Abstract
The post-operative complication of chylous leakage after breast cancer is relatively rare, and few clinical studies have been published. We report a 64-year-old woman with chylous leakage following modified radical mastectomy. We describe the patient’s diagnostic and treatment process in detail. The patient was diagnosed with grade II (left) breast invasive ductal carcinoma. Post-operatively, the patient's chest wall and axilla were pressurized, and negative pressure drainage was initiated. On the fifth post-operative day, the drainage from the chest wall and axilla increased significantly, and the patient developed chylous leakage on the eighth postoperative day. We injected meglumine diatrizoate (100 mL) and elemene (10 mL) into the patient's axilla, and the chylous leakage gradually resolved 18 days post-operatively. In this report, we focus on managing a case of chylous leakage after modified radical mastectomy for breast cancer. Meglumine diatrizoate combined with elemene is a possible treatment for the management of this rare complication.
Keywords
Introduction
Chylous leakage can occur following thyroid, esophageal, lung, cardiac, and other operations after lymph node dissection and supraclavicular lymph node biopsy. Approximately 75% to 92% of cases of chylous leakage after breast surgery occur in the left breast.1,2 However, this complication after breast cancer surgery is infrequent, with an incidence of 0.36% to 0.84%. 3
To date, limited studies have been published on chylous leakage after breast cancer surgery. Lei et al. 4 reported 14 cases of chylous leakage after modified radical mastectomy in breast cancer. A large amount of fluid can be lost with chylous leakage after breast cancer surgery, and patients are prone to complications, such as dehydration, electrolyte disturbances, and nutritional disorders, which seriously affect patients' health. 5 Chylous leakage increases patients' fear and pain, and therapy can be difficult. In this case report, we discuss a patient who experienced chylous leakage after modified radical operation for left breast cancer. We devised a new treatment method, and the therapeutic effect was excellent.
Case report
A 64-year-old female patient who underwent modified radical mastectomy for left breast cancer was admitted to our hospital for 6 months. Physical examination showed 5 × 5-cm masses in the left breast palpable in 2 to 4 locations. The patient's bilateral axillary and clavicular lymph nodes were not enlarged. Breast color Doppler ultrasonography identified 2 to 4 exploration points appearing as disordered hypoechoic nodules in the left breast. The nodules' sizes were approximately 5 × 5 × 1.2 cm3, with unclear boundaries and uneven echogenicity; the breast imaging report and the Breast Imaging Reporting and Data System (BI-RADS) indicated grade 4C. Mammography showed dense, disorganized, multiple nodular shadows in the left upper quadrant of the breast. The boundaries of the nodules were unclear, and the sizes were approximately 67 mm × 40 mm.
After being admitted to the hospital, the patient underwent mastectomy under general anesthesia. Intraoperative frozen pathology suggested invasive ductal carcinoma of the breast (Figure 1). Subsequently, the patient underwent modified radical mastectomy for breast cancer. The post-operative pathological diagnosis was left breast infiltrating ductal carcinoma, grade II (size: 6.5 × 2.5 × 2 cm3). We found no cancer metastasis in the ipsilateral axillary lymph nodes (0/25). Immunohistochemistry (IHC) indicated positive expression of estrogen receptor (ER) (3+) and E-cadherin (+) and negative expression of cytokeratin 5/6 and 34βE12. Myoepithelial cell staining of the breast duct was negative for P63, calponin, and smooth muscle actin (SMA). Negative expression of cluster of differentiation (CD)34 and D2-40 indicated no intravascular tumor thrombosis, and multidrug resistance testing identified positive expression of human epidermal growth factor receptor 2 HER2 (+) and ER (3+), negative expression of P53 and progesterone receptor (PR), and a Ki67 index of 30%. Tumor-node-metastasis (TNM) staging indicated pT3N0M0 left breast cancer.

Intraoperative frozen section and immunohistochemical staining of tumor tissue. a: frozen section staining (×200); b: frozen section staining (×400); c: calponin (×400); d: cluster of differentiation (CD)34 (×400); e: cytokeratin (CK) 5/6 (×400); f: D2-40 (×400); g: E-cadherin (×400); h: estrogen receptor (ER) (×400); i: human epithelial receptor 2 (HER2) (×400); j: Ki-67 (×400); k: P53 (×400); l: P63 (×400); m: progesterone receptor (PR) (×400); n: smooth muscle actin (SMA) (×400); o: 34βE12 (×400).
Immediately after surgery, the patient’s chest wall and axilla were pressurized, and negative pressure drainage was initiated. The drainage volume increased significantly (Table 1) during the first post-operative week, and on the eighth post-operative day, pale red milky-white drainage fluid appeared from the axillary and chest wall drains. Biochemical testing of the fluid indicated a triglyceride concentration of >1100 mg/L, and the patient was diagnosed with chylous leakage, which increased and then averaged >150 mL/24 hours. The drainage did not decrease when we continued pressurization and drainage while providing a high-protein diet6–8 (Table 2, Figure 2). On days 12 to 13 post-operatively, we injected meglumine diatrizoate (100 mL) into the patient’s axilla. We also clamped the drain, and applied compression to the axilla and chest wall to evenly disperse the agent. After 2 hours, we discontinued the negative pressure, and aspirated the diatrizoate meglumine through the drain. We then reinstituted pressure bandaging and negative pressure drainage. Fourteen days postoperatively, we injected 10 mL of elemene along the drain, drained the fluid after 30 minutes, then reinstituted pressure bandaging and negative pressure drainage. The drain was removed smoothly, and after 2 days of continuous pressure dressing, no subcutaneous fluid was seen. The patient had no subcutaneous effusion when she subsequently underwent computed tomography (CT) 18 days after discharge (Figure 3).
Chest wall and axillary drainage over time.

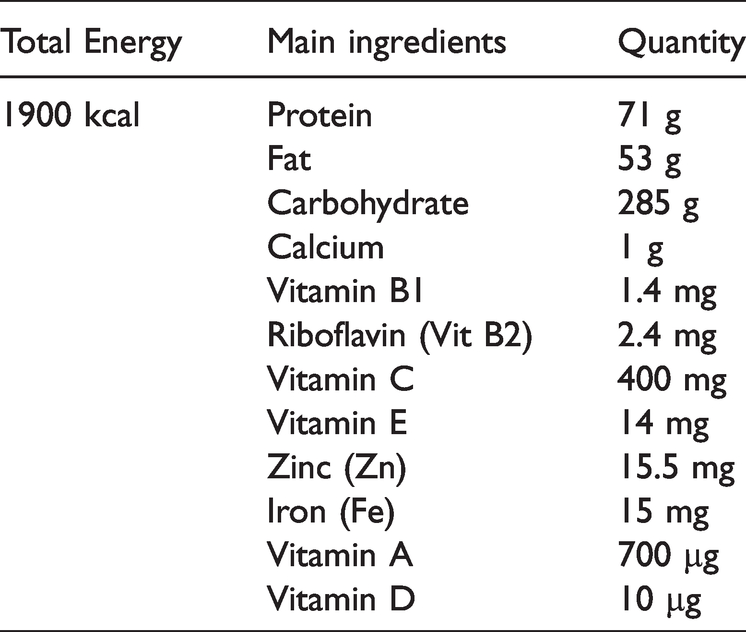
The patient’s high-protein diet after developing chylous leakage.
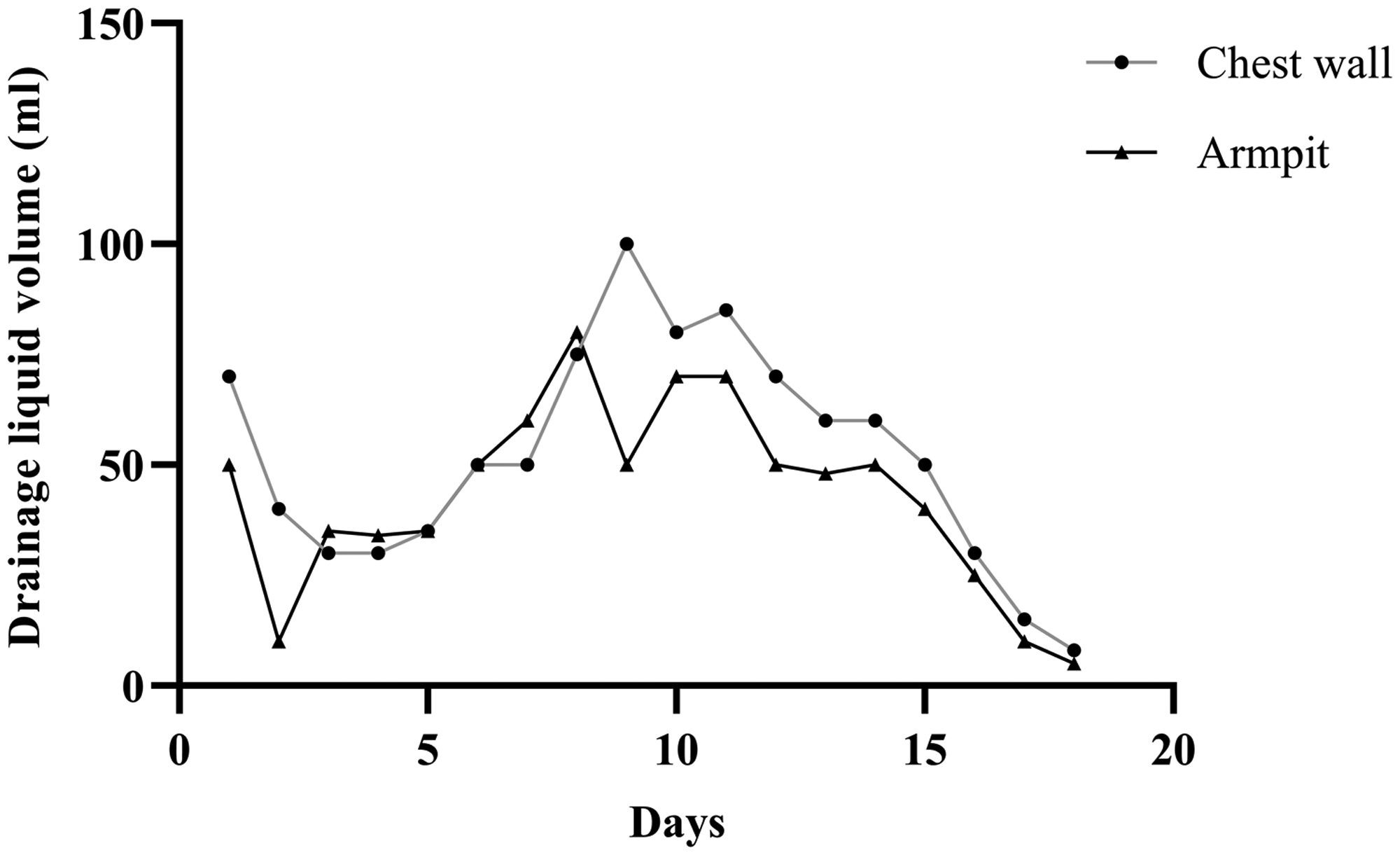
Drainage volumes on different treatment days.

Chest computed tomography (CT) 18 days after discharge from the hospital. a: enhanced CT; b: non-enhanced CT.
Discussion
Chylous leakage mainly occurs after neck lymph node dissection, and is extremely rare after modified radical mastectomy. 9 Generally, local compression and adequate drainage can reduce its occurrence. The incidence of chylous leakage after modified radical mastectomy for breast cancer is very low, and traditional treatment methods have been used. This complication extends the patient's treatment period, and increases the associated costs. Additionally, re-operation is not necessarily effective; the risk of leakage persists, and trauma from a second surgery is inevitable, increasing the patient's pain. 10 Farkas et al. 11 performed a systematic review that summarized 51 cases from 31 chylous leakage studies. The authors concluded that abnormal thoracic duct anatomy is the most likely cause of chylous leakage. When the amount of chylous leakage is less than 500 mL/24 hours, the authors recommend conservative treatment, such as no-suction flow, low-fat diet, and axillary compression bandaging. A second surgical treatment should be considered with high amounts of chylous leakage (>500 mL/24 hours), and in patients who do not respond to conservative measures.
In this study, chylous leakage occurred after modified radical mastectomy for breast cancer, and the chylous fluid volume was high. The patient was actively treated according to traditional methods, such as a controlled diet and continuous pressure bandaging. The patient also received a meglumine diatrizoate local injection, but the effect was limited. There have been reports of refractory chylothorax with meglumine diatrizoate and elemene. Meglumine can quickly form a hypertonic state around the incision, causing local tissue dehydration. When we injected meglumine 12 to 13 days post-operatively, the 24-hour drainage volume decreased by only approximately 15 mL, and the daily drainage remained at 60 mL. We then infused 10 mL of elemene to strengthen meglumine’s efficacy, 14 days after injecting the meglumine. In chylothorax treatment, elemene can stimulate a pleural response, which induces pleural hypertrophy and adherence; the pleural cavity disappears, and the fistula becomes occluded. In our patient, under the stimulation of both drugs, aseptic inflammation occurred in the axilla and subcutaneous tissues. The skin surface appeared reddish and swollen, and aseptic inflammation related to telangiectasia developed. Four days later, the drainage volume decreased sharply. No effusion was confirmed with B-ultrasonography 18 days post-operatively, and chest CT examination 18 days after discharge from the hospital confirmed no effusion, indicating complete resolution of the chylous leakage.
Conclusion
In this study, we discussed the management of a case of chylous leakage after modified radical mastectomy for breast cancer. We described a treatment method for the clinical management of this rare complication. Meglumine diatrizoate combined with elemene may be complementary treatments for refractory chylous exudation.
Footnotes
Ethics statement
The research ethics committee of the 971st Naval Hospital of the Chinese People's Liberation Army approved this study (approval number: 2018032701). Written informed consent was obtained from the patient for publication of this manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
