Abstract
Background
Levels of specific IgE (sIgE) against allergen components can be assessed using multiplex assays or with highly sensitive, quantitative methods. The aim of this study was to compare the sensitivity and specificity of different immunological methods for diagnosis of shrimp allergy.
Methods
Twenty patients with positive skin prick tests for frozen tiger shrimp were selected for further examination. Blood samples were taken to assess concentrations of sIgE against the house dust mites Dermatophagoides pteronyssinus and D. farinae, shrimp allergen extract, allergen components Der p 1, Der p 2 and Pan a 1 (ImmunoCap), and the ImmunoCap ISAC 112 panel.
Results
All patients had elevated levels of sIgE against shrimp and D pteronyssinus. Eight patients were sensitized to Pen m 1, three patients were sensitized to Pen m 2, and two patients were sensitized to Pen m 4 (ISAC). ImmunoCap ISAC detected shrimp sensitization in 50% of patients. There was a strong correlation between concentrations of sIgE against Pen m1 and Der p 10 detected by ImmunoCap.
Conclusions
The singleplex ImmunoCap system remains the reference diagnostic method, but in the case of shrimp allergy ImmunoCap ISAC provided better insight into patient allergen profiles.
Background
Shrimp allergy is an increasing problem in the European population. Symptoms associated with the consumption of shrimp by allergic individuals can range from mild, local reactions to systemic reactions and anaphylactic shock. The main allergen of crustaceans is tropomyosin, which shows high interspecies homology and is also found in house dust mites (HDMs). Other shrimp allergens have also been reported and may be associated with a severe course of sensitization.
HDM allergy is widespread in Europe, including Poland. The largest epidemiological study conducted in Poland, the Epidemiology of Allergic Diseases in Poland (ECAP) study, found that allergic rhinitis was the most prevalent allergic condition affecting both children and adults and was diagnosed in 24% to 30% of the study population. 1 Boquete et al. 2 indicated that 71% of patients allergic to HDMs also had specific IgE (sIgE) against shrimp, and 55% had increased levels of sIgE against shrimp tropomyosin. Canadian studies demonstrated a high prevalence of allergy to HDMs in 95 patients with confirmed shrimp allergy. In the study population, 86 (90.5%) patients had positive skin prick tests for HDM allergens. 3 In a recent Italian study, 9% of 526 patients with HDM allergies also had allergy to shrimp. Patients with shrimp allergies were less frequently hypersensitive to airborne allergens other than HDM than crustacean-tolerant subjects. Only 51% of patients who were tropomyosin sensitized had shrimp allergies, and only 48% of patients who were shrimp sensitized were also sensitized to tropomyosin. 4
There are several different methods of diagnosing shrimp allergy. Double blind, placebo-controlled food challenge remains the gold standard for diagnosis of food allergy. Oral food challenges are technically difficult to perform and time consuming. For patients with histories of anaphylaxis, the risks may also outweigh the potential benefits of performing the challenge.
To our knowledge, only a few studies have addressed the clinical relevance of component-resolved diagnosis of shrimp sensitization. The aim of the current study was to analyze the clinical utility of different diagnostic methods for patients with shrimp allergy. It was not feasible to use all available diagnostic methods in patients suspected of shrimp allergy. This is the first study to compare the utility of singleplex ImmunoCap using allergen extracts and allergen components with ImmunoCap ISAC in this specific group of patients. Establishing the sensitivity and specificity of different immunological methods and knowledge of their limitations may help clinicians in deciding the best diagnostic approach for patients with suspected shrimp allergies.
Methods
Patients with symptoms of persistent allergic rhinitis were selected for screening. The selection was random and was based on order of visiting the Outpatient Clinic of Allergic Diseases. Patients being treated for serious chronic diseases and patients on medication that could influence the results of the study were excluded.
A detailed interview and physical examination was conducted for each patient. All patients underwent skin prick tests (SPTs) with HDM extracts (Dermatophagoides pteronyssinus and D. farinae; Allergopharma, Hamburg, Germany) and with frozen tiger shrimp purchased from a local eco-shop. Patients with positive SPTs for frozen tiger shrimp and self-declared symptoms of shrimp allergy were selected for further examination. As a control group, patients with negative SPTs for frozen tiger shrimp who declared no symptoms of shrimp allergy were enrolled.
Blood samples were taken from all patients to assess concentrations of sIgE to the HDMs D. pteronyssinus and D. farinae, shrimp allergen extracts (Pandalus borealis, Penaeus monodon, Metapenaeopsis barbata, and Metapenaus joyneri), and the allergen components Der p 1, Der p 2, and Pan a 1. All immunological assays were performed using the highly sensitive immunofluorescent ImmunoCap system (Thermo Fisher Scientific, Waltham, MA, USA). Concentrations of IgE were evaluated as elevated when they exceeded 0.35 kUA/L (ImmunoCap) in accordance with common practice in the field. In all patients we determined levels of sIgE against 112 allergen components using the ImmunoCAP ISAC 112 panel, a semi-quantitative test whose results are reported in ISAC Standardized Units (ISU) and provide indications of sIgE levels within the range of 0.3 to 100 ISU-E. The units used in the ImmunoCap ISAC (ISU-E) were developed especially for this test. The result is shown on a scale of four values for sIgE (indeterminate, <0.3 ISU-E; low, 0.3–0.9 ISU-E; medium, 1–14.9 ISU-E; high, >15 ISU-E). Levels of sIgE in ImmunoCap ISAC tests were considered as elevated when they exceeded 0.3 ISU-E. ImmunoCap ISAC tests included allergen components from D. pteronyssinus (Der p 1, Der p 2, and Der p 10), D. farinae (Der f 1 and Der f 2), and shrimp (Peneus monodon Pen m 1, Pen m 2, and Pen m 3).
Differences between groups were assessed using Mann–Whitney and Kruskal–Wallis tests with Dunn’s post hoc test. Spearman correlations were also calculated. Analyses were conducted using R, version 3.3.1 (www.r-project.org) and MS Excel 365. The study was approved by the local Bioethics Committee (number: 147/2015). All patients provided written informed consent to participate in the study.
Results
A total of 290 patients (176 women and 114 men) were screened and 20 had positive SPTs for frozen tiger shrimp (wheel >3 mm). All 20 patients had histories of allergic reaction after consumption of shrimp. A control group of 13 patients were selected with negative SPTs for frozen tiger shrimp and no symptoms of shrimp allergy. The characteristics of the study population and the results of immunoassays are presented in Table 1. Oral food challenges were not performed for several reasons, including lack of consent, time requirements, challenges in proper performance, and risks to patients with histories of anaphylaxis. All 20 patients reported symptoms following consumption of shrimp. Urticaria was reported by seven patients, anaphylaxis by six patients (including two patients with anaphylactic shock), digestive problems by four patients, and dyspnea by three patients.
General characteristics of the study population.

Data are shown as counts or as means ± standard deviations (ranges).
HDM, house dust mite.
All patients with shrimp allergies had elevated levels of shrimp sIgE by ImmunoCap. In addition, all patients were sensitized to D. pteronyssinus and 18 (90%) were sensitized to D. farinae. ImmunoCap ISAC results for shrimp and HDM allergens are presented in Table 2. Patients with shrimp allergies were sensitized to 72 allergen components by ImmunoCap ISAC. The most common sensitization in patients with shrimp allergy was to Der f 2 (16 patients). In addition, 12 patients were sensitized to Fel d 1, 11 patients were sensitized to Bla g 7 and Cyn d 1, and 10 patients were sensitized to Bet v 1, Cor a 1 and Phl p 1. The sensitization patterns are presented in Appendix Table 1. Among the 20 patients sensitized to shrimp, ImmunoCap ISAC detected elevated levels of sIgE against at least one out of three shrimp allergen components in only 10 patients (50%). In the control group all patients were sensitized to HDM by ImmunoCap, but none were sensitized to shrimp allergen extracts (Table 2).
Results of ImmunoCap ISAC tests.

HDM, house dust mite.
Table 3 shows a comparison between the concentrations of sIgE against allergen components detected by ImmunoCap and levels of sIgE detected by ImmunoCap ISAC. The results showed good concordance. Correlations in the 20 patients studied here were >0.7, but this result should be treated with caution. Although it is an indication that the results of the ImmunoCap ISAC and singleplex ImmunoCap methods were similar, the former method is semi-quantitative and the latter is quantitative. In addition, the units and the source shrimp species of tropomyosin differ between the two assays.
Comparison between concentrations of specific IgE measured using the quantitative ImmunoCap method and the semi-quantitative ImmunoCap ISAC microarray.
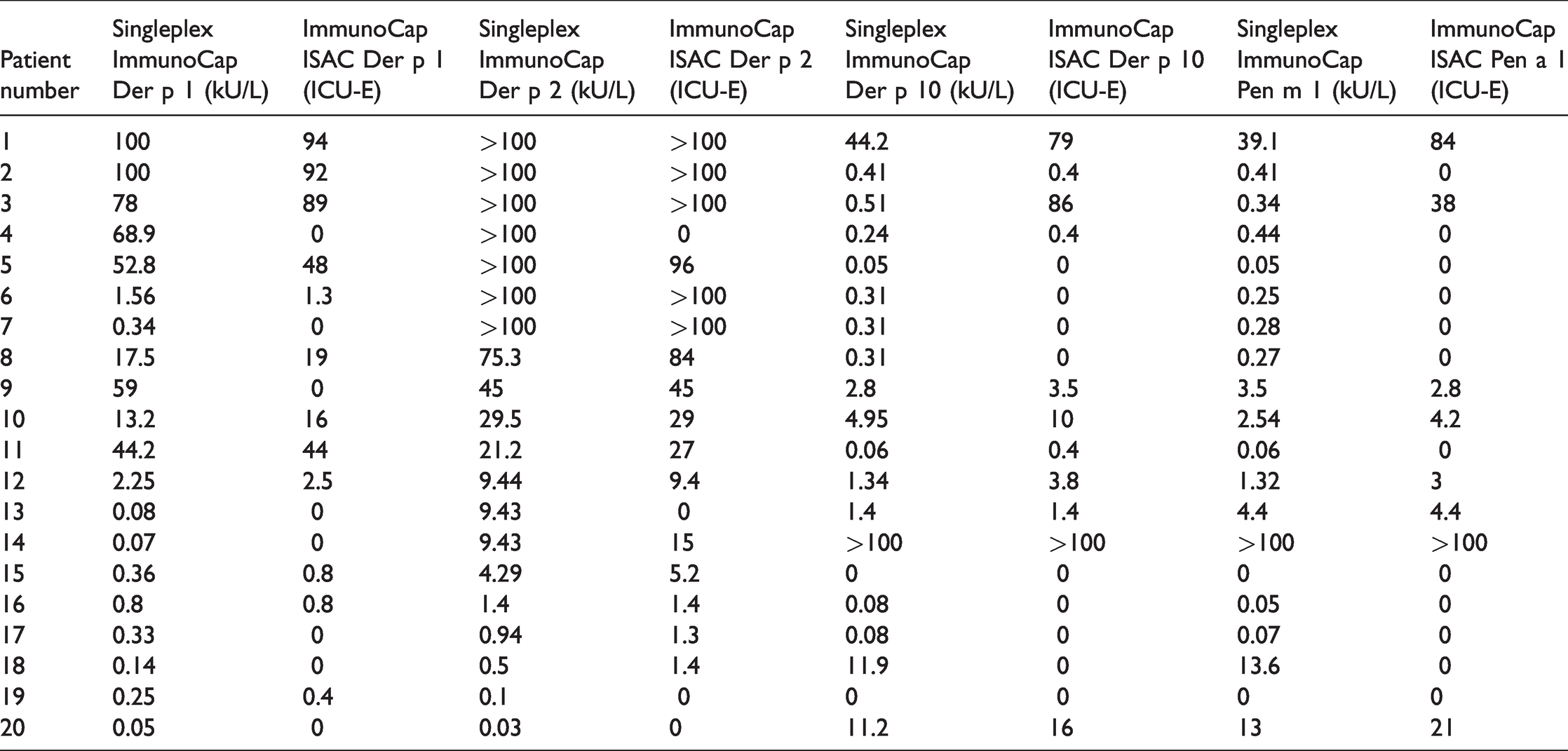
For HDM allergen components, ImmunoCap ISAC found that 84.6% patients were sensitized to Der p 1, 88.9% of patients were sensitized to Der p 2, and 90% of patients were sensitized to Der p 10. Only 50% of patients who were Pen m 1-sensitized had elevated levels of sIgE against Pen a 1 by ImmunoCap ISAC. There was a strong correlation between the concentrations of sIgE against Pen m 1 and Der p 10 in ImmunoCap assays (Figure 1).

Strong correlation (0.918, p < 0.001) between levels of IgE specific to Pen m 1 and Der p 10 in ImmunoCap (kU/L).
As shown in Table 1, ImmunoCap revealed that 18 of 20 patients sensitized to allergen extracts of D. pteronyssinus were also sensitized to Der p 2 (90%). All patients sensitized to Der p 1 were also sensitized to Der p 2, but only 10 (50%) were sensitized to HDM tropomyosin Der p 10.
Discussion
Diagnosis of shrimp allergy is based on clinical interview combined with SPTs, prick by prick tests and measurements of sIgE against allergen extracts or allergenic components. The gold standard for diagnosis of food allergy is still double-blind placebo-controlled food challenge. Although this is the reference method for diagnosis of food allergy, it is rarely performed. The main limitations of oral food challenges are that they are time consuming, expensive, technically difficult to perform, and are contraindicated in patients at risk of anaphylaxis. 5 In the current study we established a diagnosis of shrimp sensitization on the basis of reported symptoms after eating shrimp, the results of SPTs, and concentrations of sIgE in the blood.
There are currently several approaches for diagnosis of food allergy. SPTs or their derivatives (prick by prick tests) are simple, inexpensive, and yield results within minutes. Unfortunately, for food allergens SPTs have limited sensitivity. There are also contraindications to these tests, especially for patients taking certain medications or patients with skin diseases. 6
In vitro diagnosis of food allergy can be conducted using different approaches. Measuring the concentration of sIgE against food allergen extracts provides good insight into sensitization and often is sufficient to establish a diagnosis. Component resolved diagnosis takes the issue further, allowing assessment of the patient's detailed allergic profile. The concentrations of sIgE against specific proteins that mediate the allergic reaction can be measured using semi-quantitative methods such as micro-array testing or with highly sensitive, quantitative methods (e.g., ImmunoCap). A summary of the currently available methods for immunological diagnosis of shrimp allergy is presented in Table 4.7–10
Currently available methods for determining specific IgE levels against shrimp allergens.

N – natural source of allergen; E – allergen extract; R – recombinant allergen.
To our knowledge, only a few studies have addressed the clinical relevance of component-resolved diagnosis of shrimp sensitization. Ours is the first study to compare the utility of singleplex ImmunoCap assays using allergen extracts and allergen components and ImmunoCap ISAC in this specific group of patients. The present study focused on hypersensitivity to commercially available shrimp allergen extracts and allergen components in adult patients who had positive SPTs against frozen tiger shrimp and who experienced symptoms after consumption of shrimp. We also assessed the utility of singleplex vs multiplex methods of evaluating sIgE concentrations in this group of patients.
Among patients with shrimp allergies, we found a high prevalence of co-sensitization to HDM allergens. All 20 of the patients with shrimp allergies studied here were simultaneously sensitized to D. pteronyssinus and 18 were sensitized to D. farinae. This finding was not unexpected because of potential cross reactivity between shrimp and HDM allergens. Recently Asero et al. 11 emphasized in a review article that in cases of shrimp allergy, other allergens beyond tropomyosin are likely to be cross reactive with HDM and allergy to shrimp may be one factor triggering HDM-related asthma. Other shrimp allergens potentially cross reactive with HDM include arginine kinase (Der p 20), myosin light chain, and high molecular weight allergen, such as hemocyanin and paramyosin.12–15 It is likely that this list of homologous allergens will increase. 11
HDM allergens are relatively well described. In a study by Weghofer et al., over 97% of patients were allergic to a few HDM proteins including cysteine proteases (Der p 1 and Der f 1) and members of the NPC2 family (Der p 2 and Der f 2). 16 Tropomyosin is considered a minor allergen of HDMs that triggers allergy in as few as 5.6% of patients demonstrating increased sIgE levels against HDM allergens. 17 This finding was confirmed in our study: sensitization to cysteine proteases and members of the NPC2 family were the main causes of HDM sensitization in the patients studied here. Using both singleplex ImmunoCap and ImmunoCap ISAC methods, a limited number of HDM allergen components were available. Nevertheless, sIgE was detected at high prevalence compared with HDM allergen extracts (ImmunoCap).
ImmunoCap ISAC detected that 100% of patients were sensitized to D. pteronyssinus and that 88.9% of patients were sensitized to D. farinae. New multiplex platforms available for component resolved diagnosis (FABER and ALEX) include HDM allergen components (Der p 5, Der p 7, Der p 7, Der p 9 and/or Der p 23), providing additional value to these tests. However, in most cases the evaluation of sIgE against HDM allergen groups 1 and 2, as available in ImmunoCap ISAC, should be sufficient. 4 Previously, ImmunoCap ISAC was described as a valuable method for diagnosis of mite allergy. Yadzir et al. compared SPTs with the results of ImmunoCap ISAC. Among 40 patients with positive SPTs against HDM allergen extracts, elevated levels of sIgE against at least one HDM allergen component were detected in 34 patients (85%). 18
Panzer et al. in analyzed the ImmunoCap ISAC results of 1766 patients. In 1255 patients, increased levels of sIgE against at least one allergen component were detected. Sensitization to HDMs (Der p 1 or Der p 2) was detected in 32.7% of patients, and sensitization to tropomyosin (Der p 10) was detected in 1.9% of patients. Overall, allergy to tropomyosin was diagnosed in 2.2% of the study population. 19
The ImmunoCap ISAC contains three shrimp allergen components, whereas other multiplex methods include only tropomyosin. 10 Still, compared with the singleplex ImmunoCap method using shrimp allergen extract, the multiplex ImmunoCap ISAC method showed only a 50% detection rate. This indirectly shows that other shrimp allergen components are responsible for sensitization. It also shows that ImmunoCap ISAC is not sufficient for screening patients for shrimp sensitization.
The ImmunoCap ISAC allows better insight into patient sensitization profiles. In our study population, most patients who were shrimp sensitized, as detected by ImmunoCap ISAC, were also sensitized to only one of three shrimp allergen components. There were no patients sensitized to all three allergen components; only two patients were sensitized to Pen m 1 and Pen m 2, and one patient was sensitized to Pen m 1 and Pen m 4. There were many other allergen components sensitizing patients in the study population and polysensitization was an important issue complicating the clinical picture.
The relatively low prevalence of tropomyosin sensitization in our study population was consistent with the results of other studies. In 2012, Asero et al. found that only 41% of patients who were shrimp sensitized had elevated levels of sIgE against tropomyosin, whereas the sera of 52% of patients were reactive with a protein of molecular weight >60 kDa. Moreover, IgE reactive with proteins with molecular masses corresponding to arginine kinase (Pen m 2, 40 kDa), calcium-binding sarcoplasm binding protein (Lit v 4, 20 kDa), light myosin chain (Lit v 3, 20 kDa), triphosphate isomerase (Cra c 8, 27 kDa), troponin C (Cra c 6, 17 kDa), and fatty acid binding protein (15 kDa) was rarely observed (13% of patients). 12 Girffida et al. 20 identified the high molecular weight protein (>60 kDa) as hemocyanin. There are no commercially available methods of determining levels of sIgE against this allergen, but in previous studies it may have been responsible for clinically significant reactions to shrimp, including anaphylactic shock. Hemocyanins are considered to be among the allergens responsible for cross reactivity of HDM allergens.20,21 Recently, Tonomura et al. 22 identified another clinically important protein with a molecular weight of 40 kDa as fructose 1,6–bisphosphate aldolase in a case of food dependent, exercise induced anaphylaxis. In 2018, Kimura et al. 23 identified a 43-kDa shrimp allergen as a cause of food-dependent, exercise-induced anaphylaxis.
Johnston at al. investigated the sera of 21 patients who had clinical reactions to shellfish. Most subjects (13/21, 62%) showed positive sIgE to shrimp allergens by ImmunoCap; 43% of patients were sensitized to tropomyosin, while an additional 29% were sensitized to other shrimp allergens including sarcoplasmic calcium-binding protein, arginine kinase and hemocyanin. The authors highlighted the lack of standardized shrimp allergens and the inadequacy of current extracts for shrimp allergy diagnosis. 24
An important aspect of this study was to assess the utility of different immunological methods for diagnosis of shrimp allergy. As observed in our previous study, there was high concordance of sIgE against Der p 10 and Pen m 1 (ImmunoCap) (Spearman correlation 0.918, p < 0.001), which may indicate that it is economically sensible to diagnose shrimp allergy using only one of the methods. 25 We also found positive correlations between concentrations of sIgE to Der p 1, Der p 2 and Der p 10 by singleplex ImmunoCap and the corresponding allergen components in ImmunoCap ISAC. Not all patients with elevated sIgE according to the singleplex ImmunoCap method were detected, but the detection rate was above 84% in both cases. The detection rate of sIgE against shrimp tropomyosin was lower (50%); however, the ImmunoCap ISAC and singleplex ImmunoCap assays use tropomyosin from different allergen sources (P. monodon vs F. aztecus). Although published data on different immunological assays for diagnosis of shrimp allergy are limited, our data are consistent with previously published research compering the utility of ImmunoCap ISAC vs ImmunoCap assays (either extract based or component based). Griffiths et al. retrospectively analyzed 118 patients to evaluate what testing strategy (SPT, ImmunoCAP, or ISAC) was the most appropriate for diagnosis in a “real-life” clinical setting. Inpatients with nut allergy, the detection rates of SPTs (56%) and ISAC (65%) were lower than those of ImmunoCap (71%). By contrast, ISAC had a higher detection rate (88%) than ImmunoCap (69%) or SPTs (33%) for diagnosis of oral allergy. ImmunoCap results identified all nine patients with anaphylaxis resulting from wheat allergy (100%), whereas ISAC was positive in only six of nine patients (67%). ImmunoCap showed high specificity and sensitivity overall. 26
Huss-Marp et al. compared ImmunoCap ISAC with singleplex ImmunoCap in a 2015 study. A total of 101 patients with grass pollen allergy were compared in terms of sIgE detection rates against extracts vs ImmunoCap ISAC components. The authors identified four possible explanations of discrepancies between ImmunoCap ISAC and singleplex ImmunoCap results: (i) the trigger allergen was not present in the ISAC panel; (ii) the sensitivity of the ISAC is lower than that of singleplex ImmunoCap tests; (iii) the native extract-based antigen is not identical to the recombinant molecular allergen in ISAC; or (iv) the trigger allergen is absent or underrepresented in the allergen extract. In prior studies the positive percent agreement (PPA) and negative percent agreement (NPA) of corresponding allergens between the ISAC test and the extract-based singleplex ImmunoCap results at a cut-off of 0.1 kUA/L varied between 60% and 100% for PPA and 78% and 97% for NPA. At a cut-off of 0.35 kUA/L (as in our study) the PPA for D. pteronyssinus was 61%, but for timothy grass (with seven allergen components in the ImmunoCap ISAC panel: Phl p 1, 2, 4, 5, 6, 7, 11, and 12) it was 100%. Shrimp sensitization was not analyzed. 27 In our study the PPA of sIgE against D. pteronyssinus was 100%, but for shrimp allergy the PPA between extract detection and ImmunoCap ISAC was low (50%). The main cause was probably a lack of important shrimp allergen components in ImmunoCap ISAC.
Although in our study, the presence of Der p 1 and Der p 2 was sufficient to diagnose HDM sensitivity in patients, the new ImmunoCap ISACE112i, available in Poland since 2019, contains an additional recombinant HDM allergen component (Der p23). Huang et al. developed a customized microarray based on ImmunoCap ISAC technology that was produced by Phadia Austria GmbH (Vienna, Austria) and contained as many as 13 HDM allergen components (the clinically relevant nDer p 1; rDer p 2, 5, 7, 21, and 23; and Der p 4, 10, 11, 14, 15, 18, and clone 16). Streptavidin-based ImmunoCaps (o212 ImmunoCap, Thermo Fisher Scientific/Phadia) were used to prepare microarrays containing a mixture of Der p 1, 2, 5, 7, 21, and 23. The strongest correlation was identified between extract-based ImmunoCaps and molecular ImmunoCaps containing a mixture of Der p 1, 2, 5,7, 21, and 23. Interestingly, HDM-specific IgE levels determined using the molecular ImmunoCap were considerably higher than those measured using the allergen extract-based ImmunoCap. The authors found that Der p 2, Der p 5, Der p 21, and Der p 23 appeared to be underrepresented in natural allergen extracts. This finding suggested that adding components to ImmunoCap ISAC might increase detection rates of sensitized cases. An interesting suggestion was made that adding recombinant allergen molecules to natural HDM allergen extracts or preparing molecular ImmunoCaps containing the most important allergens could be of benefit in enhancing diagnosis. 28
Conclusion
Extract-based singleplex ImmunoCap methods are highly sensitive and widely available. Component resolved diagnosis adds additional information on the nature of sensitization and potential cross reactivity. The results of sIgE levels against corresponding allergen components determined using singleplex ImmunoCap and ImmunoCap ISAC were similar in most cases, with correlations above 0.7. In the case of shrimp allergy, the lack of many important shrimp allergen components, such as hemocyanin, was the probable cause of low detectability rates in molecular analysis (both singleplex and multiplex). The prevalence of HDM sensitization among patients with shrimp allergies was high, as described previously. The concordance of sIgE concentrations against Der p 10 and Pen m 1 between singleplex ImmunoCap and ImmunoCap ISAC was very high. Only 50% of patients with shrimp allergies were sensitized to tropomyosin and a lack of other important shrimp allergen components used in quantitative ImmunoCap ISAC measurements is a serious drawback for diagnosis of shrimp allergy. The results of immunoassays should be interpreted with care and a combination of extract and component-based diagnosis gives optimal insight into patient sensitization patterns.
Numbers of patients sensitized to specific allergen components in ImmunoCap ISAC. Levels of sIgE against allergen components that are not included in the table were not elevated in any patients.

sIgE, specific IgE
Footnotes
Author contributions
NUS – manuscript revision, study design, research material collection, interviews with patients, informed consent collection, manuscript preparation, final approval of the version to be published. This author agrees to be accountable for all aspects of the work related to its accuracy or integrity.
MZG – study design, immunoassays, final approval of the version to be published. This author agrees to be accountable for all aspects of the work related to its accuracy or integrity.
KL – study design, immunoassays, final approval of the version to be published. This author agrees to be accountable for all aspects of the work related to its accuracy or integrity.
RA – study design, manuscript evaluation and correction, final approval of the version to be published. This author agrees to be accountable for all aspects of the work related to its accuracy or integrity.
AK – manuscript revision, manuscript evaluation and correction, final approval of the version to be published. This author agrees to be accountable for all aspects of the work related to its accuracy or integrity.
ZB – study design, manuscript evaluation and correction, final approval of the version to be published. This author agrees to be accountable for all aspects of the work related to its accuracy or integrity.
Availability of data and materials
The dataset supporting the conclusions of this article are included within the article.
Consent for publication
All authors consented to the publication of the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by funding from the Collegium Medicum in Bydgoszcz, WL 551.
