Abstract
Transcatheter arterial embolization (TAE) is the standard of care for haemodynamically-stable patients with blunt hepatic injury but it is sometimes impossible due to unfavourable vascular anatomies. This case report describes a 43-year-old male patient with abdominal pain following a motorcycle accident. Based on computed tomography (CT) findings, he was diagnosed with high-grade hepatic injury with coeliac axis stenosis (CAS) due to compression by the median arcuate ligament, and an aberrant right hepatic artery. Contrast-enhanced ultrasonography (CEUS) demonstrated multiple high echogenic tubular and ovoid structures suggestive of active bleeding within the injured liver area. Angiography revealed unique interlobar and intrahepatic collateral vessels between the right and left hepatic arteries. Liver haemorrhages were also identified. Catheterization of the feeding arteries through the collateral pathway was unsuccessful, so a decision was made to cannulate the stenotic portion of the coeliac trunk with a 5-Fr Yashiro catheter. After several attempts, the microcatheter was successfully advanced coaxially into the common hepatic artery. Embolization was performed with a 1:2 mixture of N-butyl cyanoacrylate and iodized oil. Successful haemostasis was confirmed following TAE. CEUS helped clinicians identify active bleeding following traumatic solid organ injury. TAE was a safe and effective treatment strategy. Before performing TAE, attention should be given to the presence of CAS associated with compression by the median arcuate ligament.
Keywords
Introduction
High-grade hepatic injury comprises about 20% of all hepatic injury and hepatic injury with haemorrhage is a significant cause of morbidity and mortality. 1 The efficacy of transcatheter arterial embolization (TAE) for hepatic injury is well established, with success rates ranging from 85% to 100%.2,3 However, it is sometimes technically challenging to perform when endovascular treatment is not feasible due to limitations, such as those caused by anatomical anomalies of blood vessels.4,5 Among such anomalies, coeliac axis stenosis (CAS) is a limiting factor with respect to accessibility of the damaged hepatic vessels.5,6 For TAE to be performed successfully, selective catheterization of bleeding branches is necessary. 7 Therefore, knowledge of the collateral circulation associated with CAS, and how to overcome the accompanying challenges, may be important for the successful execution of TAE. There is limited research considering the efficacy and safety of TAE for traumatic hepatic injury in patients with CAS.4,6 In addition, although contrast-enhanced ultrasonography (CEUS) is considered a useful tool in the differential diagnosis of hepatic nodules, its modality for identifying active bleeding in hepatic injury is little-explored.8,9 This current case report describes haemorrhage following blunt hepatic injury, confirmed by CEUS, in a patient with unique collateral pathways caused by a combination of CAS and an aberrant hepatic artery. The patient was treated successfully with TAE.
Case report
In May 2020, a 43-year-old male patient without significant medical history was admitted to the Emergency Department, Chungbuk National University Hospital, Cheongju, South Korea following a motorcycle accident that took place 3 h before. He had moderate upper abdominal pain that did not involve the entire abdomen. On physical examination, the abdomen was mildly distended and localized tenderness without rebound tenderness was identified at the upper abdomen. The patient was haemodynamically stable with a blood pressure of 120/70 mmHg, heart rate of 88 beats/min, respiratory rate of 24 cycles/min and a body temperature of 36.2 °C. Laboratory tests identified mild leukocytosis (white blood cell count 12.02 × 103/µl) and abnormally high liver enzyme levels (aspartate aminotransferase 245 IU/l; alanine aminotransferase 235 IU/l) were detected; otherwise, there were no notable findings. Computed tomography (CT) revealed huge ill-defined low-density lesions involving the anterior (S5, S8) and S4 segments of the liver, suggestive of high-grade hepatic injury (American Association for the Surgery of Trauma grade IV), with a small amount of haemoperitoneum. The initial multidetector CT scans were performed on a 256-detector CT scanner (Revolution CT; GE Healthcare, Waukesha, WI, USA) within 2 h of his arrival at the Emergency Department. A total of 1.5 ml/kg body weight of nonionic contrast material (Iomeron; Bracco, Milan, Italy) was injected into an antecubital vein at a rate of 2.3 ml/s using a power injector. However, there was no definite evidence of active bleeding on the CT images (Figure 1A and 1B). Moreover, the root of the coeliac trunk was severely stenosed due to compression by the median arcuate ligament (MAL) (Figure 1C) and an anomaly in which the right hepatic artery (RHA) arose from the superior mesenteric artery (SMA), with the development of unique collateral vessels, was identified on a maximum-intensity projection image (Figure 1D). Grayscale ultrasound revealed ill-defined heterogeneous echogenic lesions suggestive of blunt injury to the liver (Figure 2A). A CEUS examination revealed multiple high-echogenic tubular and ovoid structures, not visible on the initial CT scan, which were suggestive of active bleeding within the injured region of the liver (Figure 2B). CEUS was performed using a contrast agent (Sono Vue; Bracco), which is a second-generation blood-pool contrast agent consisting of stabilized microbubbles of sulphur hexafluoride gas covered by a stabilizing phospholipidic membrane. CEUS examination was performed with an ultrasound device (LOGIQ P9; GE Healthcare, Chicago, IL, USA) using a curved transducer (C1-5-RS; bandwidth: 1–6 MHz) and a coded contrast imaging software that detects the fundamental nonlinear response of the microbubbles at low mechanical index (MI: 0.14–0.16).
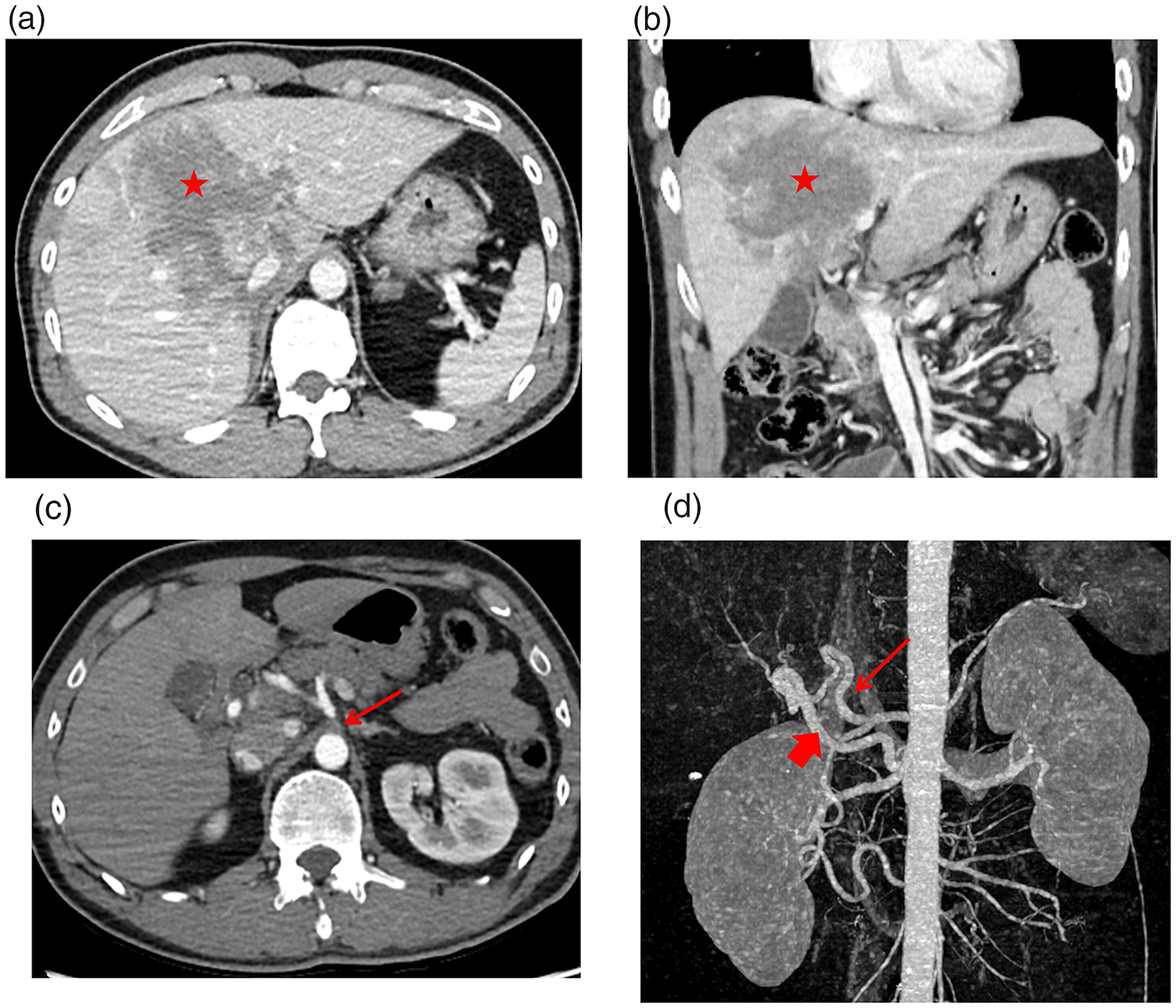
Computed tomography (CT) imaging in a 43-year-old male patient with abdominal pain following a motorcycle accident: (a and b) CT scans revealing huge ill-defined low-density lesions involving both lobes of the liver, suggestive of blunt liver injury (red asterisk). However, there was no definite evidence of active bleeding on this CT image; (c) CT axial image in the arterial phase revealing the median arcuate ligament (red arrows) compressing the coeliac axis; (d) anomaly of the right hepatic artery (thick red arrow) arising from the superior mesenteric artery with the development of unique collateral vessels (thin red arrow), identifiable on a maximum-intensity projection image. The colour version of this figure is available at: http://imr.sagepub.com.

Ultrasound imaging in a 43-year-old male patient with abdominal pain following a motorcycle accident: (a) greyscale ultrasound revealing ill-defined heterogeneous echogenic lesions suggestive of blunt injury to the liver (red circle); (b) contrast-enhanced ultrasonography revealing multiple hyper-enhanced tubular and ovoid structures (red arrow), suggestive of active bleeding within the injured region of the liver. The colour version of this figure is available at: http://imr.sagepub.com.
The patient was referred for emergency angiography. The aberrant RHA arising from the SMA was selected. Subsequently, angiography revealed interlobar and intrahepatic collateral vessels between the RHA and the left hepatic artery, and multiple sites of active bleeding in the liver, consistent with the CEUS findings (Figure 3A). A microcatheter was coaxially loaded to select the feeding arteries throughout the collateral pathway. However, catheterization of these bleeding vessels was unsuccessful due to the excessive length and severe tortuosity of the collateral pathway (Figure 3B). After a number of failed attempts at catheterization through the aberrant RHA arising from the SMA, a decision was made to cannulate the stenotic portion of the coeliac trunk with a 5-Fr Yashiro catheter (Terumo, Tokyo, Japan), and, after several attempts, a 1.9-Fr microcatheter (Asahi Intecc, Aichi, Japan) was successfully advanced coaxially into the common hepatic artery (Figure 3C). Selective angiography of the hepatic artery revealed active bleeding (Figure 3D) and embolization was performed with a 1:2 mixture of N-butyl cyanoacrylate and iodized oil. Post-embolization angiography was performed to confirm successful haemostasis without active bleeding, but small haemorrhages supplied by fine intrahepatic collateral vessels remained (Figure 4). The clinician decided to manage the residual bleeding conservatively, given the relatively small size of the haemorrhages and the stable haemodynamics of the patient. The post-interventional course was uneventful and the patient was discharged on the 13th day post-intervention. In the 7 months following the procedure, the patient displayed no further clinically-evident bleeding as monitored by multidetector CT.

Angiography in a 43-year-old male patient with abdominal pain following a motorcycle accident: (a) angiography of the aberrant right hepatic artery reveals interlobar (thin red arrow) and intrahepatic (thick red arrow) collateral vessels between the right and left hepatic arteries, and multiple sites of active bleeding (red arrowheads) in the liver, consistent with the contrast-enhanced ultrasonography findings; (b) a microcatheter was coaxially loaded to select the feeding arteries throughout the collateral pathway. However, catheterization of these bleeding vessels was unsuccessful due to the excessive length and severe tortuosity of the collateral pathway; (c) following the failed catheterization, a decision was made to cannulate the stenotic portion of the coeliac trunk with a 5-Fr RHR catheter (red arrow), and after several attempts, the microcatheter (red arrowhead) was successfully advanced coaxially into the common hepatic artery; (d) selective angiography of the hepatic artery revealed active bleeding (red circle) and embolization is performed with a 1:2 mixture of N-butyl cyanoacrylate and iodized oil. The colour version of this figure is available at: http://imr.sagepub.com.

Control angiography after embolization shows persistent small haemorrhages (red arrows) supplied by fine intrahepatic collateral vessels. The colour version of this figure is available at: http://imr.sagepub.com.
This report was approved by the Institutional Review Board of Chungbuk National University Hospital, Cheongju, South Korea (no. CBNUH 2020-12-007) and the need for informed consent was waived. Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
Discussion
Before the introduction and advancement of interventional radiological equipment and techniques, surgical management was recognized as the treatment of choice for haemorrhages resulting from traumatic hepatic injury.1,2 Currently, selective TAE is considered the most appropriate therapy for haemorrhages caused by traumatic solid organ injury.3,7 However, endovascular treatment for haemorrhages following traumatic hepatic injury is sometimes technically challenging, and occasionally even impossible, due to an unfavourable vascular anatomy, such as that caused by CAS, resulting in a difficult approach to the injured vessels.4,5,10 To safely and successfully insert the catheter into the bleeding site through the occluded coeliac axis, it is important to understand the pathological cause and anatomical structure of the occluded site, and adjust the catheterization technique accordingly. 7
The incidence of coeliac artery occlusion has been reported to range from 12.5% to 24% in Western populations.11,12 The main causes of CAS include compression of the coeliac axis by the MAL, acute and chronic dissection, and atherosclerosis, with MAL compression being described as the most frequent cause, particularly in Asian populations.6,11,12 For example, in a study of Koreans with hepatocellular carcinoma, the most common cause of CAS was compression of the coeliac axis by the MAL, which was observed in approximately 55% of patients. 6 Compared with atherosclerotic occlusive lesions, catheterizing the occluded coeliac axis is considered possible because extrinsic compression by the MAL may maintain the potential lumen of the coeliac axis.5,6,10 CT provides grounds for suspecting extrinsic compression of the coeliac axis by the MAL.11,12 Specifically, the relevant CT findings are: dilatation of the peripancreatic collateral vasculature, effacement or narrowing of the coeliac axis by an anterior soft tissue band, and poststenotic dilatation of the distal portion of the coeliac axis.4,11 In this current case, initial CT images revealed the MAL of the diaphragm to be obstructing the coeliac axis. Therefore, cannulation of the stenotic coeliac axis was attempted, and subsequently, the catheter was successfully advanced via the occluded coeliac axis.
Severe CAS is usually associated with the enlargement of collateral arteries such as the pancreaticoduodenal arcades, which supply retrograde blood flow from the SMA to the coeliac axis. 11 Moreover, anatomical variation in the site of origin of the hepatic artery is the main cause of unusual collateral route development, including that of intersegmental collateral vessels or interlobar vessels,11,12 as in the present case. These collateral routes can be a secondary route choice for hepatic embolization in patients with CAS.4,5 However, according to previously published reports, when performing microcatheter manipulation and superselective catheterization of feeding arteries, an approach through the occluded coeliac axis is better than an approach through collateral pathways such as the pancreaticoduodenal arcades.6,10 In this current case, a long and overly tortuous unique collateral pathway made it difficult to control the tip of the microcatheter, lengthening the procedure time and eventually resulting in failure to advance the catheter to the bleeding site. After the failed attempts via this approach, cannulation of the occluded coeliac axis was successfully performed using a combination of a 5-Fr Yashiro catheter and a 1.9-Fr microcatheter, and the bleeding focus was superselectively catheterized. For the inexperienced interventional radiologist, there is increased risk of arterial dissection as a result of repeated attempts at catheterization of the coeliac axis when the trunk is significantly compressed or occluded. According to the cause of the CAS, it is recommended to use appropriate catheterization techniques to minimize arterial injury. According previous research, which implemented a similar cannulation technique to the one used in the current case, catheterization of CAS caused by MAL showed a success rate of 64%, while procedure-related complications occurred in only one of 26 patients (3.8%). 6 Therefore, we recommend that access with a catheter through the occluded coeliac axis should be considered as the initial approach in TAE following traumatic hepatic injury in patients with CAS, especially when CAS is caused by MAL compression.
Abdominal CT is considered the most accurate diagnostic modality for grading liver injuries in blunt hepatic injury and it is useful for selecting the treatment option. In addition to injury grading, contrast-enhanced CT can also detect an overt haemorrhage (i.e. blush, contrast extravasation and venous phase) and concomitant intra-abdominal organ injuries, and it also quantifies the associated haemoperitoneum.13,14 The detection of a haemorrhage using CT is important because it indicates an ongoing, potentially fatal haemorrhage, which is a strong risk factor for the failure of nonoperative treatment, and it allows for prompt surgery or TAE.1,7 Evidence of an active haemorrhage with CT, or the clinical signs of an ongoing haemorrhage in the absence of other sources, should be considered as indications for TAE. However, when the hepatic injury occurs over a wide area, especially in the case of intrahepatic injury, the haemorrhage may be difficult to distinguish from normal blood vessels if it appears as foci or a blush rather than a definite active ‘jet-like’ extravasation.14,15
In recent years, a new ultrasound technique using contrast agents, CEUS, has been developed. At our institution, CEUS is regularly performed prior to any intervention in haemodynamically-stable patients suspected of hepatic haemorrhage. 9 In patients with abdominal trauma, CEUS is more sensitive than conventional ultrasonography in detecting solid organ injury, and the levels of sensitivity and specificity in CEUS have also been reported as similar to those of CT for the detection and grading of traumatic abdominal lesions.8,9 A previous study demonstrated that CEUS may be a viable new tool for the evaluation of active abdominal bleeding. 16 Another study also demonstrated that the sensitivity for detecting active bleeding in patients with blunt hepatic and splenic trauma did not differ between CEUS and multidetector CT. 8 CEUS has another advantage: its ability to continuously scan the region of interest without the burden of radiation, which eliminates the risk of missing delayed extravasations. 8 The use of CEUS for arterial injuries can facilitate the rapid detection of the anatomical source of the progressing haemorrhage and thus guide targeted angiographic management, as described in the present case. CEUS can therefore be considered an alternative diagnostic tool to contrast-enhanced CT in the follow-up of organ injury-related angiographic intervention, without the burden of radiation or the administration of contrast media, which can be harmful to patients with renal insufficiency. 9
In conclusion, the use of CEUS in this current case helped clinicians identify active bleeding following traumatic solid organ injury. TAE was an effective and safe treatment strategy for these haemorrhages. Given the increasing evidence that many CAS cases in Asian populations are associated with MAL compression, improved understanding of the causes of CAS when performing TAE should help clinicians identify appropriate access routes and ensure improved outcomes.
Footnotes
Author contributions
Y.H.S. and Y.K. contributed to manuscript drafting and reviewed the literature. Y.H.S. collected and collated the clinical data. Both authors read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
