Abstract
Pulmonary haemorrhage is an important complication of leptospirosis. We herein report an uncommon case of severe pulmonary haemorrhage and multiple organ failure caused by leptospirosis in a 49-year-old man who was previously healthy. He was a farm worker who was admitted to the hospital because of haemoptysis. He had worked in a paddy field 4 days prior to admission. Chest computed tomography revealed pulmonary haemorrhage, which rapidly deteriorated into haemorrhagic shock and multiple organ failure. Based on the patient’s possible history of contact with contaminated water and the DNA sequence of Leptospira detected in his bronchoalveolar lavage fluid, the patient was diagnosed with pulmonary haemorrhagic leptospirosis. Despite the administration of a fluid bolus, norepinephrine, broad-spectrum antibiotics, and haemostatics, and even with administration of a blood transfusion and extracorporeal life support, the pulmonary haemorrhage could not be controlled effectively. The patient eventually died of haemorrhagic shock. Leptospirosis can be a life-threatening disease despite aggressive treatment, even with extracorporeal life support. Next-generation sequencing can provide important diagnostic clues for patients with atypical leptospirotic symptoms.
Keywords
Introduction
Leptospirosis is one of the most common zoonoses worldwide, and its mortality rate is similar to or higher than that due to haemorrhagic fever of other causes. Each year, an estimated 1.03 million cases and 58,900 deaths due to leptospirosis occur worldwide. 1 Severe pulmonary haemorrhagic leptospirosis is the leading cause of mortality associated with this disease.1–3 Affected patients usually require mechanical ventilation, and the outcome is poor regardless of the use of advanced life support and antibiotics.4,5 Among all patients with pulmonary haemorrhage, 40% to 60% may rapidly die of severe acute respiratory failure. 6
Case report
The reporting of this study conforms to CARE guidelines. 7
A 49-year-old male farm worker presented to the hospital with a 4-day history of dizziness, fatigue, chest tightness, cough, chills, fever, conjunctival congestion, and myalgia. He also had a history of haemoptysis, which resulted in emergency admission. He had no significant medical history.
Upon admission to our hospital, the patient was conscious with a body temperature of 38.7°C, pulse rate of 106 beats/minute, respiration rate of 32 breaths/minute, and blood pressure of 96/62 mmHg. He also had shortness of breath and tachycardia. Laboratory examination revealed anaemia as shown by a haemoglobin level of 107.0 g/L (reference range, 130–175 g/L), thrombocytopenia as shown by a platelet count of 62.0 × 109/L (reference range, 125–350 × 109/L), inflammation as indicated by a C-reactive protein level of 138.2 mg/L (reference range, 0–8 mg/L), and liver dysfunction as shown by an aspartate aminotransferase level of 62 U/L (reference range, 10–37 U/L) and alanine aminotransferase level of 70 U/L (reference range, 10–40 U/L). Urinalysis revealed haematuria, mild proteinuria, and leukocytes in the urine. During the night of admission, the patient’s condition deteriorated and he developed massive haemoptysis, dyspnoea, and cold extremities. A computed tomography scan (Figure 1) revealed bilateral pulmonary infiltrations. The patient was then transferred to the intensive care unit.

(a) On admission, computed tomography showed diffuse intrapulmonary haemorrhage. (b) Chest X-ray examination on the night of admission showed obvious bilateral pulmonary infiltrations.
In the intensive care unit, he was placed on mechanical ventilation and treated with several doses of a fluid bolus (normal saline), broad-spectrum antibiotics (intravenous moxifloxacin at 0.4 g once daily), haemostatics (intravenous tranexamic acid at 0.5 g every 12 hours and intravenous carbazochrome sodium sulphonate at 80 mg every 12 hours), and blood transfusion (red blood cell suspension and platelets) for 3 days. However, this treatment was ineffective. On the third day of admission, he started desaturating due to continuous massive pulmonary haemorrhage despite the mechanical ventilation. He developed multiple organ dysfunction syndrome, including liver failure, renal failure, and myocardial depression. His aspartate aminotransferase, creatinine, CK-MB, and PaO2/FiO2 ratio were 4660 U/L, 247 µmol/L, 356 IU/L, and 70, respectively. Veno-venous extracorporeal membrane oxygenation and continuous renal replacement therapy were initiated. Under extracorporeal life support (ECLS), his blood pressure was 130–100/50–30 mmHg (with administration of norepinephrine at 2 µg/kg/minute), and his arterial oxygen saturation was 88% to 96%. Meanwhile, his microcirculation was suboptimal as indicated by anuria, hyperlactic acidaemia, significant acrocyanosis, cold limbs, and clamminess. His finger pulse oxygenation readings could not be obtained. On the fourth day of admission, fibreoptic bronchoscopy revealed diffuse haemorrhage in the mucosa of the bronchi and bronchioles. We sequenced the specimens collected from the airway and alveolar lavage fluid using next-generation gene sequencing (NGS). The next day, the DNA of Leptospira interrogans was detected in the bronchoalveolar lavage fluid by NGS (shotgun sequencing). With the patient’s possible history of contact with contaminated water and the DNA sequence of Leptospira detected in his bronchoalveolar lavage fluid, pulmonary haemorrhagic leptospirosis was considered, and the patient was treated with intravenous injection of 400,000 U of penicillin every 6 hours.
Unfortunately, despite aggressive treatment to control the pulmonary haemorrhage, the massive haemoptysis persisted. On the fifth day of admission, the patient’s haemoglobin level dropped from 107 to 65 g/L, his platelet count dropped from 62 to 43 × 109/L, and his fibrinogen level dropped from 7.70 to 0.76 mg/dL because of the massive haemoptysis, regardless of the blood products that had been administered during the previous 3 days (46.5 U red blood cell suspension, 40 U platelets, 3980 mL fresh frozen plasma, and 20 U cryoprecipitate) (Table 1). On day 6 of admission, the patient died of haemorrhagic shock caused by refractory pulmonary haemorrhage and irreversible multiple organ failure.
Laboratory results.
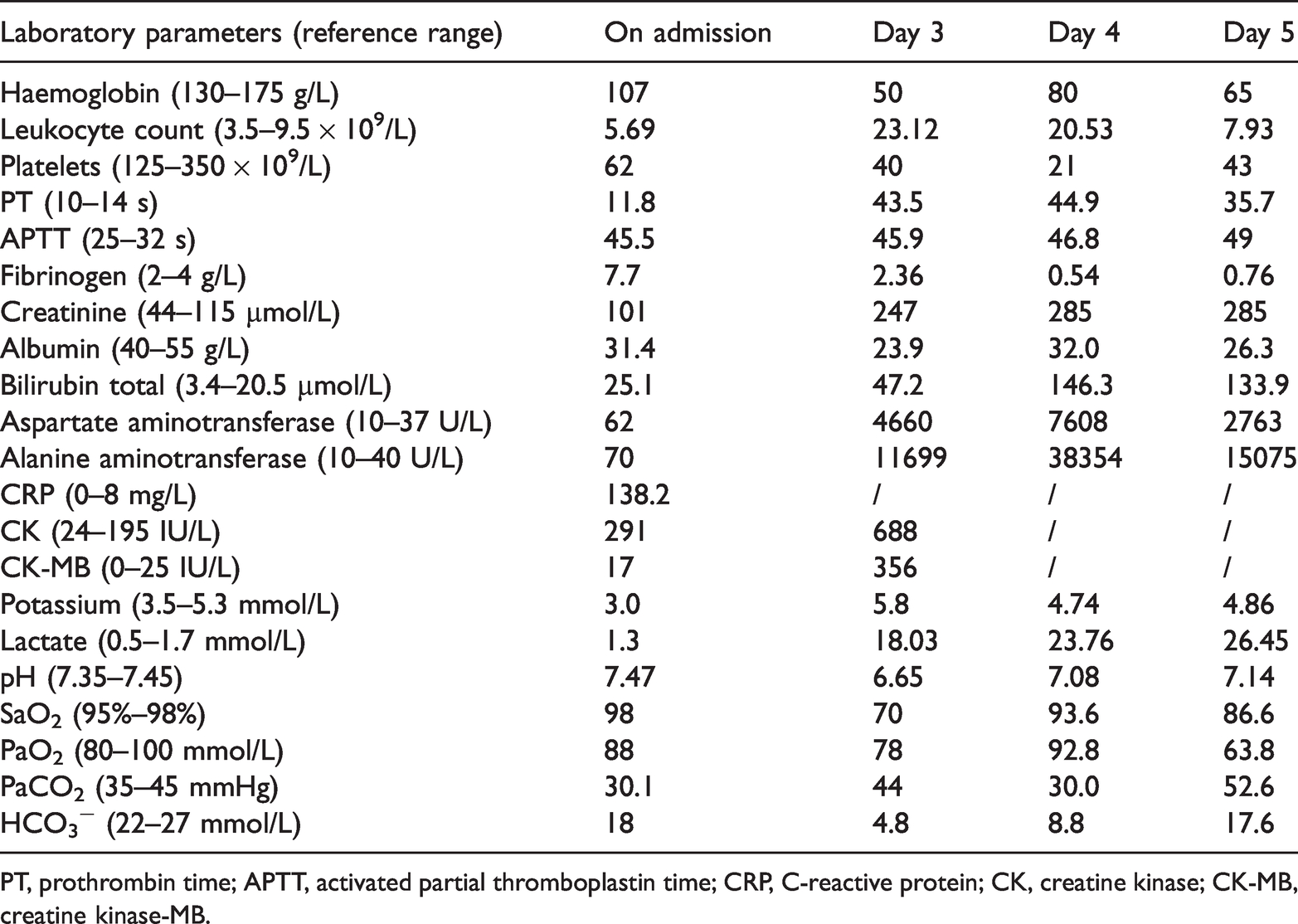
PT, prothrombin time; APTT, activated partial thromboplastin time; CRP, C-reactive protein; CK, creatine kinase; CK-MB, creatine kinase-MB.
Discussion
In this report, we have described a case of pulmonary haemorrhage in a patient with leptospirosis. In the early stage of the disease, the patient presented with chills, fever, conjunctival congestion, and myalgia; he had no jaundice, liver dysfunction, or renal impairment. 8 However, his condition rapidly deteriorated because of multiple organ failure and haemorrhagic shock, and he died soon after despite aggressive treatment including extracorporeal life support. Unfortunately, the diagnosis was delayed in this case, and the treatment of the massive pulmonary haemorrhage was not ideal throughout the patient’s hospitalisation.
Transmission of leptospirosis usually occurs through contact with the urine or tissue of carrier mammals and contaminated water, soil, or vegetation. 9 The morbidity of leptospirosis is low in non-epidemic areas, such as in temperate regions, with only 0.1 to 1.0 cases per 100,000 people each year.10,11 Prior to the present case, no case of leptospirosis had been diagnosed in our unit for more than 10 years. In addition, more than 90% of patients with leptospirosis are anicteric and have mild symptoms that resolve without treatment. Symptoms and signs are nonspecific.11,12 As a result, specific laboratory tests for leptospirosis are not available in many units, and the diagnosis can easily be missed. 13 In the present case, we could not diagnose the leptospiral infection in the early stage of the disease because we had an insufficient understanding of leptospirosis, overlooked the risk factors, and obtained no important diagnostic clues from the routine laboratory tests.
Leptospires can be detected in the blood during the initial stage of the infection. Several techniques are available for the diagnosis of leptospirosis. Leptospires culture is an aetiological test with high specificity; however, it requires a special medium, and growth requires at least 8 to 14 days. 14 This is not very helpful for diagnosis in the acute phase. The microscopic agglutination test (MAT) is the most commonly used method for diagnosing leptospirosis. A four-fold increase in the MAT antibody titre provides definite evidence of leptospiral infection, and this method also has high sensitivity. 15 However, a high antibody titre is required in this method, which means that the MAT may not be advantageous for early diagnosis. 16 Enzyme-linked immunosorbent assay (ELISA) is more sensitive than the MAT for diagnosis during the acute phase of leptospirosis. 17 Leptospiral-specific immunoglobulin M can be detected during the first week of illness, but antibody levels may be low or even undetectable during the early onset of the disease; in other words, this could easily lead to a false-negative diagnosis. 18 Polymerase chain reaction (PCR) does not require serum antibodies and is a reliable and rapid diagnostic tool. 19 It is suitable for early diagnosis of leptospirosis before seroconversion, but with less sensitivity for samples collected later in the infection. 20
NGS does not require isolation or cultivation of pathogens, and it detects pathogens by sequencing microbial nucleic acid fragments in clinical samples. It can detect Leptospira DNA in specimens quickly and accurately during the early stage of the infection.21,22 Fifteen other reports23–37 have described different diagnostic methods for severe leptospirosis requiring extracorporeal membrane oxygenation. As shown in Table 2, PCR, the MAT, ELISA, and NGS can be used to diagnose severe leptospirosis early. PCR has the shortest time from sample collection to diagnosis (average of 2 days). However, whereas the traditional detection method of leptospirosis can only help to confirm the diagnosis, NGS can help to confirm the diagnosis and provide important diagnostic clues for patients with atypical symptoms of leptospirosis. Almost all patients have a convincing transmission source.
Diagnostic methods for severe leptospirosis requiring extracorporeal membrane oxygenation.

M, male; BALF, bronchoalveolar lavage fluid; NGS, next-generation gene sequencing; PCR, polymerase chain reaction; ELISA, enzyme-linked immunosorbent assay; MAT, microscopic agglutination test.
Overall, the clinical symptoms of leptospirosis are nonspecific in the early phase. Even without treatment, most patients recover. 38 Severe pulmonary haemorrhage is rare but can be life-threatening, even with aggressive treatment including extracorporeal life support. 1 Leptospirosis should be considered in patients with unexplained pulmonary haemorrhage. A history of contact with contaminated water or animals can provide important diagnostic clues. NGS is a method that aids in the early diagnosis of leptospirosis.
Consent for publication
Written informed consent was obtained from the patient’s son for the publication of this case report and any accompanying images. A copy of the consent form is available for review by the Editor of this journal.
Footnotes
Acknowledgements
We thank Wei Tong Xiong and Ming Feng Liu from the YvShan People’s Hospital in China for consultation of this case.
Authors’ contributions
Z.C. and W.H.D. designed the study. Z.C. and W.H.D. collected the data. W.H.D. analysed the data and wrote the case report. Z.C. contributed to the discussion of the results and review of the manuscript. Both authors read and approved the final manuscript.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request. Z.C. and W.H.D. will make the data available to readers.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics approval
The requirement for ethics approval was waived because this was not a clinical study and consent for publication was provided by the patient’s son.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Research and Development Project of Jiangxi Province (Social Development Field), China (No. 20202BBGL73059).
