Abstract
Objective
To determine the risk factors for delayed radiographic resolution in children with refractory Mycoplasma pneumoniae pneumonia (RMPP) and explore the most suitable time for interventional bronchoscopy.
Methods
This retrospective study involved 142 children with RMPP who were admitted to our hospital from 1 January 2015 to 31 December 2017. They were divided into a common resolution group and a delayed resolution group based on their chest radiograph series.
Results
Among the 142 patients, 67 showed common resolution on chest radiographs and 75 showed delayed resolution. Independent risk factors for delayed resolution were a clinical course of ≥11.5 days before the performance of interventional bronchoscopy, mucus plug formation, corticosteroid resistance, and atelectasis. When bronchoscopy was performed before the disease had been present for <11.5 days, the length of hospitalization, total fever duration, and duration of time until disappearance of coughing were shorter than those in children who underwent bronchoscopy after the disease had been present for ≥11.5 days.
Conclusions
Corticosteroid resistance, the time to interventional bronchoscopy, atelectasis, and mucus plug formation were associated with delayed resolution on chest radiographs. Performance of interventional bronchoscopy before the clinical course has reached 11.5 days may help alleviate clinical symptoms and improve radiographic resolution.
Introduction
Mycoplasma pneumoniae is a leading cause of community-acquired pneumonia. 1 Infection by M. pneumoniae is usually a benign and self-limiting process and can be effectively treated with macrolides; however, the infection may still progress to refractory M. pneumoniae pneumonia (RMPP). 2 In recent years, the incidence of RMPP has been increasing annually. If children with RMPP are not treated in a timely fashion, serious lung disease may occur along with systemic damage and adverse effects on various extrapulmonary organs, including the digestive, nervous, skin, musculoskeletal, hematological, and renal systems.3–6 These changes may ultimately result in atelectasis, bronchiectasis, bronchiolitis obliterans, and other sequelae that can seriously threaten children’s health.7,8 Chest images of children with RMPP can reveal diverse problems, often displaying inflammatory changes in the lungs and/or lung segments as well as pleural effusion. Systemic corticosteroids have been used in addition to antimicrobial therapy to diminish the host immune response in children with RMPP, demonstrating satisfactory effects.9–11 However, some patients with RMPP do not show improvement of fever when they are treated with systemic corticosteroids and thus require investigation using bronchoscopy.
Previous studies have shown that resolution on chest radiography occurs about 1 to 2 months after disease onset.12–14 We retrospectively evaluated the medical records of children with RMPP diagnosed during hospitalization at our institution. Based on their clinical information and chest images after bronchoscopy, we identified the risk factors for delayed radiographic resolution and the optimal time to introduce interventional bronchoscopy.
Methods
Patients and definitions
This retrospective case-control study was conducted at the pulmonary department of Children’s Hospital of Soochow University. We collected clinical information from the medical records of children diagnosed with RMPP while hospitalized from 1 January 2015 to 31 December 2017. The diagnostic criteria for RMPP were (1) a fever (>38.5°C), paroxysmal dry hacking cough with dyspnea, pulmonary crackles, and pulmonary infiltrates on radiographs; (2) a significant rise in M. pneumoniae immunoglobulin (Ig) G or seroconversion in paired sera, or the presence of IgM together with M. pneumoniae DNA detected in nasopharyngeal aspirates; and (3) a persistent fever (>38.5°C) and radiological deterioration after >1 week of macrolide therapy.15–17 The exclusion criteria for this study were important organ or system dysfunction, evidence of coinfection with other pathogens, and incomplete clinical data.
The indication for bronchoscopic intervention was lobar pneumonia or segmental atelectasis on chest radiographs after 1 week of appropriate antibiotic and corticosteroid treatment. All patients or their legal guardians provided written informed consent before bronchoscopic treatment. This retrospective study was approved by the Ethics Committee of Children’s Hospital of Soochow University (no. 2020CS078), and all patients or their legal guardians provided written informed consent to participate. All methods were carried out in accordance with the format recommended by the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 18
Data collection
Demographic data, clinical information, laboratory examination findings, radiological manifestation, and bronchoscopic findings were retrospectively collected from the records of all children. Laboratory parameters included the white blood cell count, C-reactive protein concentration, lactate dehydrogenase concentration, erythrocyte sedimentation rate, and D-dimer concentration. Nasopharyngeal aspirates were routinely collected within 24 hours of admission. Microbiologic tests were performed to exclude other respiratory tract infections, including nasopharyngeal aspirates for common respiratory tract virus antigens, respiratory syncytial virus, influenza virus A, influenza virus B, parainfluenza virus 1, parainfluenza virus 2, parainfluenza virus 3, human rhinovirus, human metapneumovirus, human bocavirus, and adenovirus.
Chest radiographs taken 3 weeks after bronchoscopy were reviewed. Delayed radiographic resolution was defined as a <50% absorption rate on chest radiographs 3 weeks after bronchoscopy. Common radiographic resolution was defined as a ≥50% absorption rate on chest radiographs 3 weeks after bronchoscopy.
Corticosteroid resistance was defined as a persistent fever lasting >72 hours after a regimen of intravenous methylprednisolone at 2 mg/kg/day.
Diagnostic tests for M. pneumoniae
Nasopharyngeal swabs were obtained within 1 day of admission. The specimens were centrifuged and stored at −80°C. A quantitative diagnostic kit for M. pneumoniae DNA (Da An Gene Co., Ltd., Guangzhou, China) was used to measure the load of M. pneumoniae. The method is based on TaqMan polymerase chain reaction (PCR) technology, and the target is the 16S rRNA gene specific for the M. pneumoniae genome. Briefly, 1 mL of nasopharyngeal aspirate diluted by 4% sodium hydroxide was centrifuged at 12,000 rpm for 5 minutes. The sediment was collected, washed twice with 0.9% sodium chloride, blended with 50 μL of DNA extraction solution, incubated at 100°C for 10 minutes, and centrifuged at 12,000 rpm for 5 minutes. Real-time PCR was performed on the resulting supernatant of 2 μL with 43 μL of PCR mix (supplied with the kits) using the DA 7600 real-time PCR system (Applied Biosystems, Foster City, CA, USA) as follows: 93°C for 2 minutes, 10 cycles at 93°C for 45 s and 55°C for 60 s, followed by 30 cycles at 93°C for 30 s and 55°C for 45 s.
Specific IgM and IgG antibodies to M. pneumoniae were detected in serum samples of patients in the acute phase of MPP (on admission) and in the convalescent phase (on discharge), respectively, using a commercial enzyme-linked immunosorbent assay (ELISA) kit (Serion ELISA classic MP IgG/IgM; Institute Virion/Serion, Würzburg, Germany) according to the manufacturer’s instructions.
Statistical analysis
IBM SPSS Statistics for Windows, Version 25.0 (IBM Corp., Armonk, NY, USA) was used for all statistical analyses. We conducted a normal distribution test on the data. An independent-samples t test was used for data with a normal distribution, and a non-parametric test was used for data with a non-normal distribution. Count data were analyzed using the χ2 test, with the results expressed as percentages. Multivariate logistic regression analysis for 21 influential factors was performed. The variables identified by univariate analysis (p < 0.05) were entered into the multivariate regression model to identify independent risk factors for delayed resolution on chest radiographs.
Results
Clinical characteristics
In total, 5020 patients with MPP were identified during the study period, including 562 (11.2%) patients with RMPP. Among these, 142 patients with RMPP were included in the present study. The common resolution group comprised 67 (47.2%) patients, and the delayed resolution group comprised 75 (52.8%) patients. The peak body temperature of the children in the common resolution group was 39.5°C ± 0.6°C lower than that of the children in the delayed resolution group (P < 0.05). The total duration of fever was longer in the delayed resolution group (median, 10.0 days; interquartile range, 7.0–13.0 days) than in the common resolution group (median, 7.0 days; interquartile range, 6.0–10.0 days) (P < 0.01). There were 8 patients with corticosteroid resistance in the common resolution group and 30 patients in the delayed resolution group, with a statistically significant difference (P < 0.01) (Table 1).
Clinical characteristics and laboratory indicators in children in the common and delayed resolution groups.
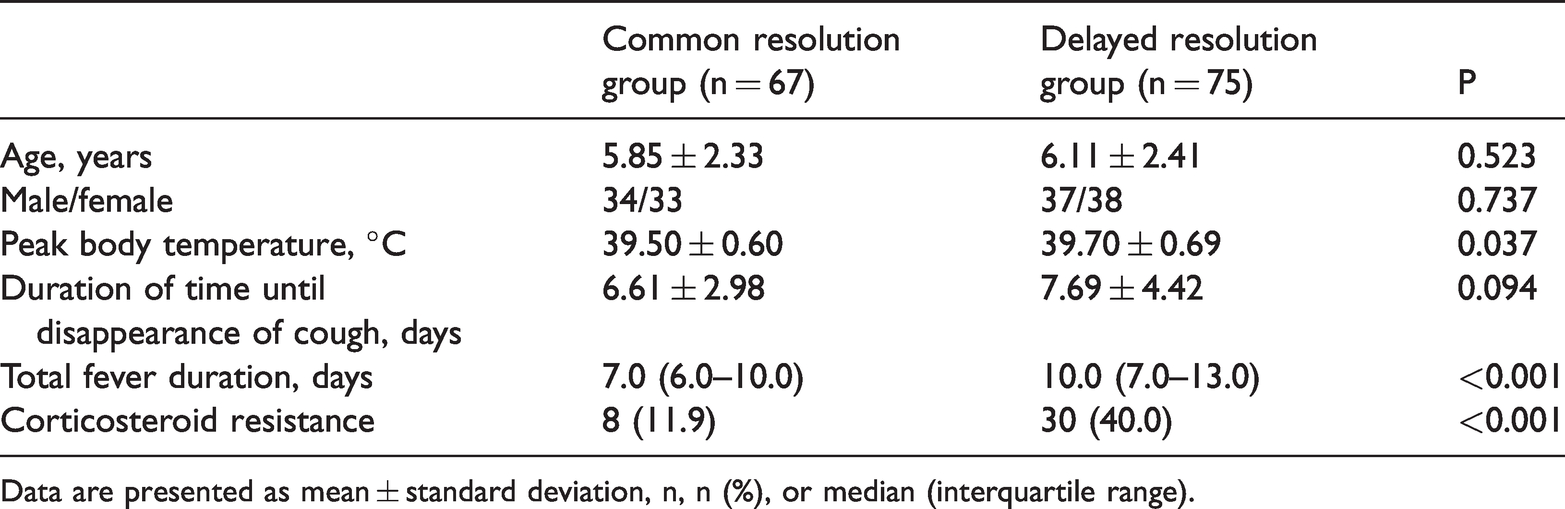
Data are presented as mean ± standard deviation, n, n (%), or median (interquartile range).
Laboratory findings
The laboratory findings in the common and delayed resolution groups are shown in Table 2. The lactate dehydrogenase and D-dimer concentrations were higher in the delayed resolution group. There were no significant differences in the white blood cell count, C-reactive protein concentration, erythrocyte sedimentation rate, IgA, IgG, IgM, or lymphocyte subpopulations between the two groups.
Laboratory values in children in the common and delayed resolution groups.

Data are presented as median (interquartile range).
WBC, white blood cell; CRP, C-reactive protein; LDH, lactate dehydrogenase; ESR, erythrocyte sedimentation rate; Ig, immunoglobulin.
Radiographic and bronchoscopic findings
Atelectasis was present in 2 (3.0%) patients in the common resolution group and in 18 (24.0%) patients in the delayed resolution group, with a statistically significant difference (P < 0.01). However, there was no significant difference in the incidence of pleural effusion or lung consolidation between the two groups. The incidence of mucus plug formation was significantly higher in the delayed resolution group (n = 36 (48.0%)) than in the common resolution group (P < 0.01). However, the mucus plugs were successfully cleared after bronchoscopy (Figure 1).
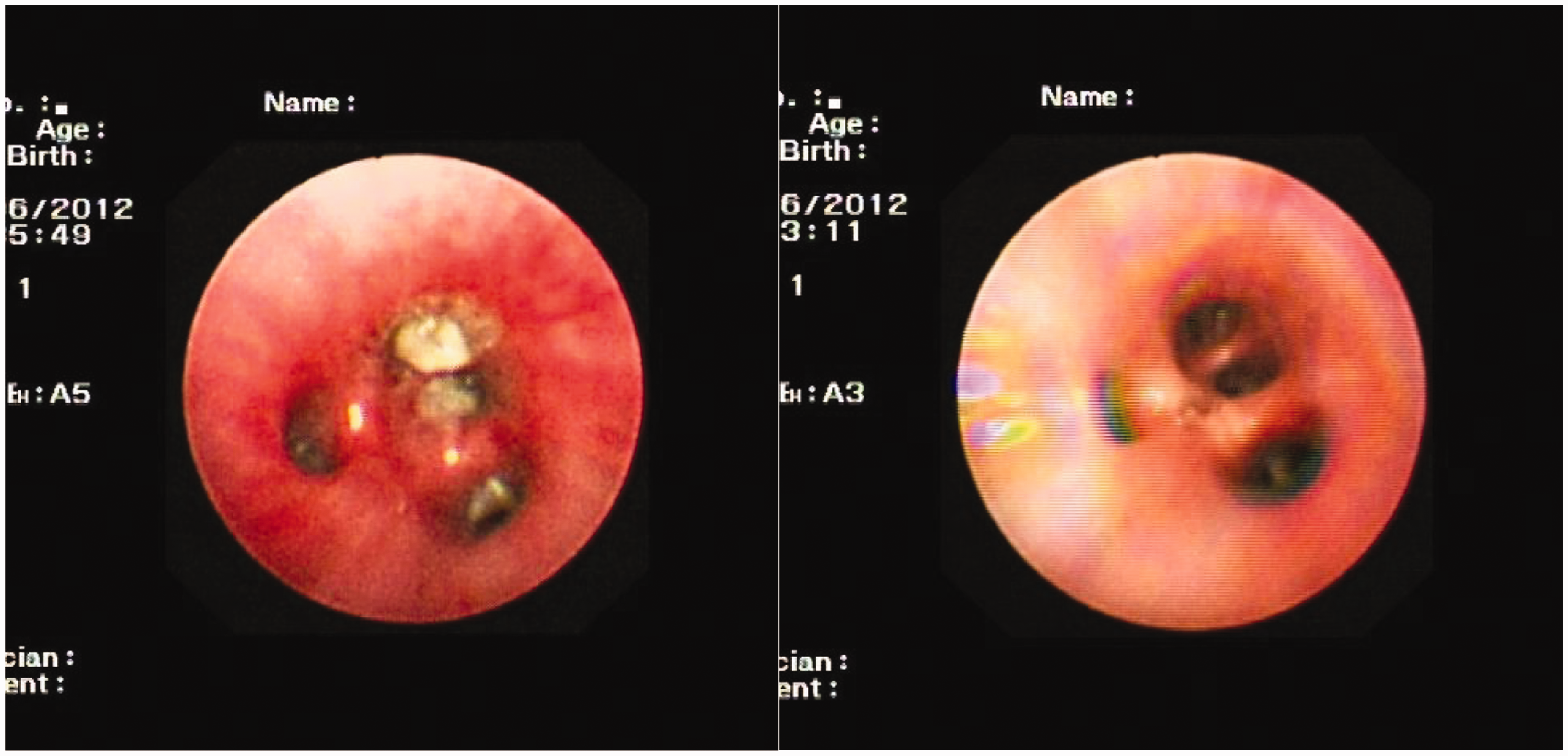
Bronchoscopic findings before and after bronchoscopic treatment.
Predictive risk factors for delayed resolution on chest radiographs
Univariate analysis identified eight variables that were significantly different between the two groups: peak body temperature, duration of time until performance of interventional bronchoscopy, total fever duration, corticosteroid resistance, lactate dehydrogenase concentration, D-dimer concentration, atelectasis, and mucus plug formation (P < 0.05). Multivariate logistic regression analysis showed that corticosteroid resistance, the duration of time until interventional bronchoscopy, atelectasis, and mucus plug formation were independent risk factors for delayed radiographic resolution in children with RMPP. A receiver operating characteristics (ROC) curve was used to determine the optimal time to perform bronchoscopy. We used “resolution” as the state variable to perform the ROC curve analysis for determination of the critical value. We then took the maximum sensitivity and specificity points in the ROC curve and determined that the critical time for interventional bronchoscopy was 11.5 days, with specificity and sensitivity of 0.681 and 0.610, respectively. We performed a logistic regression analysis and found that all the indicators were statistically significant. We scored independent risk factors according to the following odds ratio values: 1 point for a ≥11.5-day duration until bronchoscopic intervention and mucous plug formation, and 2 points for hormone insensitivity and atelectasis (Table 3). We divided the patients into two risk groups based on the total score: a high-risk group (4–6 points) and a low-risk group (0–3 points). The scores of independent risk factors indicated that the resolution rate gradually decreased as the total score increased (Figure 2).
Multivariable predictors of delayed resolution on chest imaging using the predicted scores.

OR, odds ratio; CI, confidence interval.
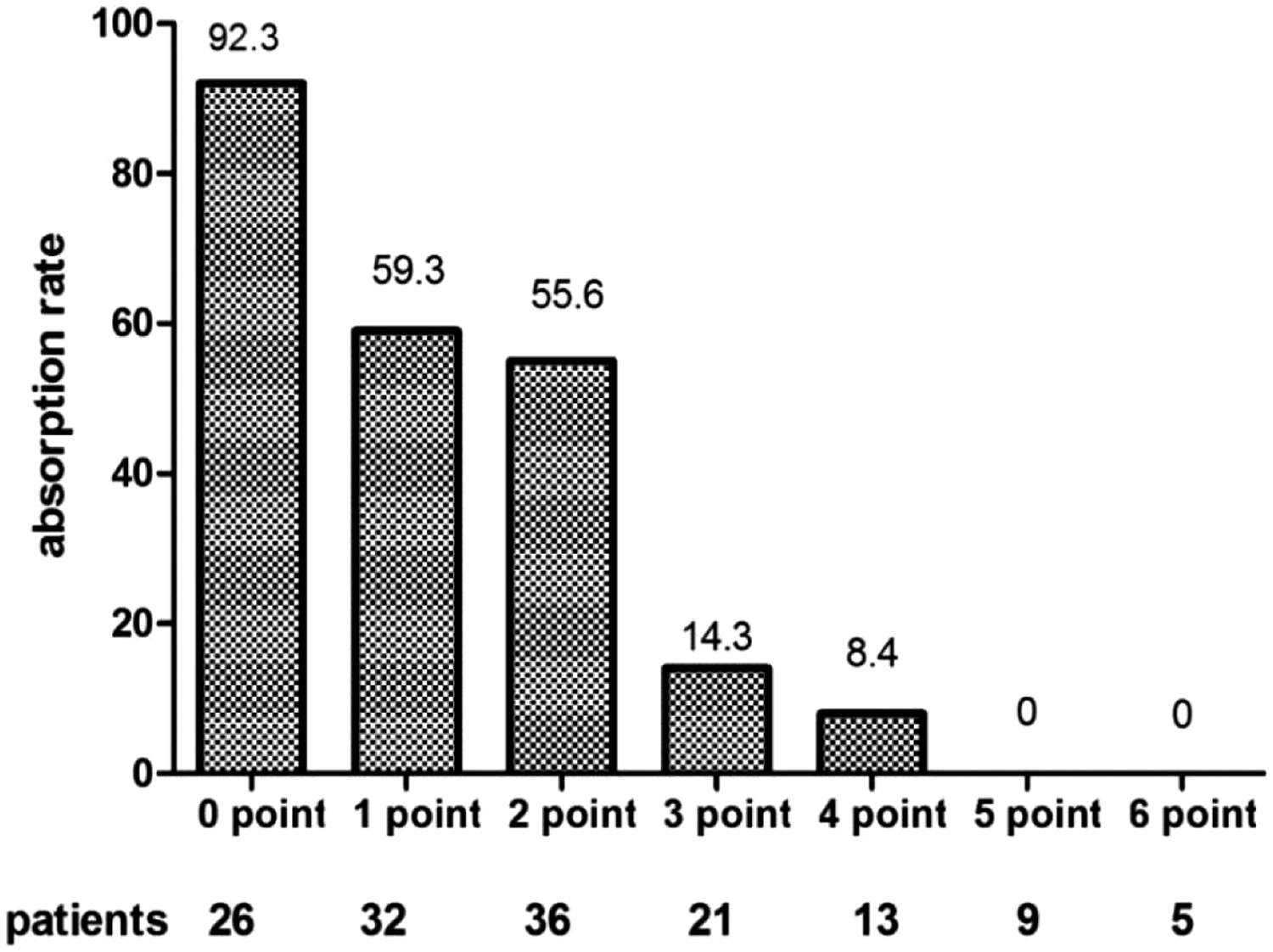
Simple scoring model evaluated for correspondence with delayed resolution.
Discussion
To the best of our knowledge, this is the first study to focus on the most suitable time to perform interventional bronchoscopy in children with RMPP. The performance of interventional bronchoscopy before the clinical course has reached 11.5 days is helpful for alleviating clinical symptoms and improving resolution on chest radiographs.
After 2000, MPP epidemics were observed in 2006, 2010, 2011, and 2012. The epidemics in 2011 and 2012 were particularly widespread and attracted public attention. 16 Large increases in the numbers of patients with MPP were also reported in Europe and other countries during this period.19,20 The number of patients increased again beginning in the summer of 2015 and reached a higher level during the winter. 16 An increase in the number of patients with MPP was also reported in China in 2015. 21 In recent years, the increasing numbers of patients with RMPP have attracted attention from experts worldwide. Complete statistical data on the incidence of RMPP are not available. One study showed that the incidence of RMPP among patients with MPP was 14.30%. 2 The incidence of RMPP in children aged <4 years was 6.83%, that in children aged 4 to 7 years was 20.86%, and that in children aged ≥7 years was 40.84%; thus, the incidence of RMPP increased with age. 2 Even after administering macrolide antibiotics to children with RMPP, the high fever often remains, along with worsening of the disease and damage to multiple organs and systems. When chest imaging shows an abnormality in children with MPP, the existence of RMPP should be considered. 22
Children with MPP display various chest radiological findings, including bronchial pneumonia, pulmonary interstitial lesions, and segmental or lobar changes. The numbers of cases of MPP with accompanying lobar pneumonia have increased in recent years. Studies have shown that the presence of large lobar changes in the lungs is an important risk factor for multiple complications and sequelae in children. 23 Zhang et al. 24 found a 10% incidence of atelectasis in children with MPP at the 1-year follow-up. The main sequelae in such patients included occlusive bronchiolitis and bronchiectasis. Obvious chest imaging abnormalities often lag behind the clinical symptoms in children. One study of 42 children with MPP using lung computed tomography for 10 to 15 days showed that more than half of the children had pulmonary inflammatory resolution. 12 Our study was a retrospective analysis; therefore, the time points at which the children’s chest images were re-examined were inconsistent. We divided the children into a common resolution group and a delayed resolution group according to their chest imaging findings 3 weeks after bronchoscopy, and we found no significant difference in the times at which the chest radiograph examinations were conducted between the two groups. Among the 142 children, 67 showed improvement after 3 weeks and 75 did not exhibit absorption. Eighteen patients in the delayed resolution group had atelectasis, whereas only 2 in the common resolution group had atelectasis (P < 0.01). Hence, atelectasis was an important risk factor that was revealed by chest imaging.
Some studies have used bronchoscopy to analyze mucus plug formation, a high-risk factor, and revealed that children with mucous plug formation have a high peak body temperature and prolonged fever. 25 Other studies have shown that the formation of mucus or mucus plugs can lead to poor bronchial ventilation, which portends a poor prognosis if active treatment is not instituted. 26 We found that the incidence of mucus plugs in the delayed resolution group was 48.0%, which was higher than that in the common resolution group (20.9%). Logistic regression analysis showed that mucus plug formation was an independent risk factor that was revealed by chest imaging. Corticosteroid resistance during treatment is another important factor. In one study, the proportion of patients with pleural effusion was higher in the corticosteroid-resistant group than in the corticosteroid-sensitive group. 27 The pleural effusion usually appeared in moderate to large amounts, which indicates that patients with corticosteroid resistance might develop more serious pulmonary complications. 27 Our study findings are consistent with those results.
Infection by M. pneumoniae can disrupt the balance of the body’s normal T-lymphocyte subsets, leading to immune disorders. T lymphocytes mainly engage in the cellular immune response by releasing cytokines to recognize antigens. Studies have shown that RMPP is not only related to T-lymphocyte subsets but is also closely related to the function of the humoral immune response.28,29 The immune complexes in serum produce humoral immunity in children with MPP. MP antigen is partially similar to antigens of the smooth muscle tissue in the human brain, heart, liver, lung, and kidney. Upon the development of MPP in children, specific antibodies such as IgM, IgA, and IgG appear in the body, which can induce the corresponding tissues of the body to produce antibodies to form an immune complex. This also results in multisystem damage throughout the body. 30 In the present study, there was no significant difference in IgA, IgG, IgM, or the lymphocyte subpopulations between the two groups. In addition, research has shown that atopy is an important cause of RMPP.31,32 Bao et al. 32 found that atopy may be a risk factor for the presence and severity of RMPP because of the high pathogen load in the airways. Another study showed that patients who developed M. pneumoniae-related extrapulmonary disease showed significantly higher levels of IgE than children with non-M. pneumoniae-related respiratory disease and children with M. pneumoniae-related respiratory disease. 33 In the present study, IgE was not a routine laboratory examination and was therefore not analyzed. Further analyses that include IgE measurement are needed.
The present study showed that corticosteroid resistance, the duration of time until performance of interventional bronchoscopy, atelectasis, and mucus plug formation were independent risk factors for delayed resolution on chest radiographs in children with RMPP. We scored the independent risk factors according to the odds ratio and found that the resolution rate gradually decreased as the total score increased. The rate of lung lesions that appeared after bronchoscopy was also predicted by this scoring system.
No relevant reports have indicated the best time to perform interventional bronchoscopy. Some have reported conducting chest imaging within 1 month after bronchoscopy in children with MPP. In one study, on the basis of whether chest imaging showed complete absorption, interventional bronchoscopy was performed at 15.5 days with a specificity and sensitivity of 0.857 and 0.563, respectively. 34 Our ROC curve of the intervention time based on whether the chest radiographs showed resolution indicated that the cutoff time for interventional bronchoscopy was 11.5 days. The length of hospitalization, total fever duration, and duration of time until disappearance of coughing were shorter among children who underwent interventional bronchoscopy before the disease had run its course for 11.5 days than in those who underwent bronchoscopy after 11.5 days. Additionally, the resolution rate on chest radiographs was higher in children who underwent bronchoscopy within 11.5 days.
The present study has several limitations. First, this was a retrospective study with inherent limitations on complete data collection; therefore, further prospective studies are needed. Second, some patients might have had coinfection with other pathogens that could not be detected, potentially leading to longer resolution on chest imaging. Finally, macrolide resistance may have also affected the resolution rate on chest radiographs; however, we lacked relevant data with which to determine this, and further analysis is needed.
Conclusions
Our research indicates that the most advantageous time to perform interventional bronchoscopy is within the initial 11.5-day period of the disease course. Hormone insensitivity, atelectasis, and mucus plug formation are independent risk factors for delayed resolution on chest radiographs in children—a higher score is associated with a lower resolution rate. Nevertheless, the optimal time for interventional bronchoscopy needs further study.
Footnotes
Authors’ contributions
Conception and design: CL Hao, QL Yan. Administrative support: CL Hao. Provision of study materials or patients: All authors. Collection and assembly of data: All authors. Data analysis and interpretation: CL Hao, QL Yan, WJ Jiang. Manuscript writing: All authors. Final approval of manuscript: All authors.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors received the following financial support for the research, authorship, and/or publication of this article: This work was supported by the 2020 Youth Research Fund Project of Children’s Hospital of Wujiang District, Suzhou (funding number 2020QN01).
