Abstract
Objective
Recurrent aphthous stomatitis (RAS), a common oral mucosal disorder characterized by chronic, inflammatory, and ovoid ulcers, has a complex etiology. The purpose of the study was to investigate the specific dietary factors influencing the prevalence of RAS.
Methods
A total of 754 participants aged 18 to 59 years were enrolled in this descriptive cross-sectional study. An anonymous questionnaire was adopted to investigate the distribution of RAS, dietary factors, self-reported trigger factors, and therapeutic methods.
Results
Among all participants, the prevalence rate of RAS was 21.4%. Univariable analysis showed that fruit, dairy products, vegetables, and water, but not fried foods, fermented foods, spicy foods, and eggs, were preventive factors against RAS. After adjusting for age and sex, multivariable regression analysis suggested that fruit (adjusted odds ratio [aOR] = 0.430, 95% confidence interval [CI] = 0.218–0.847) and water (aOR = 0.294, 95% CI = 0.119–0.726) were protective factors against RAS.
Conclusion
This study found that the consumption of fruit and water was negatively associated with RAS. These results imply a potential adjunctive and complementary role of food in RAS treatment and some feasible means of RAS prevention.
Keywords
Introduction
Recurrent aphthous stomatitis (RAS), or recurrent oral ulceration, is one of the most common oral mucosal disorders, affecting 2% to 66% of the population, according to worldwide epidemiological data. 1 Characterized by multiple small, ovoid, and recurrent ulcers with circumscribed margins, yellowish white or gray floors, and erythematous haloes, RAS can greatly impact the functions of eating, speaking, and swallowing in severe cases.2,3
To elucidate the pathogenesis of RAS, researchers have suggested many influencing factors, including genetic factors, immune dysregulation, and stress,3–6 as well as specific dietary factors.7–19 It has been reported that cow’s milk protein can induce RAS with an increase of anti-fresh cow’s milk IgA, IgG, and IgE antibodies in the serum.7,8 Other studies have suggested that some foods, such as tomatoes, oranges, lemons, and pineapple, can induce a proinflammatory cascade, leading to RAS.11–13 Additionally, deficiencies of vitamin B12, vitamin D, folic acid, ferritin, and hemoglobin may be involved in keratinocyte proliferation and immune imbalance, thereby inducing RAS.15–20 We designed this study to explore the potential associations between RAS and several dietary factors that have been suggested to be potential influence factors in the literature or in clinical settings.7,14
In this cross-sectional survey, we aimed to collect data regarding RAS incidence, dietary factors, self-reported trigger factors, and therapeutic methods to explore the relationships between RAS and dietary factors. This work can potentially encourage further study of the etiology of and prevention measures against RAS in the clinic.
Methods
Sample size, study design, and participants
A sample size of approximately 700 people was determined using the following formula:
This cross-sectional study was conducted from June 2017 to September 2017. We used a multistage random sampling method to select five districts of Nanjing, China. Four neighborhood communities were randomly chosen from each district. Forty civilians who had resided in the city for at least 1 year were selected from each community. The inclusion criteria were being age 18 to 59 years and having no mental disorders, cognitive impairments, communication disabilities, or illiteracy. In line with previous studies,7,21 RAS was diagnosed via self-report using a set of diagnostic criteria for RAS (Tables 1 and 2). Symptoms had to meet all major criteria and at least one minor criterion to be considered a definite diagnosis of RAS. Additionally, we excluded participants with oral local trauma factors and systemic disease (including hematinic deficiencies, Crohn disease, Behçet's disease, auto‐inflammatory disorders, hormonal conditions, diabetes, hypertension, or cardiovascular, renal, gastrointestinal, and liver disease).
Major criteria for RAS

RAS, recurrent aphthous stomatitis.
Minor criteria for RAS

RAS, recurrent aphthous stomatitis.
Participants were asked to complete an anonymous questionnaire, which was developed on the basis of previous studies, with minor modifications. We collected data including demographic information, dietary factors, self-reported trigger factors, and therapeutic methods.22–28 According to Dietary Guidelines for Chinese Residents, 29 daily consumption of fruit, dairy, water, and vegetables was estimated and classified into different levels. Consumption frequencies of other dietary factors were classified into three categories: often (at least 4 times per week), sometimes (1–3 times per week), never, or seldom. Consumption frequencies of eggs were classified into sometimes or often (at least 1 time per week), never, or seldom. In line with Dietary Guidelines for Chinese Residents, 29 fruit included drupe, berry, melon, pome, and citrus fruit; dairy included skim/low-fat and whole milk, condensed milk, milk powder, milk tablets, liquid yogurt, curd yogurt, ice cream, cheese, and butter; water included plain water (bottled water and tap water) and beverages (soft drinks, energy drinks, alcoholic drinks, milk, coffee, juice, tea, and other beverages), but not moisture in foods; vegetables included Cruciferae, Umbelliferae, Leguminosae, Solanum, Chenopodiaceae, and Cucurbitaceae. We excluded those participants who did not complete all questions on the questionnaire.
Statistical analysis
The original data from the pencil-and-paper questionnaire were processed and transcribed in Microsoft Excel (Microsoft Corporation, Redmond, WA, USA) and IBM SPSS 22.0 (IBM Corp., Armonk, NY, USA) was used to set up a database of the results. All categorical variables are described using frequency and percentage. We assessed the validity and reliability of the questionnaire using Cronbach’s α, the Kaiser–Meyer–Olkin (KMO) test, and Bartlett’s test of sphericity. The difference in sex or age among participants with and without RAS was assessed using chi-square analysis. Univariable analysis was performed using the crosstab function in SPSS to obtain preliminary results and screen out variables with little evidence of an association. Variables with a p value < 0.05 in the univariable analysis were included in multivariable regression analysis to evaluate the connection between dietary factors and RAS. Multivariable logistic regression was adjusted for age and sex, and statistical significance was defined as p < 0.05.
Ethics approval
This study followed the STROBE statement guidelines (Text S1) and was approved by the Ethics Committee of Nanjing Stomatological Hospital (2014NL-002 (KS)).
Results
Reliability and validity of the questionnaire
Cronbach’s α for the questionnaire was 0.797, indicating good reliability. The KMO value was 0.883, and the χ2 value in Bartlett’s test of sphericity was 70.598 (p < 0.05), suggesting good validity.
Participant characteristics
A total of 754 participants (336 men and 418 women) aged 18 to 59 years were enrolled in this study, with a RAS prevalence of 21.4% (n = 161). The demographics of the participants are summarized in Table 3. There was no significant difference in sex or age among participants with and without RAS in chi-square analysis.
Participant demographics

RAS, recurrent aphthous stomatitis; OR, odds ratio; CI, confidence interval.
Self-reported trigger factors and therapeutic methods in the RAS group are summarized in Table 4. A total 53.4%, 56.5%, 69.6%, 69.6%, and 78.9% of the RAS group self-reported stress, reduced immune function, irregular life schedule, and unhealthy diet as trigger factors, respectively. The distribution of trigger factors in the various age groups is shown in Figure 1. Nearly half (n = 89, 55.3%) of participants were not undergoing treatment for RAS; 29.2% and 15.5% of participants in the RAS group were using conventional medicine or alternative treatment (AT), respectively.
Self-reported trigger factors and the main therapeutic methods in different age groups

Unhealthy diet: Experienced RAS several hours after consuming high-temperature food, spicy food, fried food, or fermented food.
RAS, recurrent aphthous stomatitis.

Distribution of trigger factors in the different age groups
Associations of dietary factors with RAS
The distribution of RAS prevalence in the groups according to different dietary habits is shown in Figure 2. Table 5 presents preliminary results regarding the relationship between RAS and putative dietary factors in univariable analysis. The findings revealed that RAS was negatively associated with the consumption of fruit (OR = 0.402), dairy (OR = 0.470), water (OR = 0.224), and vegetables (OR = 0.619) (p < 0.05). RAS was not significantly associated with other factors, including fried foods, fermented foods, spicy foods, eggs, sex, or age. The significant variables were all included in the multivariable logistic regression, which was adjusted for age and sex (Table 6 and Figure 3). After adjustment for confounders, multivariable logistic regression revealed that RAS was negatively associated with fruit (adjusted odds ratio [aOR] = 0.430, 95% confidence interval [CI] = 0.218–0.847, p = 0.015) and water (aOR = 0.294, 95% CI = 0.119–0.726, p = 0.008).

Distribution of rates of recurrent aphthous stomatitis (RAS) in groups with different dietary habits, including consumption of fruit, dairy products, and vegetables (A), water (B), and spicy foods, high-temperature foods, fried foods, and fermented foods (C)
Univariable analysis of the relationship between dietary factors and RAS

Hardly ever or never: less than once a week and the consumption is hard to quantify. Never or seldom: less than once a week. Sometimes: 1–3 times per week. Often: at least 4 times per week.
Boldface values are statistically significant with p < 0.05.
RAS, recurrent aphthous stomatitis; CI, confidence interval; OR, odds ratio.
Multivariable logistic regression of the relationship between dietary factors and RAS

Hardly ever or never: food is consumed less than once a week and the consumption is hard to quantify.
aAdjusted for age, sex.
Boldface values are statistically significant with p < 0.05.
RAS, recurrent aphthous stomatitis; CI, confidence interval; OR, odds ratio.
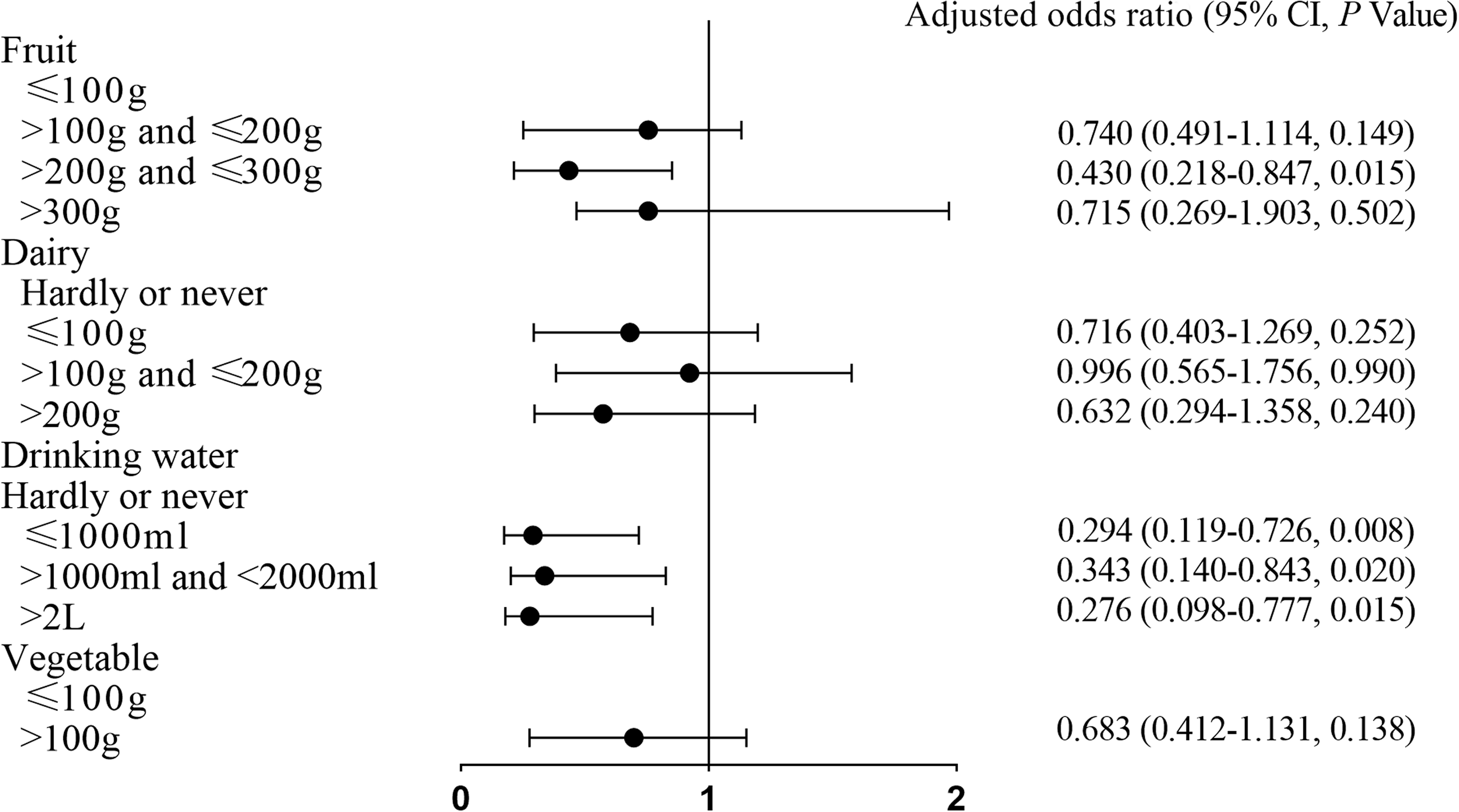
Multivariable logistic regression of the relationship between dietary factors and recurrent aphthous stomatitis
Discussion
In this cross-sectional study, we explored the distribution of RAS, dietary factors, self-reported trigger factors, and therapeutic methods among our study population and found a prevalence of 21.4%, in accordance with the literature.14,30,31 Moreover, fewer than half (72, 44.7%) of participants with RAS had ever received treatment, which was lower than the percentage reported in another study.28,32 Nearly one-third of participants who ever received treatment for RAS mainly used AT versus conventional medicine, despite a lack of randomized controlled trials to prove the effectiveness and safety of AT. Many factors, including the frequency of RAS, can greatly influence patients’ choice of therapeutic methods. Further investigation is needed to explore which factors influence therapeutic method choices and the clinical effects achieved among patients with RAS who use AT.
Univariable analysis revealed that RAS was negatively associated with the consumption of fruit, dairy, water, and vegetables (p < 0.05). RAS was not significantly associated with other factors, including fried foods, fermented foods, spicy foods, and eggs. Zinc, vitamin B12, and probiotics in dairy products may explain the preventive mechanisms of dairy against RAS.17,33–40 Several studies reported that patients with RAS had lower levels of serum zinc as compared with health controls, suggesting a role of zinc in ulcer healing and free-radical scavenging in treating RAS.36–38 Additionally, vitamin B12 has important roles in DNA synthesis and cell division, and vitamin B12 deficiency may induce oral epithelial atrophy in RAS development. 20 Other studies have reported a change in the microbiome in RAS and a treatment effect of a Lactobacillus and Bifidobacterium composition on RAS.39,40 Zinc and selenium in vegetables may explain the preventive mechanisms of consuming vegetables in RAS.40–44 Zinc has been suggested to be involved in the synthesis of protein and collagen, which contribute to wound healing, and selenium supplementation has been associated with a decrement of lipid peroxidation and incidence of infectious complications, as well as better wound healing.10,41–44
In our study, multivariable logistic regression showed that RAS was negatively associated with fruit (aOR = 0.430) and water (aOR = 0.294) (p < 0.05). It is noteworthy that RAS was first found to be associated with the total consumption of fruit and water in this study. Considering the significant decrease in folate levels among patients with RAS compared with health controls reported in several studies, and the capacity of vitamin C to reduce outbreaks and pain levels in cases of minor-type RAS,1,5,18,19 fruit may prevent RAS by supplementing folate and vitamin C. Considering the capacity of calcium to protect the oral mucosa and regulate immune capacity by maintaining tissue polarity,45,46 as well as the capacity of magnesium to reduce both serum nitric oxide and lipid peroxidation to treat ulcers in a mouse model, 47 water may prevent RAS by supplementing calcium and magnesium. Thus, the consumption of certain foods, especially fruit and water, may indirectly influence the risk of RAS by changing the composition of oral microbiota and the blood concentration of nutrients, including vitamin B12, folate, vitamin C, zinc, selenium, calcium, and magnesium.36–47
We acknowledge a bias toward a younger population in our study, which might result from a high percentage of students residing in the chosen neighborhood communities. However, considering that no significant association was found between age and RAS in this study, this bias should not affect the generalizability of our results. Nevertheless, despite our attempts to choose districts representative of different demographic characteristics, our inclusion of participants from only one city leads to some inevitable limitations in terms of generalizability and extrapolation of the results to other urban and rural areas. Additionally, our findings were generated using self-reports, which also led to a lack of the clinical type of RAS. Finally, the variables in this study were general and can be misleading. As nutrients vary greatly among different foods, the effect of these variables on RAS could result from the consumption of certain species of plant foods. Therefore, our results should be interpreted with caution.
Conclusion
The present study findings indicated that consuming fruit and drinking water might be potentially protective against RAS and might be valuable daily preventive measures against RAS. These results could provide new insights into the prevention and treatment of RAS.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211017724 - Supplemental material for Relationship between dietary factors and recurrent aphthous stomatitis in China: a cross-sectional study
Supplemental material, sj-pdf-1-imr-10.1177_03000605211017724 for Relationship between dietary factors and recurrent aphthous stomatitis in China: a cross-sectional study by Kaiyuan Xu, Chongchong Zhou, Fan Huang, Ning Duan, Yanyi Wang, Lichun Zheng, Xiang Wang and Wenmei Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605211017724 - Supplemental material for Relationship between dietary factors and recurrent aphthous stomatitis in China: a cross-sectional study
Supplemental material, sj-pdf-2-imr-10.1177_03000605211017724 for Relationship between dietary factors and recurrent aphthous stomatitis in China: a cross-sectional study by Kaiyuan Xu, Chongchong Zhou, Fan Huang, Ning Duan, Yanyi Wang, Lichun Zheng, Xiang Wang and Wenmei Wang in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Scientific Foundation of China (81870767 & 81570978), the Key Project of Science and Technology Department of Jiangsu Province (BL2014018), the Project of Jiangsu Provincial Medical Youth Talent (QNRC2016118), the Preventive Medicine Project of Jiangsu Province (Y2015004), and the Nanjing Medical Science and Technique Development Foundation (ZKX17033 and YKK16162).
Availability of data and materials
The data supporting this study are available from the corresponding author on reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

