Abstract
Objective
We aimed to determine the location of neurotrophic receptors tropomyosin receptor kinase (Trk)B, TrkC, and ciliary neurotrophic factor receptor (CNTFR)α in the retina of retinal degeneration (rd) mice and to explore the dynamic changes of B-cell lymphoma-2 (Bcl-2), Bcl-2-associated X-protein (Bax), and microtubule-associated protein light chain 3 (LC3) expression and ultrastructure in the retina of rd mice intravitreally injected with neurotrophic factors.
Methods
Rd mice aged 2 and 3 weeks post-natally (PN) received intravitreal injections of neurotrophic factors. Two weeks later, their retinas were harvested for the detection of Bax, Bcl-2, and LC3 mRNA and protein expression.
Results
TrkB and TrkC expression levels were lower at 3 weeks PN compared with 0, 1, and 2 weeks PN, but CNTFRα expression was still detected in certain layers. The three receptors were expressed in different retinal layers at the same timepoint. Bax expression was downregulated in, rhBDNF + rhCNTF, rhBDNF + rhNT-3, groups 2 weeks after intravitreal injection; Bcl-2 expression was upregulated in the rhBDNF + rhCNTF + rhNT-3 group at PN-4w; and LC3 expression was upregulated in rhBDNF + rhCNTF + rhNT-3 groups.
Conclusions
The combined use of neurotrophic factors had a more significant effect on Bax, Bcl-2, and LC3 expression than the same factors used alone.
Keywords
Introduction
Retinitis pigmentosa (RP) is a group of inherited, degenerative retinal diseases that are a major cause of adult blindness. The progression from initial symptoms of night blindness to ultimate visual acuity impairment is slow and varies among individuals because of hereditary and environmental heterogeneity. The prevalence of this disease is estimated to be approximately 1 in 4000 people. 1
There is currently no effective cure for RP 1 but several treatments have been proposed to slow its progression including gene therapy, antioxidative drugs, cell and tissue transplantation, prosthesis, and neuroprotective and neurotrophic factors.2,3 Because RP is genetically heterogeneous, treatments that are mutation-independent are desirable. 2 Multiple death signaling pathways are involved in disease pathogenesis, leading to the death of different photoreceptors and retinal ganglion cells. 4 Therefore, several neurotrophic factors such as brain-derived neurotrophic factor (BDNF), ciliary neurotrophic factor (CNTF), and neurotrophin-3 (NT-3) have drawn increasing attention as possible therapies5–8 and shown to have positive effects on retinal degenerative tissues when administered as single treatments. 9 However, when administered together, neurotrophic factors were more effective on photoreceptors than when used separately where they provided limited functional recovery and nerve regeneration. 10 , 11
The retinal degeneration (rd) mouse model is the classic animal model 12 that presents as an acute autosomal recessive form of RP. It carries a mutation affecting expression of the β-subunit of phosphodiesterase (PDE) encoded by the PDE6β gene, which leads to rod photoreceptor degeneration at postnatal day 8 (P8) and progresses to complete loss of the rods by postnatal week 3, resulting in early-onset severe retinal degeneration. 13 The rd mouse retina shows degeneration homologous to a form of RP with a rapid loss of rod photoreceptors and deficiency of retinal blood vessels.
In the present study, we used the rd mouse model to evaluate the combined effect of the neurotrophic drugs BDNF, CNTF, and NT-3 on the RP retina, and to assess tropomyosin receptor kinase (Trk)B, TrkC, and CNTFRα expression in retinas during disease progression to identify a suitable strategy for RP intervention.
Materials and methods
Animals
Rd mice and C57BL/6J mice were obtained from Laboratory Animal Center of Capital Medical University, and were kept under specific-pathogen-free conditions. Rd mice were sacrificed at 0, 1, 2, 3, and 4 weeks postnatally (PN) and their eyes were enucleated to make frozen sections for immunofluorescence. The animals were treated according to the Statement for the Use of Animals in Ophthalmic and Vision Research from the Association for Research in Vision and Ophthalmology. The study protocol was approved by the Ethics Committee of Beijing Tongren Eye Center.
Rd mice at PN-2 and PN-3 weeks were anesthetized (avertin, 12 mg/kg, i.p), and recombinant human BDNF (rhBDNF; Sigma, St. Louis, MO, USA), rhBDNF + recombinant human NT-3 (rhNT-3; Sigma), BDNF + recombinant human CNTF (rhCNTF; Sigma), or rhBDNF + rhNT-3 + rhCNTF were intravitreally injected into their left eye. Two doses of rhBDNF, rhCNTF, and rhNT-3 were tested: 50 or 100 μg/L [50 μg/L BDNF; 50 μg/L rhBDNF + 50 μg/L rhCNTF; 50 μg/L rhBDNF + 50 μg/L rhNT-3; 50 μg/L rhBDNF + 50 μg/L rhCNTF + 50 μg/L rhNT-3 or at their concentration of 100 μg/L]. Rd mice without injection served as blank controls (rd group). Rd mice that received phosphate-buffered saline (PBS) + bovine serum albumin (BSA) injections served as treatment controls (PBS group). C57BL/6J mice of the same age served as normal controls (C57BL/6J groups). Two weeks after injection (PN-4 and PN-5 weeks), all mice were sacrificed and retinas were harvested.
Immunofluorescence
Retinas were embedded in O.C.T. compound (Sakura, Torrance, CA, USA) and frozen sections of 7 μm were cut, fixed in acetone, and blocked with 5% BSA for 1 hour at room temperature. They were then incubated with rabbit anti-mouse TrkB (1:1000 dilution; Abcam, Cambridge, UK), rabbit anti-mouse TrkC (1:1000 dilution; Abcam), or goat anti-mouse CNTFRα (1:200 dilution; Santa Cruz Biotechnology, Santa Cruz, CA, USA) primary antibodies at 4°C overnight. The sections were washed with PBS, then incubated with fluorescein isothiocyanate (FITC)-conjugated goat anti-rabbit IgG polyclonal antibody (1:200 dilution; ZSGB-BIO, Beijing, China) or FITC-conjugated rabbit anti-goat IgG polyclonal antibody (1:200 dilution; ZSGB-BIO) for 45 minutes at 37°C. Sections incubated with PBS instead of the primary antibody served as negative controls. Finally, the samples were mounted (Applygen Technologies Inc., Beijing, China) and imaged using a DM4000B microscope system (Leica Microsystems, Wetzlar, Germany).
Real-time quantitative PCR
Retinas were collected and total RNA was extracted using TRIzol reagent (Life Technologies Co., Grand Island, NY, USA), and cDNA was reverse-transcribed using ReverTra Ace-α-™ (Toyobo, Osaka, Japan), according to the manufacturer’s protocol. PCR primers are shown in Table 1. The PCR protocol was: 95°C for 10 minutes, then 34 cycles of 95°C for 15 seconds and 55°C for 1 minute, with a final extension at 72°C for 2 minutes. Relative mRNA expression levels were calculated by the equation 2ΔΔCt method. GAPDH was used as the reference gene.
PCR primer sequences.

Western blot
Total protein was extracted from retinas using radioimmunoprecipitation assay lysis buffer (Beyotime Institute of Biotechnology, Haimen, China). Protein concentrations were determined using the BCA method (Beyotime Institute of Biotechnology). Equal amounts of proteins (20 µg/lane) were separated by 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis, then transferred to polyvinylidene difluoride membranes (Millipore Corp., Billerica, MA, USA) which were blocked with 5% (w/v) non-fat milk at room temperature for 1 hour. They were incubated with antibodies against Bcl-2-associated X-protein (Bax) (1:1000 dilution; Cell Signaling, Danvers, MA, USA), B-cell lymphoma-2 (Bcl-2) (1:1000 dilution; Cell Signaling), LC3A/B (1:1000 dilution; Cell Signaling), and GAPDH (1:1000 dilution; Cell Signaling) at 4°C overnight. A corresponding goat anti-rabbit IgG secondary antibody conjugated to horseradish peroxidase (1:5000 dilution; Abgent Inc., San Diego, CA, USA) was then incubated at room temperature for 1 hour and protein bands were detected by enhanced chemiluminescence (Thermo Fisher Scientific, Waltham, MA, USA). Protein bands were evaluated by densitometry using Quantity One analysis software (Bio-Rad, Hercules, CA, USA). Protein expression was normalized to GAPDH.
Transmission electron microscopy
An incision was made in the cornea of enucleated eyes, which were subsequently submerged in 2.5% glutaraldehyde (100 mmol/L phosphate buffer, pH 7.4) for 4 hours. The retina was post-fixed in 1% buffered osmium tetroxide and dehydrated using gradient ethanol solutions. Semi-thin sections (1 μm) stained with toluidine blue were produced and examined to determine the location for observation. Ultrathin sections (0.5 μm) of the fixed area were made and stained with uranyl acetate and lead citrate for transmission electron microscopy (Hitachi, Tokyo, Japan).
Statistical analysis
Data were expressed as the mean ± standard deviation (SD). Differences among groups at different time points were determined by one-way analysis of variance with the LSD post hoc test. Two-sided P-values <0.05 were considered statistically significant.
Results
Characteristics of neurotrophic factor receptor expression in retinitis pigmentosa tissues
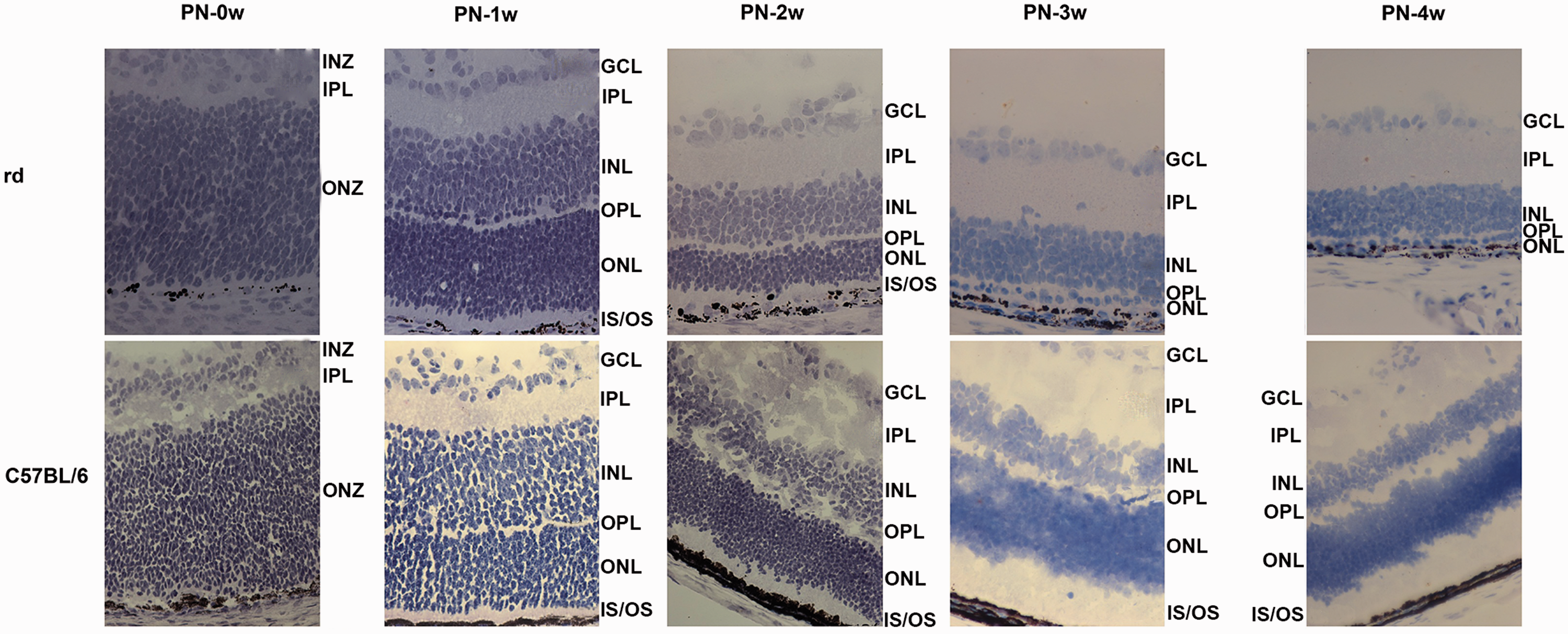
At PN-0 week when the retina was not completely developed, one broad neuroblastic zone flanking the inner plexiform layer (IPL) was observed both in rd and C57BL/6J mice (Figure 1). TrkB was strongly expressed in the central-outer neuroblastic zone, TrkC was mainly localized in the outer neuroblastic zone, including some cells near the IPL, and CNTFRα was strongly expressed both within and outside the neuroblastic zones (Figure 2).
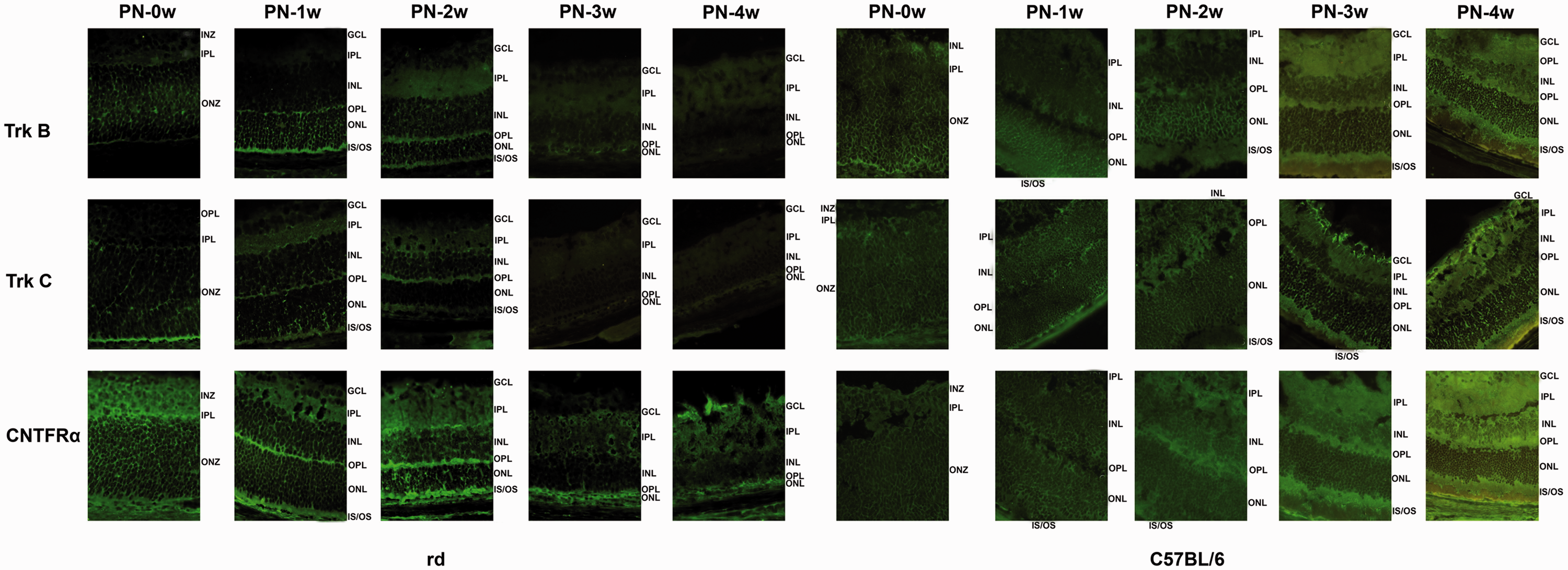
At PN-1 week, all retinal layers had clearly formed both in rd and normal control groups (Figure 1). TrkB was localized to inner and outer segments (IS/OS) and the outer nuclear layer (ONL), and limited immunoreactivity was detected in the outer plexiform layer (OPL) and inner nuclear layer (INL). TrkC was present in IS/OS, the ONL, OPL, INL, IPL, and ganglion cell layers (GCL). CNTFRα was expressed in IS/OS, the ONL, OPL, INL, IPL, and GCL (Figure 2).
The retina of rd mice began to atrophy at PN-2 weeks and the rods and cones disappeared, ultimately leaving only a line of ONL at PN-3 weeks (Figure 1). TrkB was labeled in IS/OS, the ONL, OPL, INL, and IPL, TrkC was expressed in IS/OS, the ONL, OPL, INL, and GCL, and CNTFRα was expressed in IS/OS, the ONL, OPL, INL, and GCL (Figure 2).
At PN-3 weeks, the retina of rd mice was thinner than that of normal controls (Figure 1). TrkB was present in the ONL and INL, and weakly detectable in the IPL and GCL. TrkC expression was barely visible. CNTFRα was strongly expressed in the ONL, OPL, INL, IPL, and GCL (Figure 2). At PN-4 weeks, the retina of rd mice showed atrophy, was thinner than that of C57BL/6 mice, and was not developing, while the retina of C57BL/6 appeared to still be developing (Figure 1). TrkB was expressed weakly in the IPL and GCL, TrkC showed no detectible expression, and CNTFRα was expressed in the ONL, OPL, and GCL (Figure 2). No reaction was observed in negative control samples.
Dynamic changes of Bax, Bcl-2, and LC3 after neurotrophic combination therapy
At PN-4 and PN-5 weeks, the relative mRNA and protein expression of Bax in the rd retina was significantly higher than in the C57BL/6J retina (P < 0.05). At PN-4 weeks, Bax expression was also significantly higher in the rd group than in the PBS group (P < 0.05), but there were no differences between the two groups at PN-5 weeks. Two weeks after treatment with 50 μg/L of drugs (rhBDNF, rhBDNF + rhCNTF, rhBDNF + rhNT-3, or rhBDNF + rhCNTF + rhNT-3), the relative mRNA expression of Bax was significantly increased in the rd retina of the rhBDNF group compared with rd and PBS groups at PN-4 and 5 weeks (P < 0.05); it was also downregulated in rhBDNF + rhCNTF and rhBDNF + rhNT-3 groups at PN-4 weeks and upregulated at PN-5 weeks. Two weeks after treatment with 100 μg/L of drugs (rhBDNF, rhBDNF + rhCNTF, rhBDNF + rhNT-3, or rhBDNF + rhCNTF + rhNT-3), the relative mRNA and protein expression of Bax was significantly decreased in the rd retina of rhBDNF + rhCNTF, rhBDNF + rhNT-3, groups compared with rd and PBS groups at PN-4 weeks (P < 0.05). At PN-5 weeks, the relative mRNA and protein expression of Bax was significantly decreased both in rhBDNF and combination drug groups (rhBDNF + rhCNTF, rhBDNF + rhNT-3, and rhBDNF + rhCNTF + rhNT-3) compared with rd and PBS groups (P < 0.05). Bax expression in the rhBDNF group was significantly higher than that of the rhBDNF + rhCNTF group at PN-4 weeks (P < 0.05) (Figures 3, 4).
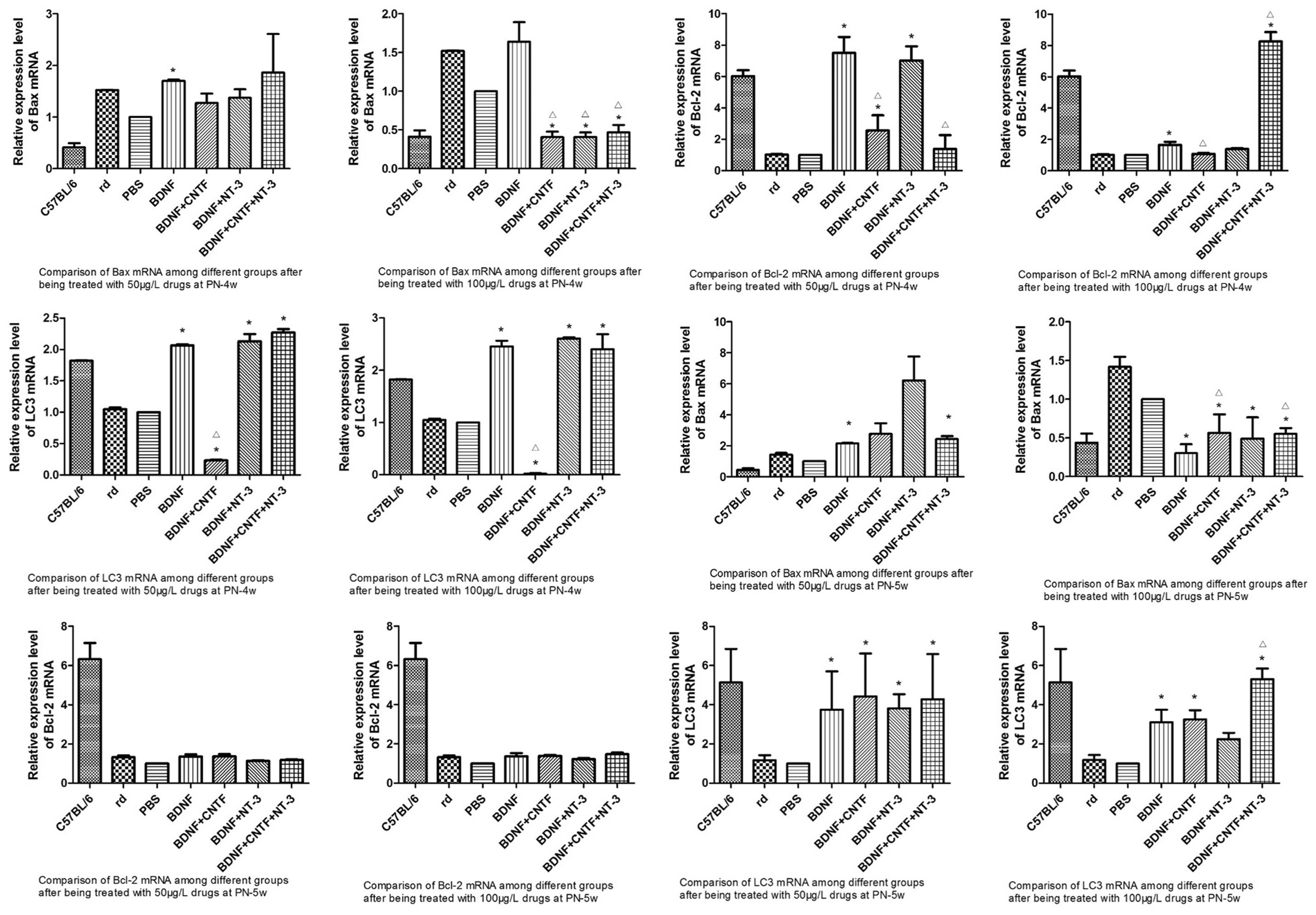

At PN-4 and PN-5 weeks, the relative mRNA and protein expression of Bcl-2 in the rd retina was significantly lower than in the C57BL/6J retina (P < 0.05). There were no differences between rd and PBS groups. Two weeks after treatment with 50 μg/L of drugs (rhBDNF, rhBDNF + rhCNTF, rhBDNF + rhNT-3, or rhBDNF + rhCNTF + rhNT-3), the relative mRNA expression of Bcl-2 in the rd retina was significantly increased in rhBDNF, rhBDNF + rhCNTF, and rhBDNF + rhNT-3 groups compared with rd and PBS groups at PN-4 weeks (P < 0.05), with the rhBDNF group being higher than rhBDNF + rhCNTF and rhBDNF + rhCNTF + rhNT-3 groups. However, protein expression levels were comparable between groups. Two weeks after treatment with 100 μg/L of drugs (rhBDNF, rhBDNF + rhCNTF, rhBDNF + rhNT-3, or rhBDNF + rhCNTF + rhNT-3), the relative mRNA and protein expression of Bcl-2 in the rd retina was significantly higher in rhBDNF + rhCNTF + rhNT-3 groups compared with rd and PBS groups at PN-4 weeks, with the rhBDNF + rhCNTF + rhNT-3 group being higher than the rhBDNF group (P < 0.05). There were no significant differences among other groups (Figures 3, 4).
At PN-4 and PN-5 weeks, the relative mRNA and protein expression of LC3 in the rd retina was significantly lower than in the C57BL/6J retina (P < 0.05). There were no differences between rd and PBS groups. Two weeks after treatment with 50 μg/L of drugs (rhBDNF + rhNT-3 and rhBDNF + rhCNTF + rhNT-3), the relative mRNA and protein expression of LC3 in the rd retina was significantly higher than in rd and PBS groups (P < 0.05). At PN-4 weeks, LC3 mRNA expression was significantly decreased in the rhBDNF + rhCNTF group compared with rd and PBS groups (P < 0.05). Two weeks after treatment with 100 μg/L of drugs (rhBDNF, rhBDNF + rhCNTF, rhBDNF + rhNT-3, or rhBDNF + rhCNTF + rhNT-3), the relative mRNA and protein expression of LC3 in the rd retina was significantly higher in the rhBDNF + rhCNTF + rhNT-3 group compared with rd and PBS groups at both PN-4 and 5 weeks, but LC3 mRNA was significantly decreased in the rhBDNF + rhCNTF group (P < 0.05). LC3 expression in the rhBDNF + rhCNTF + rhNT-3 group was significantly higher than in the rhBDNF group (P < 0.05; Figures 3,4).
Ultrastructural changes after neurotrophic combinative therapy
At PN-4 and PN-5 weeks after drug treatment, some ultrastructural changes were seen in rd retinas. Two weeks after injection, the IS/OS were barely visible, but many ribosomes were observed between the retinal pigment epithelium and the ONL. Slightly higher numbers of damaged organelles and swollen mitochondria were seen in the INL and GCL compared with pre-treatment retinas. The mitochondria and ribosomes in the INL and GCL showed a slightly improved morphology, with no obvious membrane segregation, reduced vacuolization and plasma destruction, and reduced damage and swelling of mitochondria. Improved synaptic structure was also seen in the IPL (Figure 5).

Discussion
In the present study, the structure of the rd retina and the expression patterns of TrkB, TrkC, and CNTFRα were the same as in normal mice in the first 2 weeks after birth. The retina then began to atrophy and the photoreceptors began to undergo apoptosis after PN-2 weeks, with lower expression of TrkB and TrkC receptors observed from PN-3 weeks onward. TrkB was expressed in the IS/OS, ONL, OPL, INL, and IPL after full development and was still expressed in the ONL, INL, IPL, and GCL after photoreceptor apoptosis.
BDNF may improve visual function in RP animal models, and long-term interventions of BDNF appear to protect photoreceptors in animal models with retinal light damage or inherited retinal degeneration. 4 BDNF functions by binding to a high affinity receptor (TrkB) or a low affinity receptor (p75NTR). The effects of the latter have been disputed, so BDNF effects are thought to be mainly mediated by two key signaling pathways induced via TrkB, including mitogen-activated protein kinases and phosphatidylinositol 3-kinase (PI3K)/Akt. 14 Single treatments of BDNF and NT-3 were more effective at treating degenerative models induced by light damage.
CNTF and NT-3 both promote the survival of retinal neurons, exerting protective effects during retinal development and after retinal injury. 5 NT-3 may specifically bind TrkC, then trigger a series of reactions for neurite outgrowth, neuron development, differentiation, survival, cytoskeleton remodeling, and synapse formation. 15 CNTF appears to activate signal transduction by binding to a complex consisting of CNTFRα (which controls retinal development and function), 16 glycoprotein 130, and leukemia inhibitory factor-β.
Endogenous BDNF has neuroprotective effects on several neural tissues. For example, intrinsic BDNF/ cAMP response element-binding protein (CREB) signaling plays an important role against the apoptosis, inflammation, and oxidative stress induced by nicotine-induced neurotoxicity, 17 and endogenous BDNF was reported to increase Bcl-2 protein levels and reduce Bax levels. 18 , 19 Light-induced retinal injury and some treatments were also shown to promote the intrinsic generation of BDNF and CNTF, which upregulates Bcl-2 and downregulates Bax, 20 , 21 while the systemic administration of CNTF promoted the neuroprotection of motor neurons through increasing Bcl-2 and decreasing Bax. 22 Moreover, a single intravitreal injection of CNTF protein was found to increase the Bcl-2/Bax ratio in the retina after laser injury to improve cone survival, 23 and single CNTF use protected photoreceptors of animal models with inherited retinal degeneration. 24 , 25 NT-3 regulates CREB phosphorylation and affects the levels of Bcl-2 in oligodendrocyte progenitor cells, thus playing an important role in linking proliferation and survival pathways in oligodendrocyte progenitors. 26
TrkB has several isoforms, which are temporally and spatially differentially expressed in Müller cells. BDNF transfers signals and protects photoreceptors under stress conditions by recruiting appropriate receptor isoforms and then triggering dimerization of the receptors. 25 BDNF-TrkB signaling stimulates the Müller cells to become endogenous retinal progenitor cells and then proliferate and differentiate in the degenerating retina for photoreceptor rescue. 27 There are differences in TrkB expression among retinas of different species. In the developing mouse retina, TrkB is expressed in the inner part of the neuroblastic zone and IPL in both rd/rd mice and C57BL/6J mice at 2 days after birth. At the end of the first 2 weeks, immunohistochemistry has shown that TrkB is present in the OPL, INL, IPL, several cells of the GCL, and bundles of axons that form the optic nerve in both mouse groups. TrkB-like immunoreactivity is also seen in the outer and inner segments of photoreceptors. 20 The location of expression in zebrafish retina does not change from embryo to maturity, but the intensity is altered. 28 BDNF/TrkB signaling is essential for the development and maintenance of synaptic connections between photoreceptors and bipolar cells, as well as the dopaminergic network. 20
TrkC is expressed in IS/OS, the ONL, OPL, INL, and GCL after full development, and is barely visible after photoreceptor apoptosis. Previous research on mouse retinas showed it to be expressed in the INL, amacrine, and GCL of wild-type mice and weakly expressed in the INL and GCL of rd/rd mice 2 days PN. TrkC is also expressed in the OPL, IPL, and GCL 4 days PN, with rd/rd mice having higher expression than wild-type. TrkC-like immunoreactivity is also found in the outer and inner segments of photoreceptor cells. Expression is higher at PN-4 weeks in rd/rd than wild-type mice, possibly because the retinal neurons have largely died at this point, resulting in upregulation of the receptors at PN-3 weeks. 20 In pigeon retina, there is no expression of TrkC before hatching, whereas expression in the GCL increases after hatching and TrkC is ultimately expressed in retinal ganglion cells and the IPL. This pattern is related to NT-3 regulation of retino-tectal development and synapse formation. 29
At PN-3W and 4W points of the rd mouse model, TrkB is expressed weakly and TrkC shows no detectible expression. mRNA and protein levels of neurotrophic factors (nerve growth factor, BDNF, NT-3, and glial cell line-derived neurotrophic factor) and their receptors (TrkA, TrkB, and TrkC) previously showed marked and persistent down-regulation in the rat retina from 3 days after permanent bilateral common carotid artery occlusion surgery. 30 This may explain the changes in TrkB and TrkC expression induced by RP in our study, and we plan to determine whether TrkB and TrkC expression is up-regulated in rd mice treated with BDNF and NT-3 in a future study.
CNTFRα is expressed in IS/OS, the ONL, OPL, INL, and GCL after full development and remains detectable in the ONL, OPL, INL, IPL, and GCL after photoreceptor apoptosis. It has been defined as the specific receptor required for CNTF binding and signaling, yet its location in the retina is still controversial. Some researchers have shown that CNTFRα is expressed in the mature photoreceptors of mice or in the immature photoreceptors of rats, 31 , 32 while others detected it in the photoreceptors of adult non-rodent mammalian retinas. 16 CNTF acts either directly on photoreceptor precursors through CNTFRα or indirectly through receptors expressed in Müller cells. 32 , 33 In the rd retina of the present study, the location of the three retinal receptors in the was different and overlapping, which made the combinative use of their ligand reasonable.
In the present study, Bax expression was decreased at transcriptional and translational levels in rhBDNF, rhBDNF + rhCNTF, rhBDNF + rhNT-3, and rhBDNF + rhCNTF + rhNT-3 groups 2 weeks after injection with 100 μg/L of drugs at PN-3 weeks, while Bcl-2 mRNA and protein levels were increased in the rhBDNF + rhCNTF + rhNT-3 group after injection with the same dose at PN-2 weeks. These results indicate that the combined use of neurotrophic factors may affect apoptosis, especially with rhBDNF and rhNT-3 at a higher dosage.
Bcl-2 is one of the most important anti-apoptosis proteins. It is mainly located in the mitochondrial outer membrane, nuclear membrane, and endoplasmic reticulum, where it blocks cytochrome C release into the cytoplasm then inhibits a series of apoptotic reactions as caspase-9. As a pro-apoptosis protein, Bax binds to the homologous structural domain of Bcl-2 to form a heterodimer, blocking the biological activities of Bcl-2. Balancing Bcl-2 and Bax levels is important in maintaining mitochondrial homeostasis, 34 , 35 and the Bcl-2 to Bax ratio also plays a key role in neuron survival; indeed the ratio is altered in many degenerative fundus diseases such as Leber’s and glaucoma.36–40
Programmed cell death, or apoptosis, is critical to both tissue development and maintenance, as well as to the destruction of injured cells. (BCL-2) family proteins, including both pro- (e.g., BCL-2-associated X protein (BAX)) and anti- (e.g., BCL-2) apoptotic proteins, are major regulators of the apoptotic process. 41 A diverse range of death signals leads to a shift in the balance of anti- and pro-apoptotic BCL-2 family members. 42 Autophagy is a highly conserved catabolic process in which cellular components are degraded through autophagosome formation, and it is involved in a number of human diseases and physiological processes43–46such as retinal degeneration and sphingolipid disorders. 47 It is also related to photoreceptor activities such as membrane disc metabolism, rhodopsin circulation, and visual cycle formation. 48
Recently, parts of the autophagy machinery, particularly microtubule-associated protein 1 light chain 3 (LC3), were found to be specifically recruited to phagosomal membranes resulting in phagosome–lysosome fusion and efficient degradation of internalized cargo. 49 LC3 is the mammalian homolog of Atg8 and is located on the inner membrane of the autophagosome. 50 An increase in the rate of cytosolic LC3-I conversion to LC3-II is commonly used as a marker for the autophagosome, which reflects autophagic activity.45,51 LC3-II levels are increased under conditions of increased autophagic initiation or decreased autophagic degradation or a combination of the two. 52 BDNF has been shown to promote neuronal survival through the modulation of autophagy, and there is a crossover between the pathways whereby BDNF affects autophagy and other biological processes. 52
In the present study, LC3 expression was increased both at the transcriptional and translational levels in rhBDNF + rhNT-3 and rhBDNF + rhCNTF + rhNT-3 groups 2 weeks after injection with two doses of drugs. This indicated that the combined administration of neurotrophic factors may be more effective at altering autophagic conditions in the rd retina than a single neurotrophic factor alone. BDNF is essential for neuroprotection as well as neuroregeneration because of its role in neurogenesis and the formation of new dendrites as well as in neuro-plasticity, which requires a suitable degradation process to remove unnecessary and undesirable connections. 53 It has also been found that BDNF can trigger the mammalian target of rapamycin (mTOR) signaling pathway to affect autophagy processes, then activate neurons for autophagy-related synaptic remodeling in other neurodegenerative diseases. Increased BDNF signaling therefore enhances autophagic flux, promoting the survival of degenerative neurons. 54 BDNF has also been shown to upregulate autophagy through the PI3K-Akt pathway, which is upstream of mTOR. LC3 upregulation is related to Akt/mTOR/p70S6K pathways by BDNF. 55
In conclusion, we observed that the the three neurotrophic factor receptors localized to different areas during retina development and disease progression. The combined use of neurotrophic factors may dynamically change Bax, Bcl-2, and LC3 expression and improve the rd retinal ultrastructure.
Supplemental Material
sj-pdf-1-imr-10.1177_0300060520967833 - Supplemental material for Impact of neurotrophic factors combination therapy on retinitis pigmentosa
Supplemental material, sj-pdf-1-imr-10.1177_0300060520967833 for Impact of neurotrophic factors combination therapy on retinitis pigmentosa by Xiaobei Yin, Ting He, Rui Chen, Hui Cui and Genlin Li in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_0300060520967833 - Supplemental material for Impact of neurotrophic factors combination therapy on retinitis pigmentosa
Supplemental material, sj-pdf-2-imr-10.1177_0300060520967833 for Impact of neurotrophic factors combination therapy on retinitis pigmentosa by Xiaobei Yin, Ting He, Rui Chen, Hui Cui and Genlin Li in Journal of International Medical Research
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
