal of International Medical Research
Footnotes
Acknowledgment
The authors wish to acknowledge financial support from the Catholic Medical Center Research Foundation provided in the program year of 2019.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author contributions
Conceptualization: Jin woo Choi, Hong Soo Jung.
Data curation: Jin-deok Joo, Jang hyeok In, Daewoo Kim, Yongshin Kim, Seung Tae Choi.
Investigation: Jin woo Choi, Hong Soo Jung, Jung Han Kim.
Writing-original draft: Jin woo Choi, Hong Soo Jung.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
