Abstract
Objective
Interleukin (IL)-17 is a multifunctional cytokine with important roles in inflammatory and autoimmune diseases. This case–control study explored the relationships of IL-17A rs2275913 and IL-17F rs763780 single-nucleotide polymorphisms (SNPs) with recurrent aphthous ulcer (RAU) morbidity and severity.
Methods
IL-17A rs2275913 and IL-17F rs763780 SNPs were measured in 125 patients with RAU and 116 healthy control participants. The genotype distributions, disease risks, and relationships with RAU severity were analyzed.
Results
RAU risk was associated with rs2275913 after adjustment for age, body mass index, sex, smoking status, and drinking status (AA vs. GG: odds ratio [OR], 2.759; 95% confidence interval [CI], 1.381–5.512; A allele vs. G allele: OR, 1.783; 95% CI, 1.242–2.560). TC and CC genotypes in rs763780, and the corresponding C allele, demonstrated greater prevalence among patients with RAU, compared with the TT genotype (TC vs. TT, OR: 1.895; 95% CI: 1.088–3.301; CC vs. TT, OR: 4.080, 95% CI: 1.079–15.425; C allele vs. T allele, OR: 1.969, 95% CI: 1.257–3.083). Serum IL-17 concentrations were also higher in patients with RAU than in control participants. These concentrations were associated with IL-17 polymorphisms.
Conclusions
IL-17 polymorphisms might be associated with greater risk of RAU pathogenesis.
Keywords
Introduction
Recurrent aphthous ulcer (RAU; recurrent aphthous stomatitis) causes recurrent episodes of oral ulcers. RAU affects approximately 20% of the population worldwide.1,2 The etiology of RAU is multifactorial, such that genetic, immune, nutritional, and microbial factors, as well as local trauma, contribute to its onset. 3
The etiology of RAU is unclear, although inflammatory factors presumably play key roles in its development.4,5 Cytokine genes in peripheral blood mononuclear cells (e.g., interleukin [IL]-2, tumor necrosis factor-α, and IL-6) contribute to the development of RAU. 6 Additionally, IL-10 mRNA levels are reduced in patients with RAU, which suggests a failure to suppress inflammatory reactions in oral mucosa. 7
To the best of our knowledge, only a few studies have reported associations between inflammatory gene polymorphisms and risk of RAU. The IL-1β-3954 C/T (rs1143634), IL-6-174G/C (rs1800795), IL-10-1082A/G (rs1800896), and IL-10-819C/T (rs1800871) polymorphisms are correlated with risk of RAU in Chinese individuals. 1 However, no analysis of IL-17 polymorphisms and RAU has been reported.
IL-17 plays important roles in innate and adaptive immune functions.8,9 The subtypes IL-17A and IL-17F are mainly produced and secreted by T helper 17 (Th17) cells, which constitute a unique lineage of CD4+ effector cells.10,11 Th17 cells contribute to several human diseases, including autoimmune conditions, allergies, tumor development and progression, and transplanted organs/bone marrow acceptance or rejection. Th17 cells are presumed to be actively recruited to and/or expanded in pathological sites, where they secrete proinflammatory cytokines to enhance inflammation. 11 In patients with psoriasis, Th17-type cytokines are overexpressed in lesional skin and serum; Th17 cells also infiltrate into lesional skin. Targeting the IL-12/IL-23 axis is an effective treatment for psoriasis, which indicates that Th17 cells are important in psoriasis pathogenesis. 12 IL-17 stimulates the release of pro-inflammatory cytokines and chemokines, matrix metalloproteinases, and antimicrobial peptides from myeloid, mesenchymal, and epithelial cells. 13 Thus, IL-17 is considered an important driver in the pathogenesis of autoimmune diseases (e.g., systemic lupus erythematosus and membranous glomerulonephritis).14,15
Genetic polymorphisms of IL-17 are associated with susceptibility to several immune-mediated diseases, including ulcerative colitis, systemic lupus erythematosus, membranous glomerulonephritis, and rheumatoid arthritis.16–19 Here, we hypothesized that IL-17 is a susceptibility factor for RAU and explored the associations of IL-17 polymorphisms (IL-17A rs2275913 and IL-17F rs763780) with risk of RAU (i.e., morbidity and severity) in a Han Chinese population.
Materials and methods
Participants and study design
This study included patients who were diagnosed with RAU in the Second Hospital of Hebei Medical University from April 2017 to December 2019. The diagnostic criteria were ≥1 horizontal oral ulcers covered with a yellow pseudomembrane, presence of surrounding hyperemia, central sag and obvious causalgia; ulcers were cyclical, self-limiting, and exhibited distinct phases. Inclusion criteria included: (1) ≥6 months of regularly recurrent episodes of oral aphthous ulcer; (2) at least two ulcers per month within 6 months; (3) normal full blood count with serum B12 of 200 to 900 ng/L, red cell folate of 110 to 700 µg/L, and serum ferritin of 20 to 400 µg/L. Exclusion criteria included: (1) pregnancy; (2) history of oral ulcers caused by systemic diseases (e.g., Behçet’s syndrome, celiac disease, Crohn’s disease, ulcerative colitis, or autoimmune deficiency syndrome); (3) combined treatment with systemic steroids, immunomodulatory drugs, or cytotoxic drugs. Healthy control participants without RAU were recruited from among individuals undergoing routine examinations in our hospital during the study period. Participants in the control group were confirmed to have no history of RAU; they were matched with patients in the study on the basis of sex and age. Written informed consent was obtained from all participants for inclusion in the study. The study protocol was approved by the Ethics Committee of the Second Hospital of Hebei Medical University (approval number: SH20170832).
Clinical assessment of disease severity in patients with RAU
Six ulcer characteristics were used to generate an ulcer severity score (USS): number, size, duration, ulcer-free period, site, and pain. For the purposes of RAU severity scoring, the characteristics of ulcer attacks in the preceding 3 months were recorded using a standardized form, established on the basis of published literature. 20 Patients were surveyed to ascertain the mean ulcer size, number of ulcers per attack, ulcer duration, ulcer attack frequency, sites affected, and intensity of pain caused by ulcers. A patient’s description of ulcers was verified clinically whenever possible. Ulcer characteristics were converted into numerical data to provide a score for objective comparison of disease severity, using a previously described method. 20
Sample collection
Ten milliliters of fasting peripheral blood were collected into ethylenediaminetetraacetic acid-containing tubes by venipuncture from each participant for DNA isolation, as described in a subsequent section. The serum was separated from these blood samples for assessment of biochemical, physiological, and immune parameters, as described in a subsequent section.
Genomic DNA isolation
A DNA Blood Mini Kit (Qiagen, Hilden, Germany) was used for extraction of genomic DNA from 200 µL of whole blood, in accordance with the manufacturer’s instructions. DNA concentrations were measured by spectrophotometry (NanoDrop 2000, Thermo Fisher Scientific, Waltham, MA, USA), then diluted to approximately 40 ng/µL.
DNA sequencing analysis
IL-17 sequences were amplified by polymerase chain reaction using the following polymorphism primers: rs2275913 forward: 5′-
Serum IL-17A and IL-17F concentrations
Total serum was separated from non-anticoagulant-treated blood samples by centrifugation of clotted blood at 1200 × g for 10 min at 4°C. Serum concentrations of IL-17A and IL-17F were measured by sandwich enzyme-linked immunosorbent assay (Abcam, Cambridge, MA, USA), in accordance with the manufacturer’s instructions and previously published protocols.21,22 The optical density of each sample well was measured at 450 nm using an S190 microreader (Molecular Devices, Sunnyvale, CA, USA). Concentrations of IL-17A and IL-17F were calculated based on a standard curve.
Statistical analysis
The effective sample size and statistical power were determined using Genetic Power Calculator software developed by Purcell et al. 23 (http://pngu.mgh.harvard.edu/∼purcell/gpc/). The parameters for predicting sample size included the frequencies of IL-17A rs2275913 and IL-17F rs763780, incidence of RAU, and predicted odds ratios of IL-17A and IL-17F polymorphisms. The specific assumptions for 85% statistical power were 5% disease prevalence, 5% minor allele frequencies, complete linkage disequilibrium, 1:1 case-to-control ratio, odds ratio of 2, and 5% type I error rate. For both the control and patient cohorts, the minimum number of participants was 109 each.
Clinical data are described as mean ± standard deviation in each group; these data were analyzed using Student’s t-test. The genotype and allelic frequencies were calculated by Hardy–Weinberg equilibrium using an online calculator (http://www.oege.org/software/hardy-weinberg.html), then assessed by chi-squared analysis or Fisher’s exact test. IMB SPSS Statistics, version 19.0 (IBM Corp., Armonk, NY, USA) was used to calculate odds ratios and 95% confidence intervals for associations between single-nucleotide polymorphisms (SNPs) and RAU risk; this software was also used for multivariate analysis (i.e., multinomial logistic regression models) to clarify whether differences between patients with RAU and healthy control participants were due to their genotypes. The threshold for statistical significance was set at P < 0.05.
Results
Participant characteristics
This study recruited 125 Han Chinese patients with RAU and 116 age and sex-matched healthy Han Chinese control participants. Power analysis indicated a power of 0.89 to detect associations between IL-17A and IL-17F polymorphisms and the risk of RAU at a level of significance of 0.05. The detailed demographic characteristics of the study population are listed in Table 1. There were no significant differences in age, sex, body mass index, or smoking status between the two groups. However, a greater proportion of patients with RAU reported a drinking habit (P = 0.032, Table 1).
Demographic characteristics of the study population.

Data are shown as mean ± standard deviation or number.
IL, interleukin.
A allele in rs2275913 and C allele in rs763780 were associated with increased risk of RAU
The distributions of genotypes and alleles are shown in Table 2; these distributions exhibited Hardy–Weinberg equilibrium. The genotype and allele distribution in rs2275913 significantly differed between healthy control participants and patients with RAU (Table 2). In addition, AA genotype carriers had a significantly greater risk of RAU, compared with genotype GG carriers (P = 0.006). Moreover, A allele carriers had a significantly greater risk of RAU, compared with G allele carriers (P = 0.002). Finally, the AA genotype and A allele were substantially more common in women than in men (P < 0.05; Table 2).
Distributions of IL-17 single-nucleotide polymorphism genotypes in each group and analyses of associations between polymorphisms and risk of recurrent aphthous stomatitis.

RAU, recurrent aphthous ulcer; OR, odds ratio; CI, confidence interval.
Similar results were observed concerning genotype and allele distribution in rs763780 (Table 2); these significantly differed between patients with RAU and healthy control participants. Notably, C allele carriers had a greater risk of RAU, compared with T allele carriers (Table 2). Furthermore, women with the C allele exhibited greater risk of RAU, compared with women with the T allele (P = 0.021). However, men with the CC genotype or C allele did not exhibit a significantly greater risk of RAU.
Multinomial logistic regression analysis was conducted to control for possible confounding factors (age, sex, body mass index, smoking status, and drinking status). As shown in Table 3, after controlling for confounding factors, the A allele of rs2275913 and C allele of rs763780 remained significantly associated with risk of RAU (P = 0.043 and P = 0.038, respectively).
Multivariate analyses of potential risk factors for recurrent aphthous ulcer in Han Chinese individuals.

RAU, recurrent aphthous ulcer; OR, odds ratio; CI, confidence interval.
IL-17 polymorphisms were related to RAU severity
RAU severity was evaluated based on the USS for each patient. The median USS was 32 (range, 18–60). Based on the median score, all patients with RAU were classified into two sub-groups: < 32 USS (low score group) and ≥32 USS (high score group). The distribution of SNP according to RAU severity is shown in Table 4; there were significant differences in all genotypes and alleles between groups, which suggested that these two IL-17 polymorphisms are risk factors for increased RAU severity. Furthermore, serum IL-17A and IL-17F concentrations were significantly higher in the high score group than in the low score group (P = 0.015 and P = 0.038, respectively; Table 4).
Association of polymorphism genotypes and alleles with recurrent aphthous ulcer severity.

IL, interleukin; SD, standard deviation; RAU, recurrent aphthous ulcer; USS, ulcer severity score.
Serum IL-17A and IL-17F concentrations were higher in patients with RAU
The mean serum IL-17A and IL-17F concentrations were significantly higher in patients with RAU than in healthy control participants (P = 0.0221 and P = 0.0314, respectively; Table 1). Furthermore, there were significant differences in serum IL-17A and IL-17F concentrations among all three genotypes in both loci (Table 5), which indicated that the A allele in rs2275913 and C allele in rs763780 might lead to increased secretion of IL-17A and IL-17F, respectively, in patients with RAU.
Serum concentrations of IL-17A and IL-17F according to genotype in patients with recurrent aphthous ulcer.
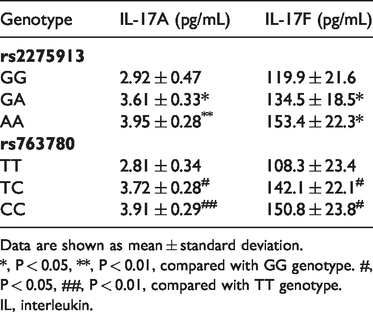
Data are shown as mean ± standard deviation.
*, P < 0.05, **, P < 0.01, compared with GG genotype. #,P < 0.05, ##, P < 0.01, compared with TT genotype.
IL, interleukin.
Discussion
Recent studies have indicated that Th17 cells and the IL-17/IL23 pathway play important roles in the pathology of inflammation-related diseases. 15 A previous meta-analysis has demonstrated relationships between polymorphisms of IL-17A (rs2275913) and IL-17F (rs763780) and the pathogenesis of inflammatory diseases (e.g., periodontitis, systemic lupus erythematosus, glomerulonephritis, rheumatoid arthritis, and inflammatory bowel disease). 14 Furthermore, the rs2275913 polymorphism of IL-17A is associated with osteitis after Bacillus Calmette–Guerin vaccination,24,25 and a variant of IL-17F (rs763780) may participate in the development of immune thrombocytopenia 26 and necrotizing enterocolitis. 27 Taken together, these findings indicate that IL-17 polymorphisms are connected to a wide array of immune-mediated diseases.
A previous study indicated that increasing levels of IL-17A and IL-17F are involved in the pathogenesis of minor aphthous, particularly in the ulcerative stage. 28 To the best of our knowledge, no study has reported the relationships of rs2275913 and rs763780 polymorphisms with the risk of RAU. Thus far, there has been insufficient evidence to determine whether these two SNPs affect the serum concentrations of IL-17A and IL-17F. Our findings imply that these two SNPs affect the serum concentrations of IL-17A and IL-17F (Table 4). In particular, the A allele in rs2275913 and C allele in rs763780 were associated with elevated plasma concentrations of IL-17A and IL-17F respectively, which may explain why these alleles were associated with elevated risk of RAU (Table 2). Additionally, rs2275913 A allele carriers had a greater risk of RAU, compared with G allele carriers; rs763780 C allele carriers were also at greater risk of RAU. Further evaluation of the effects of IL-17 polymorphisms on risk of RAU did not show significant sex-related differences. We suspect that a larger study will provide more comprehensive data concerning any differences related to patient demographics.
The present study showed higher mean serum concentrations of IL-17A and IL-17F in patients with RAU, compared with healthy control participants. Chronic wounds exhibit persistent inflammation with markedly delayed healing, including oral wounds. 29 Human oral epithelial cells are considered important inflammatory cells in RAU; serum IL-17A and IL-17F reportedly stimulate oral keratinocytes via IL-17RA/IL-17RE, thus promoting production of pro-inflammatory cytokines. 30 Because the IL-17 family contains proinflammatory cytokines implicated in various immune-mediated diseases in the gut and connective tissue, as well as inflammatory skin conditions, they may contribute to the pathogenesis of ulcer wounds; in particular, elevated concentrations of IL-17A and IL-17F may delay wound repair. 29 Analysis of inflammatory cells, such as Th17 cells, has revealed strongly positive IL-17A+ cells in the vascularized superficial connective tissue of human oral mucosa, which might aggravate RAU. Cytokine genes such as IL-17A and IL-17F are associated with Th17 cell development and function. We presume that IL-17 polymorphism-related regulation of the Th17 cell population might be involved in the RAU-associated inflammatory response.
There were some limitations in this study. First, it contained a small sample population limited to Han Chinese individuals; thus, the findings might not be generalizable to other populations. These results should be validated in studies involving patients of other ethnicities. Second, this study only investigated two SNPs in the IL-17 gene. Additional SNPs should be examined for their influence on the risk of RAU. Third, allele frequency differences between patients and healthy control participants after population stratification might have contributed to spurious associations due to systematic ancestry differences. 31 Therefore, more detailed statistical analysis is necessary to confirm our conclusions. Fourth, this study did not investigate the molecular mechanism by which IL-17A and IL-17F contribute to the risk of RAU. Fifth, although participants’ RAU statuses were confirmed by medical records and inquiries, a small proportion of healthy control participants might have exhibited RAU, thus weakening the strength of associations identified in this study. Finally, there were few smokers in both patient and healthy control groups, which prevented evaluation of the effect of smoking on any gene associations.
In summary, our results indicated that IL-17 functional polymorphisms were significantly correlated with the risk of RAU. The variant alleles rs2275913 AA and rs763780 CC might lead to elevated serum concentrations of IL-17A and IL-17F, higher risk of RAU, and increased disease severity. Taken together, these findings imply that inhibition of IL-17A and IL-17F might be an effective strategy for prevention and treatment of RAU, although the current cost of IL-17 inhibitors is substantial.
Footnotes
Authors' contributions
Conception and design: HX and QG; Acquisition of data, HX, DC, HG, and YW; Statistical analysis, ZJ.
Availability of data and materials
The relevant raw data are freely available to any scientist wishing to use them for non-commercial purposes, without breaching participant confidentiality.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
