Abstract
Objective
To identify predictors of the ovarian response to clomiphene citrate (CC) in infertile patients with polycystic ovary syndrome (PCOS).
Methods
We performed a prospective cohort study of infertile patients with PCOS. The participants underwent assessments of their physical, endocrine, and metabolic characteristics, and treatment with CC at an initial dose of 50 mg/day and a maximum of 100 mg/day between days 3 and 7 of their menstrual cycles. Participants who ovulated were identified as responders and those who did not as non-responders.
Results
Of the 72 participants, 48 (66.7%) were identified as responders and 24 as non-responders. Sex hormone-binding globulin (SHBG) (odds ratio 1.022, 95% confidence interval: 1.000–1.045) was found to be associated with the ovarian response to CC using logistic multivariate regression analysis. Receiver operating characteristic analysis also showed that SHBG was a significant predictor of the response to CC (area under the curve 0.799).
Conclusion
We have shown that SHBG is the best prognostic indicator of an ovulatory response to CC. However, larger prospective studies, in which more variables are assessed, are required to confirm this finding and to identify appropriate cut-off values.
Keywords
Introduction
Polycystic ovary syndrome (PCOS) is the most common endocrine and metabolic disorder, affecting 6% to 10% of women of reproductive age. 1 PCOS is often associated with oligo-anovulation and results in infertility, which necessitates the artificial induction of ovulation. Indeed, infertility is 10 times more prevalent in patients with PCOS than in healthy individuals. 2 Clomiphene citrate (CC) is a selective estrogen receptor modulator (SERM) that induces the secretion of a pulse of gonadotropin‐releasing hormone (GnRH), which inhibits the negative feedback effects of circulating estrogen and induces gonadotropin secretion from the anterior pituitary gland. The stimulation of the ovaries by these endogenous gonadotropins stimulates follicular development. 3 CC is widely used as a first-line therapy to induce ovulation in patients with PCOS. 4 However, 20% to 25% of women are CC-resistant. 5 Patients with PCOS who demonstrate hyperandrogenism, insulin resistance (IR) and obesity are reported to be less likely to respond to CC, which implies that these defects may be the principal causes of CC resistance.6,7
Various clinical, metabolic and hormonal factors might affect the response to CC. Ellakwa et al. found that the total circulating testosterone (TT) concentration was the best individual predictor of the response to CC treatment. 8 Sachdeva et al. 9 reported that body mass index (BMI), fasting insulin concentration and antral follicle count (AFC) were the best anthropometric, metabolic and ovarian reserve predictors, respectively, of non-responsiveness to CC. In addition, Xi et al. 10 showed that serum anti-Müllerian hormone (AMH) concentration may be useful for the identification of patients with PCOS who were more likely to respond to CC treatment. However, there have been no studies that have assessed the relationship between sex hormone-binding globulin (SHBG) concentration and the response to CC. In our previous studies, SHBG was shown to be a predictor of gestational diabetes mellitus (GDM) and pre-eclampsia (PE) in infertile patients with PCOS.11,12 SHBG is synthesized in the liver and is the major plasma transport protein for testosterone, with 95% of this hormone being bound to SHBG. 13 Therefore, the circulating SHBG concentration may predict the response to CC. In the present study, we aimed to identify predictors of ovulation in response to CC in women with PCOS by performing a prospective cohort study, and to provide clinical basis for the individualized treatment of patients with PCOS and infertility.
Methods
Study design
A prospective cohort study was carried out in the Obstetrics and Gynecology Hospital of Fudan University, Shanghai, between January 2014 and December 2017. We enrolled patients with PCOS who were infertile, were 20 to 40 years old, and attended the Reproductive Endocrinology Clinic. Infertility was diagnosed using the World Health Organization criteria: the inability to conceive despite regular sexual intercourse over a year, without the use of any contraception. 14 Diagnoses of PCOS were made using the Rotterdam criteria (2003). At least two of the following three criteria had to be met: clinical and/or biochemical signs of oligo- and/or anovulation, hyperandrogenism, and polycystic ovary on ultrasonographic examination. In addition, related diseases were excluded, including adrenal congenital hyperplasia, Cushing’s syndrome, and androgen-secreting tumors. 15 As shown in Figure 1, all the women with PCOS were administered an initial dose of 50 mg/day and a maximum dose of 100 mg/day CC between days 3 and 7 of their menstrual cycles, after spontaneous or withdrawal bleeding.

Diagram of the study design
The study was approved by the ethics committee of the Obstetrics and Gynecology Hospital of Fudan University (number [2013]25). All the patients provided their written informed consent for the use of their medical records and their data were anonymized in the present manuscript.
Patient characteristics
The inclusion criteria were as follows: women between 20 and 40 years old, who had been diagnosed with PCOS and infertility, and then treated with 50 to 100 mg/day of CC. The exclusion criteria were as follows: women aged >40 or <20 years; the presence of a related disease, such as adrenal hyperandrogenism or diabetes mellitus; and the induction of ovulation with >100 mg/day CC or another medication, such as letrozole.
The participants were subsequently identified as responders and non-responders. Responders were defined as having PCOS if they ovulated during CC therapy, irrespective of the dose administered. Non-responders were defined as participants who did not ovulate following treatment with 100 mg/day CC over three cycles. When one or two dominant follicles of >18 mm in diameter were identified on ultrasonographic examination, 5000 to 10,000 units of human chorionic gonadotropin (Guangzhou TianCheng Medical Technology Co., Ltd, Guangzhou, China) were injected intramuscularly.
It has been reported that between 15% and 40% of patients with PCOS show persistent anovulation following three successive treatments with CC5,16. The required sample size was calculated using this range, 90% power, α=0.05, and the equation n=µ 2 α/2π(1-π)/δ 2 ; and was determined to be between 49 and 92.
Outcome measures
Prior to treatment, the height, body mass, waist circumference (WC), hip circumference, Ferriman–Gallway (F-G) score for acne, Rosenfield score for acanthosis nigricans (AN) were recorded for all the participants. The serum concentrations of sex hormones (follicle‐stimulating hormone (FSH), luteinizing hormone (LH), estradiol (E2), prolactin (PRL), progesterone, TT, dehydroepiandrosterone sulfate (DHEA‐S), SHBG and 17α-hydroxyprogesterone (17α-OHP)) were measured during a morning of the third to fifth day of the menstrual cycle or progestogen-induced withdrawal bleeding. The fasting glucose, insulin, total cholesterol (TC), triglyceride (TG), high-density lipoprotein-cholesterol (HDL), and low-density lipoprotein-cholesterol (LDL) concentrations; and the glucose and insulin concentrations obtained during an oral glucose tolerance test (OGTT) (75 g glucose, 3‐hour duration) were also recorded. Serum samples were analyzed in the laboratory of the Obstetrics and Gynecology Hospital of Fudan University. The SHBG and insulin concentrations were measured using a Modular E170 analyzer (Roche Diagnostics, Mannheim, Germany) after calibration with the international standard preparation 95/560, which had a within‐run coefficient of variation of 1.0% at 21 nmol/L. Glucose, TC, TG, HDL-cholesterol and LDL-cholesterol concentrations were measured using a Hitachi 7180 automatic biochemical analyzer (Hitachi, Tokyo, Japan). FSH, LH, PRL, E2, progesterone, T, DHEA‐S and 17α-OHP concentrations were measured using a DxI800 immune analyzer (Beckman Coulter, Pasadena, CA, USA). Transvaginal ultrasonography was performed to diagnose PCOS and to quantify the number and sizes of the follicles in both ovaries using an SSD 5500 (Aloka Inc, Tokyo, Japan). Follicular growth was monitored in subsequent cycles until ovulation.
Body mass index (BMI) was calculated
17
to estimate the severity of obesity and the waist-to-hip ratio (WHR) was calculated as an index of central obesity. Homeostasis model assessment-insulin resistance (HOMA-IR)
18
Calculations for outcome measures
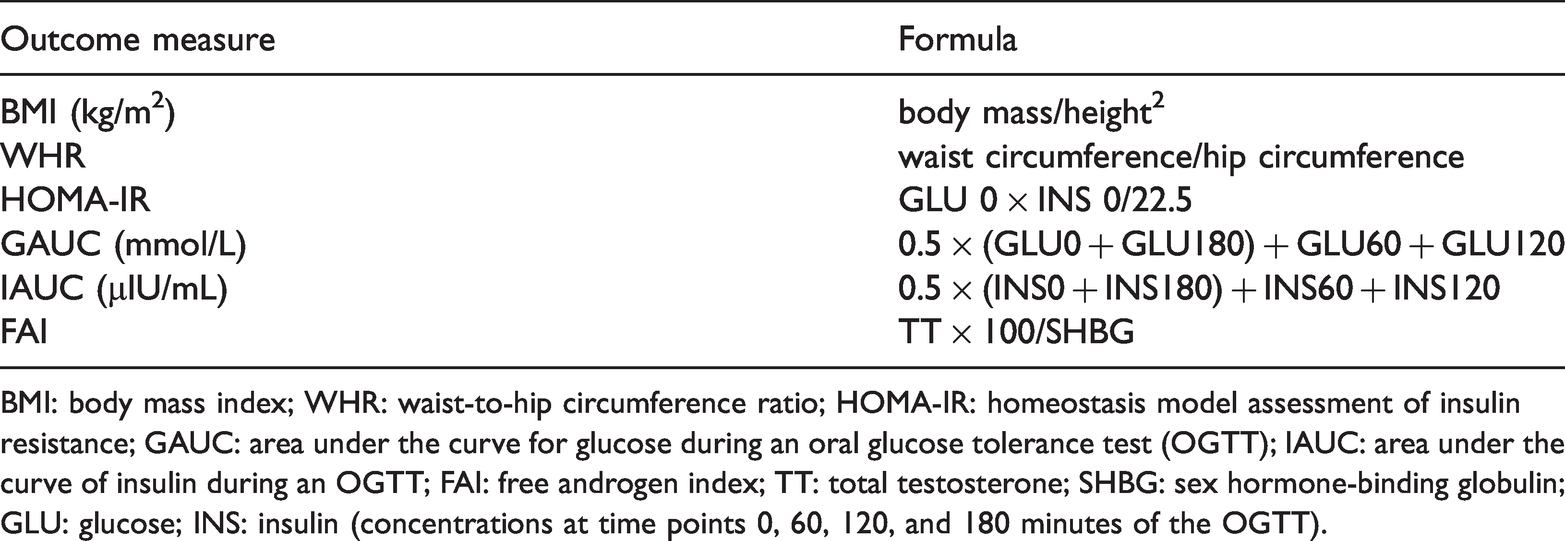
BMI: body mass index; WHR: waist-to-hip circumference ratio; HOMA-IR: homeostasis model assessment of insulin resistance; GAUC: area under the curve for glucose during an oral glucose tolerance test (OGTT); IAUC: area under the curve of insulin during an OGTT; FAI: free androgen index; TT: total testosterone; SHBG: sex hormone-binding globulin; GLU: glucose; INS: insulin (concentrations at time points 0, 60, 120, and 180 minutes of the OGTT).
Statistical analysis
Student’s t-test was used to compare data that were normally distributed and showed homogeneity of variance. Otherwise, the Mann–Whitney U test was used. Logistic regression analyses were performed to identify associations between variables and the response of the participants to CC treatment. Receiver operating characteristic (ROC) curves were plotted to assess the prognostic value of parameters identified in this way, with the area under the ROC curve being taken to represent the accuracy of the prediction of the ovulatory response to CC. P <0.05 was considered to represent statistical significance. Statistical analyses were performed using SPSS Version 21.0 (IBM, Inc., Armonk, NY, USA).
Results
We studied 72 Chinese women with PCOS and anovulatory infertility, of whom 48 ovulated after CC treatment and were defined as responders, and 24 did not, and were therefore defined as non-responders. As shown in Table 2, the WHR was higher in non-responders (P <0.05), but there were no differences in the body mass, height, BMI, WC, F-G Score, Rosenfield Score or prevalence of AN between the two groups (all P >0.05). The concentrations of LH, TT, FAI and DHEA-S were higher, whereas that of SHBG was lower in the non-responders (all P <0.05) (Table 2). The GAUC and IAUC of the non-responders were also higher than those of the responders (both P <0.05), which implied that resistance to CC was associated with IR and glucose intolerance. However, there was no difference in the lipid profile of the two groups of participants (Table 2).
Demographic characteristics; and hormone, glucose, and lipid concentrations of infertile PCOS patients prior to the induction of ovulation

Values are expressed as mean ± standard deviation. Data were analyzed using Student’s t-test, the paired t-test, or Mann–Whitney U test, as appropriate.
BMI: body mass index; WC: waist circumference WHR: waist-to-hip circumference ratio; AN: acanthosis nigricans; FSH: follicle-stimulating hormone; LH: luteinizing hormone; PRL: prolactin; E2: estradiol; TT: total testosterone; SHBG: sex hormone-binding globulin; FAI: free androgen index; DHEA-S: dehydroepiandrosterone sulphate. GLU: glucose; INS: insulin (concentrations at time points 0, 60, 120, and 180 minutes of an oral glucose tolerance test (OGTT)); IAUC: area under the insulin curve during an OGTT; HOMA-IR: homeostasis model assessment-insulin resistance; GAUC: area under the curve for glucose during an OGTT; TC: total cholesterol; TG: triglycerides; HDL: high-density lipoprotein-cholesterol; LDL: low-density lipoprotein-cholesterol.
Logistic regression was performed to identify associations between the parameters that were significantly different according to Student’s t-test and the response to CC in women with PCOS. Univariate logistic regression analysis showed that WHR, TT, FAI, SHBG, LH, DHEA-S, IAUC and GAUC were all significantly associated (all P <0.05). All of these variables were then subjected to multivariate logistic regression analysis, which showed that only SHBG (odds ratio 1.022, 95% confidence interval (CI): 1.000–1.045, P<0.05) was significantly associated with the response (Table 3).
Logistic regression analyses of variables that were associated with the response to clomiphene citrate in infertile women with PCOS

Italicized values are significant.
WHR: waist-to-hip circumference ratio; LH: luteinizing hormone; TT: total testosterone; SHBG: sex hormone-binding globulin; FAI: free androgen index; DHEA-S: dehydroepiandrosterone sulphate; GAUC: area under the curve for glucose during an OGTT; IAUC: area under the curve for insulin during an OGTT.
Subsequently, the predictive value of SHBG was analyzed using ROC analysis. The area under the ROC curve was 0.799 (95% CI: 0.679–0.918; P <0.05) and the sensitivity and specificity for the use of SHBG as a predictor were determined to be 70.0% and 76.9%, respectively. However, the optimal cut-off value could not be calculated because of the small sample size (Figure 2).

Receiver operating characteristic (ROC) curve showing the value of sex hormone-binding globulin (SHBG) for the prediction of the ovarian response to clomiphene citrate (CC) treatment in infertile PCOS women
Discussion
In the present study, we aimed to identify predictors of the ovarian response to CC treatment in infertile patients with PCOS. We found significant associations of WHR, TT, SHBG, FAI, LH, DHEA-S, GAUC and IAUC with the ovarian response to CC, and that SHBG represented the best predictor of ovulation in response to CC, using multivariate logistic regression and ROC analysis.
We found that non-responders had higher WHR, LH, TT, FAI, DHEA-S, GAUC and IAUC and lower circulating SHBG concentration than responders. Similarly, Sachdeva 21 et al. reported in 2019 that CC-resistant patients with PCOS have significantly higher circulating androgen concentrations, longer menstrual cycles, more disturbed metabolic profiles, and higher BMIs than those who respond to CC. Therefore, they speculated that abdominal obesity, hyperandrogenism, hyperglycemia and hyperinsulinism may be associated with a poor ovarian response to CC.
SHBG was shown to be a predictor of complications of pregnancy, such as GDM and PE, in infertile women with PCOS in our previous studies.11,12 In the present study, we identified a difference in serum SHBG concentration between responders and non-responders and found that serum SHBG was associated with the response to CC. Low plasma SHBG concentration results in a higher unbound testosterone concentration, leading to hyperandrogenism, which also predisposed toward CC resistance in patients with PCOS. 22 In 1998, a significant negative correlation was found between SHBG concentration and IAUC (r= −0.29, P= 0.039). 23 However, the pathogenetic link between SHBG and the ovarian response to CC remained unknown. Nevertheless, an increase in baseline serum SHBG might improve the ovarian response to CC. The estrogen in combined oral contraceptive pills (COCP) stimulates the liver to synthesize SHBG and reduces the serum concentration of free androgens. Therefore, COCP are recommended for adult women with PCOS for the management of hyperandrogenism and low SHBG concentration, to improve their response to CC. 24 Therefore, comprehensive management, including the use of COCP, may be effective for infertile women with PCOS, because this would increase their serum SHBG and thereby improve their sensitivity to CC.
A larger cohort study or a multi-center study is required to identify the most appropriate cut-off value of SHBG for the prediction of the response to CC in patients with PCOS. Following this, the responses to CC could be compared in infertile patients with PCOS who have higher and lower baseline serum SHBG concentrations to determine whether pre-treatment of PCOS might be required before the induction of ovulation.
The small sample size was a limitation of this prospective cohort study, because the most appropriate cut-off value of SHBG could not be calculated. A further limitation was that markers of ovarian reserve, including AMH and AFC, were not included because the present study focused on the associations of clinical, hormonal, and metabolic parameters with the response to CC. Other parameters, including glycosylated hemoglobin, should also be assessed as potential prognostic factors in the future.
In conclusion, the ovarian response to CC treatment is associated with a number of parameters in women with PCOS (WHR, LH, TT, FAI, SHBG, DHEA-S, GAUC and IAUC), and serum SHBG might be a useful indicator of an ovulatory response to CC. Larger prospective multi-center cohort studies are needed to identify better predictors of the ovarian response in infertile women with PCOS.
Footnotes
Acknowledgement
The authors thank Qing Li, a qualified biostatistician, for his technical support with the data analysis.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Science and Technology Commission of Shanghai Municipality [grant no. 16411963400].
