Abstract
Objective
To evaluate the efficacy and safety of radiofrequency ablation for the treatment of knee osteoarthritis.
Methods
A literature review was conducted using the PubMed, Cochrane Review, Embase, and Google Scholar databases. Two reviewers independently assessed the eligibility of all retrieved studies. The research was reported based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines to ensure the reliability and verity of the results. The statistical analysis was performed using STATA version 13.0.
Results
Nine randomized controlled trials were collected for the data extraction and meta-analysis. Significant differences in the pain score at 4, 12, and 24 weeks were found between patients treated with radiofrequency ablation and those treated with placebo. Furthermore, the use of radiofrequency ablation was associated with an improved outcome of the Western Ontario and McMaster Universities Arthritis Index at 4, 12, and 24 weeks. No serious adverse events were observed in any patients who underwent radiofrequency ablation.
Conclusion
Radiofrequency ablation is efficacious and safe for reducing pain and improving knee function in patients with knee osteoarthritis, without increasing the risk of adverse effects.
Introduction
Knee osteoarthritis (OA) is a slowly progressive, disabling joint disorder that may cause damage to the hyaline cartilage and subchondral bone. 1 It is most frequently found in the elderly, with a relatively high prevalence of about 40% among people aged 70 to 74 years. 2 The number of patients with knee OA has increased in tandem with the aging of the general population. Total knee arthroplasty is considered the treatment of choice for end-stage knee OA and can provide excellent postoperative pain relief, remarkable deformity correction, and satisfactory functional recovery.3,4 However, not all patients are appropriate candidates for this treatment because of age, comorbidities, or other factors.
Current treatments concentrate on symptom remission with the aim of pain relief and functional recovery. Various nonsurgical treatments have been used for knee OA, including oral nonsteroidal anti-inflammatory drugs, intraarticular injections of hyaluronic acid and platelet-rich plasma, extracorporeal shockwave therapy, and local infiltration of analgesics.5–8 However, most patients experience continuing knee pain and undesirable side effects of these treatments.
Radiofrequency ablation (RFA) was first described in 1891. 9 The pulse generator of RFA creates an electromagnetic field surrounding the electrode tip that activates adjacent molecules, thus generating frictional heat. 10 It is currently used to improve joint function and relieve pain by destroying nerves that innervate painful tissue or by disturbing the transmission of pain signals. RFA is reportedly a reliable method for the management of chronic knee pain related to knee OA. Its noninvasive nature and low complication rate make it a more advantageous technique than other conservative treatments. However, despite the popularization of RFA, there is a lack of reliable evidence to support its use. Therefore, we performed a meta-analysis of randomized controlled trials (RCTs) to determine the efficacy and safety of RFA in treating knee OA.
Materials and methods
Study selection
A literature review was conducted using the PubMed, Cochrane Review, Embase, and Google Scholar databases. The articles identified as relevant to our study were subsequently reported in line with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, and the meta-analysis was registered at Research Registry (identification no. researchregistry1099). The search was independently performed by two researchers using the following key words: “radiofrequency ablation,” “neurotomy,” “knee,” and “osteoarthritis.” The reference lists of reviews and selected studies were also manually searched to identify additional articles.
The requirement for ethics approval was waived because of the nature of this study (literature review).
Eligibility criteria and study selection
Two independent reviewers assessed the eligibility of all of studies retrieved from the databases. Any disagreement between the reviewers was resolved through discussion or by consultation with a third reviewer. A study was included if it (1) was an RCT, (2) compared RFA with placebo for the treatment of knee OA, and (3) included outcome measurements such as pain scores, the Western Ontario and McMaster Universities Arthritis Index (WOMAC) score, range of motion, Lequesne index score, and adverse effects. The exclusion criteria were as follows: (1) randomized studies without a control group; (2) incomplete data and an unclear outcome; (3) abstracts, case reports, conference presentations, editorials, and expert opinions; and (4) studies without available data.
Data extraction
All data were extracted from article texts, tables, and figures by two independent investigators. The following data were obtained: first author, publication year, country of origin, study design, sample size, patients’ demographics, intervention methods, length of follow-up, and clinical outcomes. The clinical outcomes included the pain scores at different time points, WOMAC score, Lequesne index score, and adverse effects. The corresponding author was contacted to request any missing data. Any disagreement between the reviewers was resolved by a third reviewer.
Quality assessment
Two independent reviewers used the Cochrane risk-of-bias tool to evaluate the risk of bias of the included RCTs. 11 The quality of each RCT was assessed using the following seven items: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other bias. Disagreement was resolved through discussion and consensus between the reviewers. Based on the information obtained from the included studies, each item was recorded as low risk of bias, high risk of bias, or unclear (lack of information or unknown risk of bias). A funnel plot was used to assess publication bias. If publication bias was present, the funnel plot was asymmetric. The evidence grade was assessed using the guidelines of the Grading of Recommendations Assessment, Development and Evaluation (GRADE) system. 12 The evidence grades were divided into the following categories: (1) high: further research is very unlikely to change confidence in the estimate of effect, (2) moderate: further research is likely to have an important impact on confidence in the estimate of effect and may change the estimate, (3) low: further research is very likely to have an important impact on confidence in the estimate of effect and is likely to change the estimate, and (4) very low: any estimate of effect is very uncertain.
Statistical analysis
The risk difference with 95% confidence interval (95% CIs) was calculated for dichotomous data, and the weighted mean difference (WMD) with 95% CI was calculated for continuous data. Heterogeneity between the studies was assessed by the χ2 test (significance level of P < 0.10) and the I2 statistic (I2 > 50% indicating significant heterogeneity). The results were pooled using a fixed-effects model for P > 0.10 and I2 < 50% or a random-effects model for P < 0.10 and I2 > 50%. Publication bias was assessed using the symmetric construction of a funnel plot. The statistical analysis was performed using STATA version 13.0 (Nordic Cochrane Center, Cochrane Collaboration, Copenhagen, Denmark).
Results
Search results
In total, 226 relevant citations were identified from the databases and another 4 publications were found through an additional manual search of the reference lists. Of these 226 articles, 197 were excluded because of duplication and 24 were excluded because they met the exclusion criteria. Finally, nine articles published from 2010 to 2020 were included in the meta-analysis.13–21 The detailed collection process is shown in Figure 1.

Flow diagram of study selection.
Characteristics of included RCTs
A total of 802 patients took part in this meta-analysis (404 treated with RFA and 398 treated with placebo). The patients in the intervention group underwent RFA for pain management, and the patients in the placebo group were treated with traditional analgesics. The patients’ average ages ranged from 57 to 70 years, and the follow-up period ranged from 12 to 24 weeks. Details of the included studies are presented in Table 1.
Characteristics of the included studies.

RCT, randomized controlled trial; RFA, radiofrequency ablation; RF, radiofrequency.
Quality assessment
Figure 2 shows the details of the risk-of-bias assessment for all studies. All articles stated the specific way in which the random allocation was applied. The allocation concealment of two studies was not adequately illustrated. Three RCTs used double-blinding of participants and surgeons, and two RCTs attempted to blind clinical assessors. All studies demonstrated a low risk of selective reporting and incomplete outcomes. Selective reporting and incomplete outcomes were identified as a low risk of bias. The risk of bias for each item was expressed in terms of the percentage across all of the included articles, which indicated the proportion of risk levels for each item (Figure 3).
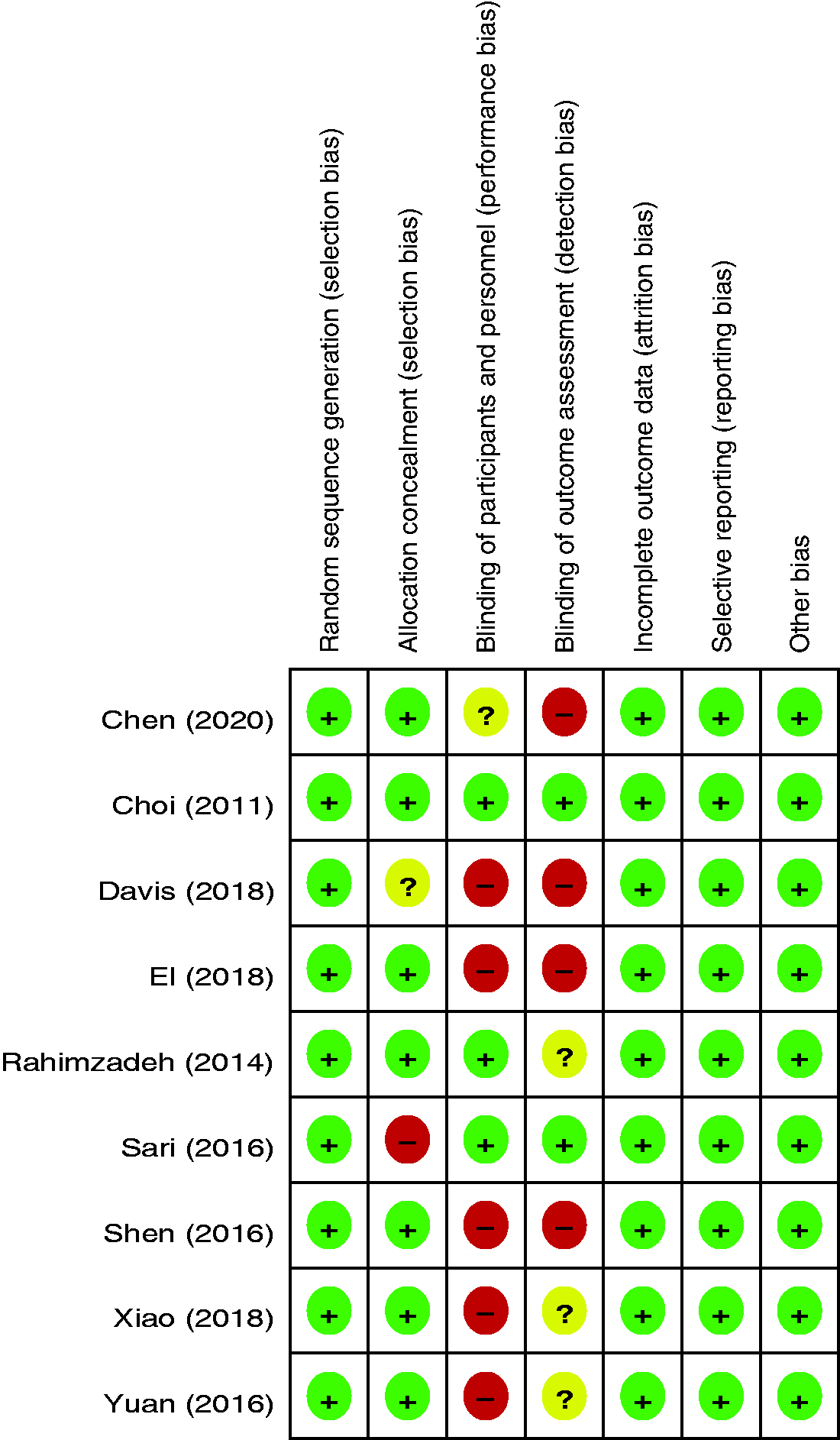
Risk-of-bias summary.

Risk-of-bias graph.
Outcomes of meta-analysis
Pain score at 4 weeks
Six RCTs provided data on the pain score at 4 weeks. There was no significant heterogeneity, and a fixed-effects model was applied (I2 = 0%). The studies showed a significant difference in the pain score at 4 weeks between the two groups (WMD, −0.503; 95% CI, −0.670 to −0.335; P < 0.001) (Figure 4).
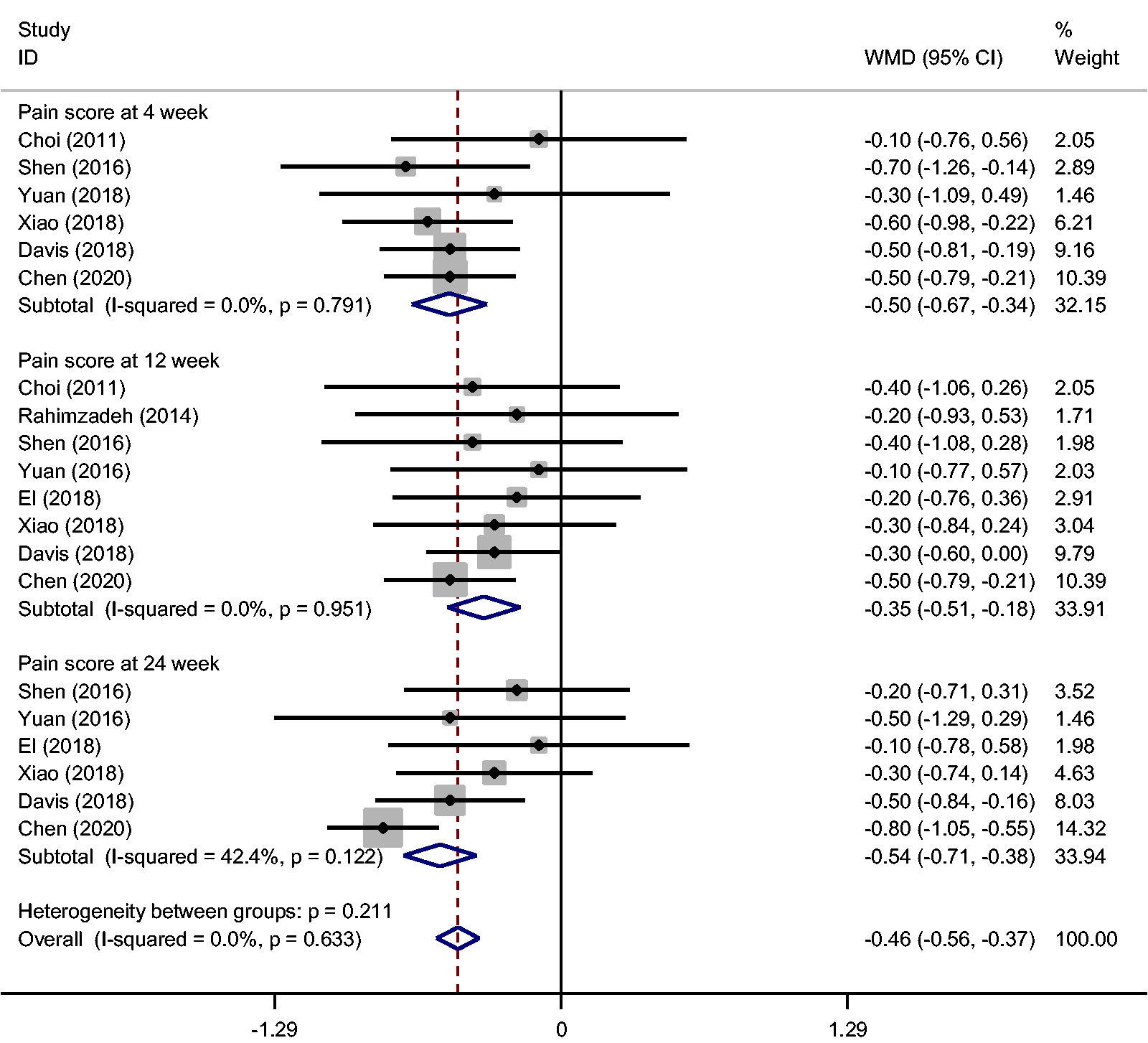
Meta-analysis of pain score.
Pain score at 12 weeks
Eight RCTs reported the pain score at 12 weeks. Based on the available data, the pooled results exhibited no significant heterogeneity (I2 = 0%), and a fixed-effects model was used. The present meta-analysis indicated that the use of RFA was associated with improved pain relief at 12 weeks (WMD, −0.348; 95% CI, −0.511 to −0184; P < 0.001) (Figure 4).
Pain score at 24 weeks
Six RCTs provided data for the pain score at 24 weeks. There was no significant heterogeneity (I2 = 42.4%), and a fixed-effects model was used. The pooled results showed a significant difference between the two groups (WMD, −0.545; 95% CI, −0.708 to −0.382; P < 0.001) (Figure 4).
WOMAC score at 4 weeks
Five RCTs reported the mean change from baseline in the WOMAC score at 4 weeks. The pooled results indicated that the mean change in the WOMAC score was significantly superior in the RFA group than in the control group (WMD, −3.231; 95% CI, −5.741 to −0.721; P = 0.012) (Figure 5). No statistical heterogeneity was found in the mean change in the WOMAC score at 4 weeks (I2 = 0%).

Meta-analysis of Western Ontario and McMaster Universities Arthritis Index.
WOMAC score at 12 weeks
Five RCTs evaluated the mean change from baseline in the WOMAC score at 12 weeks. Significant differences were found between the RFA and control groups (WMD, −4.526; 95% CI, −7.100 to −1.953; P = 0.001) (Figure 5). A fixed-effects model was used because of the lack of statistical heterogeneity identified through the meta-analysis (I2 = 0%).
WOMAC score at 24 weeks
Five RCTs provided data on the WOMAC score at 24 weeks. There was no significant heterogeneity, and a fixed-effects model was applied (I2 = 0%). The studies indicated a significant difference in the WOMAC index at 24 weeks between the two groups (WMD, −2.986; 95% CI, −5.125 to −0.847; P = 0.006) (Figure 5).
Lequesne index
The Lequesne index score was available in three studies. Based on the available data, the pooled results exhibited significant heterogeneity (I2 = 84.8%, P = 0.001), and a random-effects model was used. The present meta-analysis indicated that the use of RFA was associated with an improved Lequesne index score (WMD, −2.064; 95% CI, −3.466 to −0.663; P = 0.004) (Figure 6).

Meta-analysis of Lequesne index.
Adverse effects
Five RCTs reported the adverse events after completion of the treatment. There was no significant heterogeneity among the studies (I2 = 0%), and a fixed-effects model was adopted. No significant differences in terms of adverse events were found between the two groups (risk difference, −0.010; 95% CI, −0.036 to 0.016) (Figure 7).

Meta-analysis of adverse effects.
Quality of evidence and recommendation strengths
The quality of evidence for each of the outcomes was high to moderate. Therefore, we concluded that the overall quality of evidence was moderate. This indicates that the true effect is likely to be close to the estimate of the effect, but there is a possibility that it could be substantially different (Table 2).
Quality of evidence and recommendation strengths.

RCT, randomized controlled trial; WOMAC, Western Ontario and McMaster Universities Arthritis Index; WMD, weighted mean difference; CI, confidence interval; RD, risk difference.
Publication bias
As illustrated by the funnel plots of the pain score at 24 weeks, the scatter points were basically symmetrical (Figure 8). However, publication bias in literature reviews is unavoidable and difficult to overcome. This should be taken into consideration when analyzing the results.
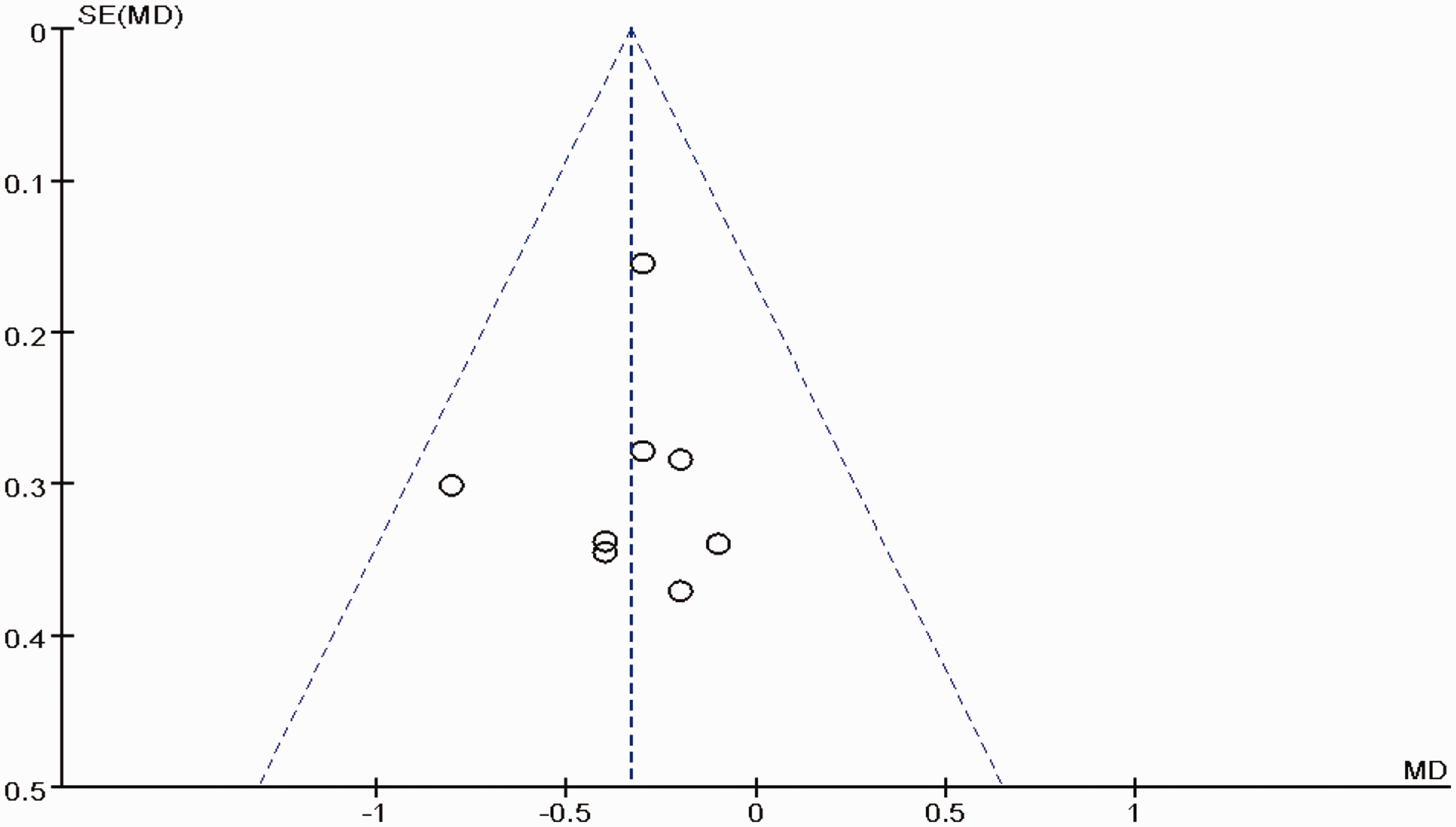
Publication bias.
Discussion
To the best of our knowledge, this is the first meta-analysis of RCTs to evaluate the efficacy and safety of RFA for treating knee OA. The most important finding of the study is that the use of RFA was associated with improvements in pain relief and the WOMAC score. There were significant differences in the Lequesne index score between the two groups. Furthermore, no serious adverse effects were observed in any of the patients who underwent RFA treatment.
Knee OA is a major cause of pain and locomotor disability worldwide; the pain is chronic and restricts the function of the knee joints. Age is a major risk factor for knee OA. Given the increasing prevalence and incidence of OA, it is now considered a major worldwide public health problem. It is predicted that 67 million people in the United States will have been diagnosed with OA by the year 2030. 22 The weight-bearing joints, such as the knees and hips, are the most frequently affected. Effective pain management plays a vital role in treating OA and improving the prognosis. Conservative treatment is the first choice for early-stage OA and may involve physical therapy, intra-articular injections (platelet-rich plasma, glucocorticoids, glucosamine, and hyaluronic acid), and oral anti-inflammatory drugs.
The concept of RFA was introduced in 1891, and the technique has since been used to treat cancer, cardiac arrhythmia, varicose veins, and other diseases.23,24 In recent years, its use has been extended to the treatment of chronic pain, such as degenerative facet joints, sacroiliac pain, trigeminal neuralgia, chronic plantar fasciitis, and refractory shoulder pain. The pulse generator of the RFA creates an electromagnetic field surrounding the electrode tip that activates adjacent molecules, thus generating frictional heat. After reaching the temperature threshold, the sensory nerve is partially denervated, relieving the refractory pain. Leggett et al. 25 found that RFA significantly reduced chronic low back pain associated with the lumbar facet joints and sacroiliac joints, discogenic low back pain, and coccygeal pain in short-term follow-ups. Amr et al. 26 reported that RFA intervention acted faster, provided a longer duration of analgesia, worked in a higher proportion of patients, and had a better safety profile in the management of refractory cancer pain. However, few studies have determined whether RFA can actually relieve knee OA pain. The present meta-analysis indicated that the use of RFA is associated with improved pain relief within 24 weeks. However, because of the limited number of available studies, the evidence does not clarify the effectiveness of different types of RFA. Further research in this important field is required.
Intra-articular injections reduce osteoarthritic pain through several pathways, including lubrication, anti-inflammation, an anabolic effect, and chondroprotection. However, the long-term benefit and cost-effectiveness of these treatments are still being debated. Published studies have shown that intra-articular injections are associated with accelerated degeneration of articular cartilage, which may change the underlying subchondral bone structures.27,28 The destruction of the articular cartilage renders it less capable to distribute large loads, making it more susceptible to stress and less capable to reduce friction within the joint space. As a consequence, knee motions are limited. With respect to outcome measures, the WOMAC has been used to evaluate functional restoration. El-Hakeim et al. 18 reported significant changes in the pretreatment WOMAC scores and the WOMAC scores during the entire follow-up period in both groups. However, Yuan et al. 17 found no statistically significant differences in the WOMAC scores. Four RCTs provided data regarding the WOMAC scores, and the present meta-analysis indicated that the use of RFA can significantly improve the WOMAC score. The effect of RFA generally lasts from 1 month to 2 years after treatment; however, this is dependent upon on the type of RFA and the target nerve. Long-term follow-ups are still required to determine the efficacy of RFA.
Although the outcome of RFA is currently considered satisfactory, there are potential concerns with this treatment, including local hemorrhage, hematoma formation, dysesthesias, thermal injury, and infection. Therefore, the use of RFA will hold less clinical value if there is a higher risk of adverse effects. Conger et al. 29 reported the development of a pes anserinus injury following RFA of the inferior medial genicular nerve. Kim et al. 30 reported the risk of genicular vascular complications under fluoroscopic guidance. Four RCTs reported adverse events, and no significant differences in terms of adverse events were found between the two groups that were the focus of this study. All adverse effects were mild, and no further treatment was required.
The results of the present study should be interpreted in light of the study limitations. First, the included studies and sample sizes were relatively small, which may affect the overall results. Second, the possibility of other confounding factors affecting the results could not be completely ruled out, such as the types of RFA and the characteristics and health of the patients. Third, the overall quality of evidence was moderate. Future high-quality research that explores the effects of RFA on larger samples is required to confirm the findings of this study. Finally, publication bias that is derived from a literature review is limiting and difficult to avoid.
Conclusion
RFA is efficacious and safe for reducing pain and improving knee function in patients with knee OA, without increasing the risk of adverse effects.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Hangzhou Medical and Health Technology Planning Program (20180533B61).
