Abstract
Objective
Literature on trial of labor after cesarean section (TOLAC) in women with isthmoceles is scarce because of complications associated with the procedure. This study investigated TOLAC's safety and feasibility in patients with isthmoceles.
Methods
The study group comprised 34 pregnant women with isthmoceles who vaginally delivered. The control group comprised 102 pregnant women without isthmoceles who vaginally delivered during the same period. Scar diverticula were measured using color Doppler ultrasonography; between-group delivery outcomes were compared.
Results
Of the study group patients, 27/34 had isthmoceles diagnosed by ultrasound before pregnancy. Nineteen (70.37%) of these patients had mild defects and eight (29.63%) had moderate defects. The scar diverticula's mean length, depth, and width were 1.05 ± 0.62, 0.54 ± 0.28, and 1.20 ± 0.70 cm, respectively. The residual muscle layer's mean thickness was 0.27 ± 0.07 cm. The mean diverticulum depth/residual muscular thickness ratio was 2.39 ± 2.58. The duration of the first stage of labor was significantly shorter and the neonatal weight was significantly lower in the study group than control group.
Conclusion
Successful vaginal delivery is possible for women with mild and moderate isthmoceles. Further large-scale studies are needed to improve TOLAC's safety in pregnant women with isthmoceles.
Keywords
Introduction
Cesarean section (CS) rates have risen rapidly in recent years,1–4 reaching an average of 27.5% of all deliveries worldwide. 5 This trend is particularly evident in Asian countries irrespective of the geographical region, patients’ socioeconomic groups, or type of treating hospital. 6 A recent study showed that the overall CS rate in China increased from 28.8% in 2008 to 50.8% in 2014. 7 Recent changes in the family planning policy have also contributed to a significant increase in the number of multiparous women with a scarred uterus resulting from a previous CS. 8
Evidence suggests that this increase in the rate of CS not only has deleterious effects on maternal and fetal well-being 9 , 10 but is also associated with a declining rate of vaginal birth after CS (VBAC). This is due to a higher incidence of short- and long-term complications such as uterine rupture, pathologically adherent placenta in subsequent pregnancies, and development of a cesarean scar defect (also known as an isthmocele) in patients with VBAC. 11 , 12 An isthmocele, also known as a diverticulum or niche, is defined as a reservoir-like pouch on the anterior wall of the uterine isthmus at the presumed site of the incision. 13 It develops because of poor local healing of the uterine incision and absence of myometrial layer continuity after CS. 14 An isthmocele may lead to several gynecological signs and symptoms such as abnormal uterine bleeding (AUB), secondary infertility, and ectopic pregnancy (cesarean scar pregnancy).15–18 It is mainly an ultrasonographic finding and can be diagnosed by several methods including transvaginal ultrasound, saline infusion sonohysterography, hysterosalpingography, hysteroscopy, and magnetic resonance imaging. 19 The severity of an isthmocele can be determined using several criteria such as the number and size of the diverticula, 20 , 21 the ratio between the myometrial thickness at the level of the defect and the thickness of the adjacent myometrium, 22 and manifestation of clinical symptoms such as AUB. 23
Because of the disruption in the myometrium at the site of the cesarean scar, an unrepaired isthmocele is considered to be a risk factor for potential uterine rupture and is one of the greatest concerns when attempting a trial of labor after CS (TOLAC). 24 Owing to the high risk of complications, TOLAC usually requires women to fulfill several strict criteria such as an interval of ≥12 months between CS and the next conception, a vertex presentation, a single lower uterine segment (LUS) transverse scar, a radiologically adequate pelvis, a low fetal weight (<3500 g), and the absence of other medical or obstetrical factors. 25 , 26 Pregnant women with an unrepaired isthmocele, especially those with severe defects, are at a higher risk of uterine rupture.
Literature on TOLAC in women diagnosed with isthmoceles is extremely scarce because of the associated complications. Despite strict guidelines, however, some reports have described successful vaginal deliveries in patients with an unknown uterine scar. 27 Smith et al. 27 demonstrated that pregnant women with an unknown scar have lower odds of uterine rupture than women with a known low transverse scar. Because isthmoceles are often underdiagnosed, the development of strategies to further improve the safety of TOLAC in such patients is important to gain a better understanding of the possible impact of an undiagnosed isthmocele on the obstetrical outcome. Therefore, this retrospective study was performed to assess the safety and feasibility of TOLAC in patients with unrepaired isthmoceles.
Materials and methods
Study design and population
All procedures involving human participants in this study were performed in accordance with the standards of the ethics committee of the Fujian Maternity and Child Health Hospital, Affiliated Hospital of Fujian Medical University (No. 2018Y0005) and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Written informed consent was obtained from all patients prior to the surgical procedures.
We retrospectively reviewed the medical records of women with isthmoceles who underwent delivery in Fujian Maternal and Child Health Hospital from January 2015 to December 2018. Isthmoceles were diagnosed by vaginal ultrasonography during non-pregnancy or early pregnancy (within 10 weeks of pregnancy) based on the presence of a fluid sonolucent area in the LUS at the incision site of the previous CS, which was connected to the uterine cavity. 14 Pregnant women with imaging-confirmed isthmoceles who successfully underwent vaginal delivery post-CS were selected as the study group. The control group comprised pregnant women with previous CS without isthmoceles who underwent VBAC within the same time period. The rate of CS at the hospital was 36% during the study period.
The inclusion criteria were a history of one CS in the LUS, a record of transvaginal or abdominal ultrasonography of the myometrium of the LUS performed in our hospital during non-pregnancy or early pregnancy (within 10 weeks of pregnancy) or after delivery, regular prenatal care performed in our hospital, and follow-up until after delivery. Patients who did not undergo transvaginal or abdominal ultrasonography in early pregnancy were excluded from the study. We also excluded patients with other complications or comorbidities in the current pregnancy, such as severe gestational hypertension, maternal heart disease, intrahepatic cholestasis syndrome, placental abruption, and placenta previa, because these conditions may affect vaginal delivery and interfere with labor.
Data collection
All data were retrieved from the patients’ medical records. Data regarding age, time since last pregnancy, gravidity, parity, pre-pregnancy body mass index, and weight gain during pregnancy were collected to assess the general condition of the women in both groups. In the study group, we assessed the details of the patients’ symptoms, number and size of scar diverticula, thickness of the residual myometrial layer [i.e., residual myometrial thickness (RMT)], and ratio of the diverticulum depth to the RMT. In addition, to evaluate the intrapartum conditions and delivery outcomes in both groups, we collected data on the thickness of the lower uterine myometrial layer measured within 2 weeks before delivery, gestational age at delivery, duration of the three stages of labor, incidence of fetal distress, forceps delivery rate, intrapartum blood loss, neonatal birth weight, neonatal 1-minute Apgar score, neonatal asphyxia rate, and admission rate to the neonatal intensive care unit (NICU). To assess uterine rupture, the uterine cavity was routinely palpated by hand along the anterior uterine wall after delivery of the placenta to detect any breach or depression. If any abnormality was found, color Doppler ultrasound was used to confirm the rupture.
Grading of isthmoceles
Isthmoceles were scored based on the RMT; percentage of the residual myometrial layer (PRM), defined as the ratio between the myometrial thickness at the level of the defect and the thickness of the adjacent myometrium; AUB; number and size of the diverticula; and CS number (Table 1). The final score was used to grade each isthmocele as mild (2–3 points), moderate (4–6 points), or severe (7–9 points) as previously described. 23
Grading of isthmoceles.
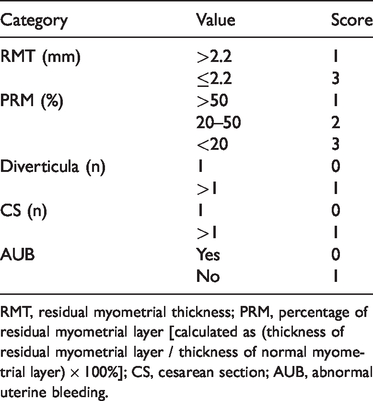
RMT, residual myometrial thickness; PRM, percentage of residual myometrial layer [calculated as (thickness of residual myometrial layer / thickness of normal myometrial layer) × 100%]; CS, cesarean section; AUB, abnormal uterine bleeding.
Statistical analysis
Normally distributed data are expressed as mean ± standard deviation, and enumeration data are expressed as frequency and percentage. Categorical data were compared using the chi-square test and Fisher’s exact test. The bivariate relationship of quantitative data was assessed using an unpaired Student’s t test. Statistical analyses were performed using IBM SPSS Statistics for Windows, Version 24.0 (IBM Corp., Armonk, NY, USA). Differences were considered statistically significant at P < 0.05.
Results
Characteristics of patients
The patients’ general characteristics are shown in Table 2. Of 38 women with isthmoceles who underwent delivery during the study period, 34 underwent vaginal delivery and 4 underwent elective CS because of occipital transverse positioning of the fetus. These four patients were therefore excluded from the study. The final study group comprised 34 women with a mean age of 31.94 ± 3.96 years. Among these 34 patients, isthmoceles were diagnosed during pre-pregnancy in 27 women and during the postpartum period in 7 women. The isthmoceles in these latter seven women were diagnosed immediately after delivery during a routine uterine examination and further confirmed by ultrasound.
Patients’ general characteristics.
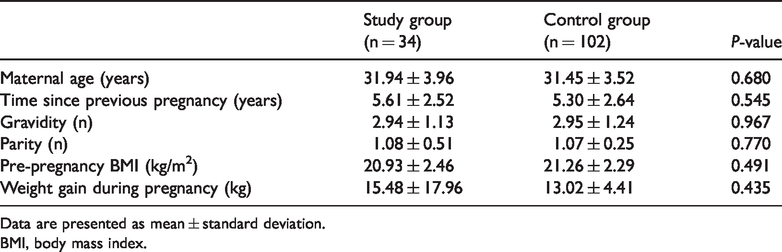
Data are presented as mean ± standard deviation.
BMI, body mass index.
The control group comprised 102 women with a mean age of 31.45 ± 3.52 years. There were no statistically significant differences in the interval between the previous CS and the subsequent pregnancy, gravidity, parity, pre-pregnancy body mass index, or weight gain during pregnancy between the study group and the control group (Table 2).
Surgical history and clinical symptoms
All patients in the study and control groups previously had a full-term pregnancy. One (2.94%) patient in the study group and seven (6.86%) patients in the control group had a history of trial of labor, with no statistically significant difference (Table 3). In the study group, 28 (82.35%) patients underwent surgery in tertiary hospitals, while 6 (17.65%) underwent surgery in private hospitals. The corresponding proportions in the control group were 81.37% and 18.63%, respectively. No patients in either group developed postpartum infections or postpartum blood loss. Five (14.7%) patients in the study group and eight (7.84%) patients in the control group reported menstrual changes after CS. No patients had a history of diverticulum repair because most patients were asymptomatic and the diverticula were only found after routine ultrasound.
Details of medical histories of study participants.

Data are presented as n (%).
*A tertiary hospital is defined as a cross-regional hospital with comprehensive and specialized medical care.
TOLAC, trial of labor after cesarean section; CS, cesarean section; CSD, cesarean scar defect.
Characteristics and grading of diverticula
The length, depth, width, and RMT of the scar diverticula measured by transvaginal ultrasonography for patients diagnosed before and after pregnancy are presented in Table 4. Based on the isthmocele grading system, of the 27 patients diagnosed pre-pregnancy, 3 (11.1%) patients had a score of 2, 16 (59.2%) patients had a score of 3, 3 (11.1%) patients had a score of 4, 3 (11.1%) patients had a score of 5, and 2 (7.5%) patients had a score of 6. Among the 27 patients, 19 (70.37%) were classified as having a mild defect (score of 2–3) and 8 (29.63%) as having a moderate defect (score of 4–6). Of the 27 patients in the study group, 6 had an RMT of ≤2.2 mm.
Dimensions of uterine scar diverticulum.
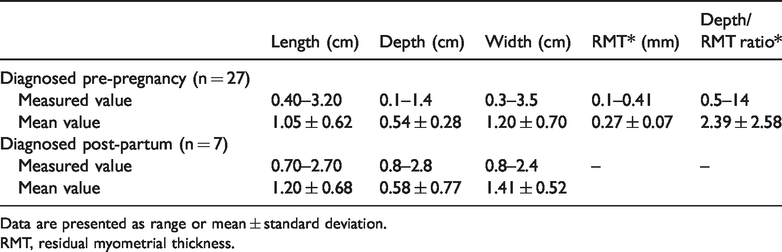
Data are presented as range or mean ± standard deviation.
RMT, residual myometrial thickness.
Comparison of intrapartum conditions and delivery outcomes
Of all 34 women in the study group, 33 had natural labor and 1 underwent induced abortion by ethacridine lactate at 37 weeks because of fetal death. There were no statistically significant differences in the uterine scar thickness or gestational age at delivery between the study group and control group (Table 5). The first stage of labor in the study group (224.88 ± 202.40 min) was significantly shorter than that in the control group (319.71 ± 217.84 min) (P = 0.027), but no such difference was noted for the second and third stages of labor or for postpartum blood loss.
Comparison of delivery outcomes between the study and control groups.
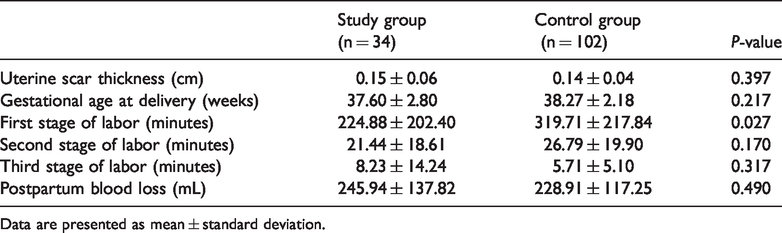
Data are presented as mean ± standard deviation.
The neonatal weight in the study group (2922.50 ± 599.51 g) was significantly lower than that in the control group (3185.68 ± 450.45 g) (P = 0.024) (Table 6). Two (5.88%) cases of fetal distress occurred in the study group compared with nine cases in the control group, but the difference was not statistically significant. Two (5.88%) neonates in the study group and eight (7.84%) in the control group underwent forceps delivery. There were no statistically significant differences in the neonatal Apgar score or neonatal asphyxia rate between the two groups. A higher number of neonates required NICU admission in the study group (n = 5, 14.71%) than in the control group (n = 2, 2.94%), but the difference was not statistically significant.
Comparison of delivery outcomes between the study and control groups.

Data are presented as n (%) or mean ± standard deviation.
NICU, neonatal intensive care unit.
Discussion
The primary finding of this study is that successful TOLAC can be achieved in women with mild and moderate isthmoceles. Our results also highlight the need for further development of strategies to improve the safety of TOLAC in pregnant women with possible isthmoceles, especially because isthmoceles are often underdiagnosed.
Characteristics and diagnosis of isthmoceles
No universal grading standard for the severity of isthmoceles has been established. One way to classify the defect is based on the size of its surface, with grade 1, 2, and 3 corresponding to a surface area of ≤15 mm2, 16 to 25 mm2, and >25 mm2, respectively 20 , 21 Other approaches are based on the ratio between the myometrial thickness at the level of the defect and the thickness of the adjacent myometrium, 22 or RMT. 14 A more detailed and complete system was recently proposed by Tower and Frishman, 23 wherein several factors are assessed simultaneously including the RMT, the PRM (remaining myometrial thickness / adjacent myometrial thickness), the number of CS scars, the CS number, and the menstrual conditions. Based on the total score, the isthmocele can then be classified as mild, moderate, or severe. 23 According to this classification system established by Tower and Frishman, 23 most patients in our study group were diagnosed with a mild or moderate defect, and no patients were diagnosed with a severe isthmocele.
In this study, transvaginal ultrasound was used to diagnose isthmoceles in the study group because this method allows the size of the diverticulum and the RMT to be measured. 22 Of all 34 women in the study group, 7 were diagnosed with an isthmocele by abdominal ultrasonography only after the delivery. Most isthmoceles are asymptomatic and incidentally detected by ultrasound. 14 Although transvaginal ultrasound is a reliable diagnostic tool, cases may be missed because of the small size of the diverticulum or insufficient awareness of isthmoceles among sonographers, leading to a missed diagnosis. 14 , 22 Such factors may have contributed to the missed diagnoses in our cohort as well. In addition to ultrasonography, hysteroscopy has also gained popularity not only for diagnosis but also for definitive management of isthmoceles in the past decade. A recent systematic review and meta-analysis demonstrated that hysteroscopy improves bleeding symptoms in about 80% of symptomatic patients. 21 However, the review failed to find evidence on the role of hysteroscopy in improving fertility or reducing the risk of obstetrical complications such as uterine rupture in asymptomatic women. 21 Despite several treatment options for scar diverticula, there are no standard guidelines for management. Treatment is often restricted to patients with symptoms and a reduced quality of life. 12 , 14 Only five patients had menstrual changes in our study group, and all had mild symptoms that did not affect their quality of life. Therefore, none of the patients included in our study were recommended to undergo surgery for the diverticulum.
Maternal and fetal outcomes after TOLAC in women with isthmoceles
In our study, TOLAC in women with isthmoceles did not affect the overall maternal outcomes. There were no reported incidences of uterine rupture in the study group, and there were no statistically significant differences in the postpartum blood loss between the study group and control group. Additionally, the isthmoceles had no impact on the length of the second and third stages of labor. However, we noticed a significantly shorter first stage of labor in the study group than in the control group (P < 0.05). A recent retrospective cohort study of 510 women demonstrated that women who underwent their first VBAC had a shorter first stage of labor, especially if the CS was performed in the advanced stage of labor, than did primiparous women who delivered vaginally. 28 Because we had no information about the exact timing of the previous CS in the participants of the current study, it is possible that the variations in CS timing accounted for the observed difference in the duration of the first stage. Further large-scale studies are required to accurately assess the possible impact of isthmoceles on labor progression and duration.
In our study, the gestational age at delivery was similar in the study and control groups. In the study group, a single case of stillbirth was diagnosed at 37 weeks. The patient had a scarred uterus and gestational diabetes, and she underwent induced labor with no complications. In terms of fetal outcomes, the neonatal weight was significantly lower in the study group than in the control group (P < 0.05). This observation is in agreement with previous reports suggesting that thinning of the LUS in women is strongly correlated with lower neonatal birth weight. 29 Because isthmoceles are associated with a markedly thinned residual myometrium in the LUS,30–32 it is possible that this thinning accounts for the observed differences in neonatal weight. Moreover, several studies have indicated that the decrease in LUS thickness may have a negative impact on fetal outcomes. In a study by Gupta et al., 33 pregnant women with a thinner LUS delivered neonates with significantly lower Apgar scores. Although our study showed no differences in the rate of fetal distress, the rate of neonatal asphyxia, or the 1-minute Apgar scores between the study and control groups, we observed a 2.5-fold increase in the rate of neonatal NICU admission associated with TOLAC in women with isthmoceles. Although this difference was not statistically significant, it is important to note that the number of participants in the study group was smaller than that in the control group. Further studies with larger sample sizes are needed to assess the impact of TOLAC in women with isthmoceles on NICU admission rates.
Prediction of uterine rupture
The primary manifestations of isthmoceles include thinning of the myometrial layer in the lower uterus, scar separation in the lower uterus, and partial or complete uterine rupture. These complications can occur sequentially or independent of one another. 34 A recent meta-analysis examining the uterine thickness after prior CS concluded that a lower uterine thickness of <2 mm suggests an increased risk of uterine rupture, 35 , 36 highlighting the importance of measuring the uterine thickness, especially within the lower uterus, after a previous CS. The literature also suggests that the size of a patient’s isthmocele can impact the risk of future complications; specifically, large diverticula (RMT of ≤2.2 mm) can increase the risk of scar dehiscence up to 42.9%. 37 However, because of the limited number of patients in our study, this could not be verified. The most recent consensus is that the only parameter that can accurately predict CS scar dehiscence in a subsequent pregnancy is the ratio of the isthmocele depth to the RMT. 31 A depth/RMT ratio of <0.785 correlates with a minimal probability of scar separation, and the risk of scar rupture increases to >50% when the depth/RMT ratio is >1.30.30,31
Color Doppler ultrasonography in all 34 pregnant women in our study group did not suggest scar separation in the lower uterus, and there were no reported uterine ruptures as a result of TOLAC. There were no detected changes in the uterine scar thickness (compared with pre-pregnancy), indicating good quality of uterine scar healing. However, the depth/RMT ratio in the study group was <1.0 in 5 patients and >1.3 in 22 patients, with a mean value of 2.39 ± 2.58. This further strengthens the observation that while uterine rupture is considered the major complication of isthmoceles, it is still a relatively low-probability event. Ultrasonographic measurements can also be affected by various factors such as the experience of the operator, measurement positions, measurement methods (transvaginal or abdominal), degree of bladder filling, and fetal presentation. 38 In our study, 7 of 34 women were diagnosed with an isthmocele by abdominal ultrasonography only after the delivery. Because of the difference in the sample sizes of the two groups in this study and the potential differences in measurements obtained by different sonographers, we believe that multicenter, large-sample studies are needed to further investigate the correlation between isthmoceles and the risk of scar dehiscence. Further control regarding the methodology of isthmocele diagnosis (transvaginal vs. abdominal) is also an important variable to be considered in future studies.
The current study has two main limitations. First, given the retrospective nature of the study, data on potential confounding variables were unavailable. These variables include maternal height and any history of previous vaginal deliveries, which are known to be associated with the success rate of TOLAC. Second, no unified, standard method of scar thickness measurement has been established to date. 39 As a result, this measurement can be affected by several factors, such as the measuring time and position, the method of detection (abdominal vs. transvaginal ultrasonography), and the qualifications of the ultrasound specialists.
Conclusion
Our results suggest that successful TOLAC can be achieved in women with mild and moderate isthmoceles. However, because isthmoceles are often underdiagnosed, it is important to develop strategies to further improve the safety of TOLAC in pregnant women with potential isthmoceles. Such strategies include early prenatal diagnosis, accurate assessment of changes in pregnant women’s conditions, well-monitored maternal weight gain, diversified comprehensive management, and development of an individualized delivery plan for strict monitoring and timely prevention and treatment of uterine rupture during labor.
Footnotes
Acknowledgements
The authors acknowledge support from the study participants and their families as well as the staff at the Department of Obstetrics and Ultrasound of Fujian Maternity and Child Health Hospital, Affiliated Hospital of Fujian Medical University.
Authors’ contributions
XQ Zheng was involved in the project development, data collection, and major manuscript writing. JY Yan and ZZ Liu were involved in the project development and manuscript editing. XC Wang was involved in the project development, data analysis, and manuscript writing. RL Xu and LY Li were involved in the manuscript editing. Z Lin, LH Zheng, M Liu, and Y Chen were involved in the data collection. All authors read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research was supported by National Health and Family Planning Commission Science Foundation (2019-WJ-04), Fujian Science and Technology Project (2018Y0005), Fujian Provincial Health Technology Project (2017-CX-11), and Key Clinical Specialty Discipline Construction of Fujian, P.R.C. ([2015] no. 593).
