Abstract
Objective
Reactive oxygen species (ROS) production by neutrophils induces pulmonary endothelial cell damage and results in acute lung injury (ALI). We previously reported that deferasirox (DFS), an iron-chelating agent, inhibits the ROS production and neutrophil extracellular trap (NET) formation induced by phorbol myristate acetate and formylmethionylleucylphenylalanine
Methods
After DFS administration for 7 days, ALI was induced in mice by LPS via intratracheal administration.
Results
LPS treatment induced neutrophil invasion in the lung tissues, along with NET formation and a significant increase in the quantity of double-stranded DNA in the bronchoalveolar lavage fluid, while pre-administered DFS inhibited these phenomena. However, alteration of neutrophil morphology in the cytoplasm in terms of shape and vacuolization was not inhibited by the pre-administration of DFS, possibly through ROS production.
Conclusions
DFS suppressed neutrophil invasion into lung tissues and reduced the double-stranded DNA content released by the neutrophils. These results suggest that DFS can potentially be used to prevent diseases related to neutrophil activation including ALI, thrombosis, and vascular endothelial dysfunction.
Keywords
Introduction
Neutrophils play an important role in immune function. 1 Activated neutrophils bind to endothelial cells and produce reactive oxygen species (ROS), resulting in injury. Redundant neutrophil activation induces various pathological conditions, including acute lung injury (ALI). The inhibitory effect of deferasirox on ROS production may help to prevent ALI development. 2
Deferasirox (DFS, Exjade; Novartis Pharma, Basel, Switzerland), an oral iron chelator, has been reported to exert biological effects via mechanisms other than iron chelation. These mechanisms include inhibiting myeloid leukemia cell growth by suppressing the mammalian target of rapamycin signaling, 3 disrupting the growth of solid tumors in xenograft models, 4 preventing the nuclear translocation of nuclear factor-κB,2,5 and reducing ROS production.2,5–8 However, neutrophil vacuoles are formed by the fusion of endosomes with autophagosomes and secondary granules in a ROS-dependent manner.9,10 The addition of agonists such as phorbol myristate acetate results in the formation of autophagosomes independent of phagocytosis, leading to subsequent vacuolization.11,12
Neutrophil extracellular traps (NETs) are reportedly involved in the development of ALI. 13 NETs are webs of long chromatin fibers decorated with granule proteins that are released by neutrophils, and these webs trap and kill pathogens in the blood and tissues. 14 They have been reported to occur in response to the generation of high levels of ROS by nicotinamide adenine dinucleotide phosphate oxidase. 15 Neutrophils with NETs can be identified by immunostaining using antibodies against the citrullinated histones in the neutrophil nucleus. 16 Moreover, NETs can be quantified by measuring the release of double-stranded DNA (dsDNA). 17
Using an
Materials and methods
Animals
Ten-week-old, male C57BL/6J mice (Oriental Yeast, Tokyo, Japan) were maintained in sterilized cages with a 12-hour light/dark cycle. Three mice were used per treatment group. The animals were provided with pellet chow and sterilized tap water
Blood collection
Ethylenediaminetetraacetic acid (EDTA)-anticoagulated peripheral blood was collected from the heart and analyzed using an XN-2000 hematology analyzer (Sysmex, Kobe, Japan). Platelet-poor plasma samples were prepared by centrifugation at 700 ×
BALF collection
BALF was collected from the right lung with 0.05 mM EDTA and 0.1% bovine serum albumin in phosphate-buffered saline (PBS) and analyzed using the XN-2000. Supernatant samples and precipitated cells were prepared by centrifugation at 700 ×
Transmission electron microscopy (TEM)
For TEM analysis, neutrophils separated from the blood, and cells precipitated from the BALF, were fixed in 1% glutaraldehyde (Electron Microscopy Sciences, Hatfield, PA, USA) in PBS for 16 hours at 4°C. The fixed cells were then attached to silanized glass slides using Cytospin (Thermo Fisher Scientific, Rockford, IL, USA) and post-fixed in 1% osmium tetroxide for 45 minutes at 4°C. Following osmium fixation, the samples were dehydrated in a graded series of ethanol and invert-embedded in Quetol 812 (Nisshin EM, Tokyo, Japan). The samples were then cut into sections with a thickness of 80 nm to 100 nm using the Ultracut UCT ultramicrotome (Leica Microsystems, Wetzlar, Germany) and observed using an H-7500 TEM system (Hitachi High-Technologies, Tokyo, Japan).
dsDNA measurement
The dsDNA content in platelet-poor plasma from the blood and supernatant from the BALF was measured using an AccuBlue High Sensitivity dsDNA Quantitation Kit (31006; Biotium, Hayward, CA, USA).
Lung tissue
The left lung was collected, fixed with 4% paraformaldehyde in PBS, and embedded in paraffin. Paraffin sections were prepared and deparaffinized in xylene and a graded series of ethanol. The sections were subjected to hematoxylin (Mayer’s Hematoxylin Solution, 131-09665; Fujifilm Wako Pure Chemical, Osaka, Japan) and eosin (1% Eosin Y Solution, 051-06515; Fujifilm Wako Pure Chemical) staining, as well as immunohistochemical staining for citrullinated histones with an anti-histone H3 antibody at 10 mg/L (ab5103; Abcam, Cambridge, MA, USA).
Statistical analysis
Normal data distribution was confirmed by performing the Kolmogorov–Smirnov test and the F-test. Welch’s t-test was employed for comparisons between two groups. EZR version 1.32 software (Jichi Medical University, Tokyo, Japan) was used for all statistical analyses. A
Results
Neutrophil morphology
Only macrophages were observed in the BALF of mice without LPS administration regardless of DFS administration (data not shown). In the LPS-administered mice, the neutrophils changed into an active shape and vacuolization in the cytoplasm was observed (Figure 1a, b). These neutrophils were noticeably different in morphology from those in the control mice, which had a stationary shape (Figure 1c). DFS administration had no significant effect on neutrophil shape.
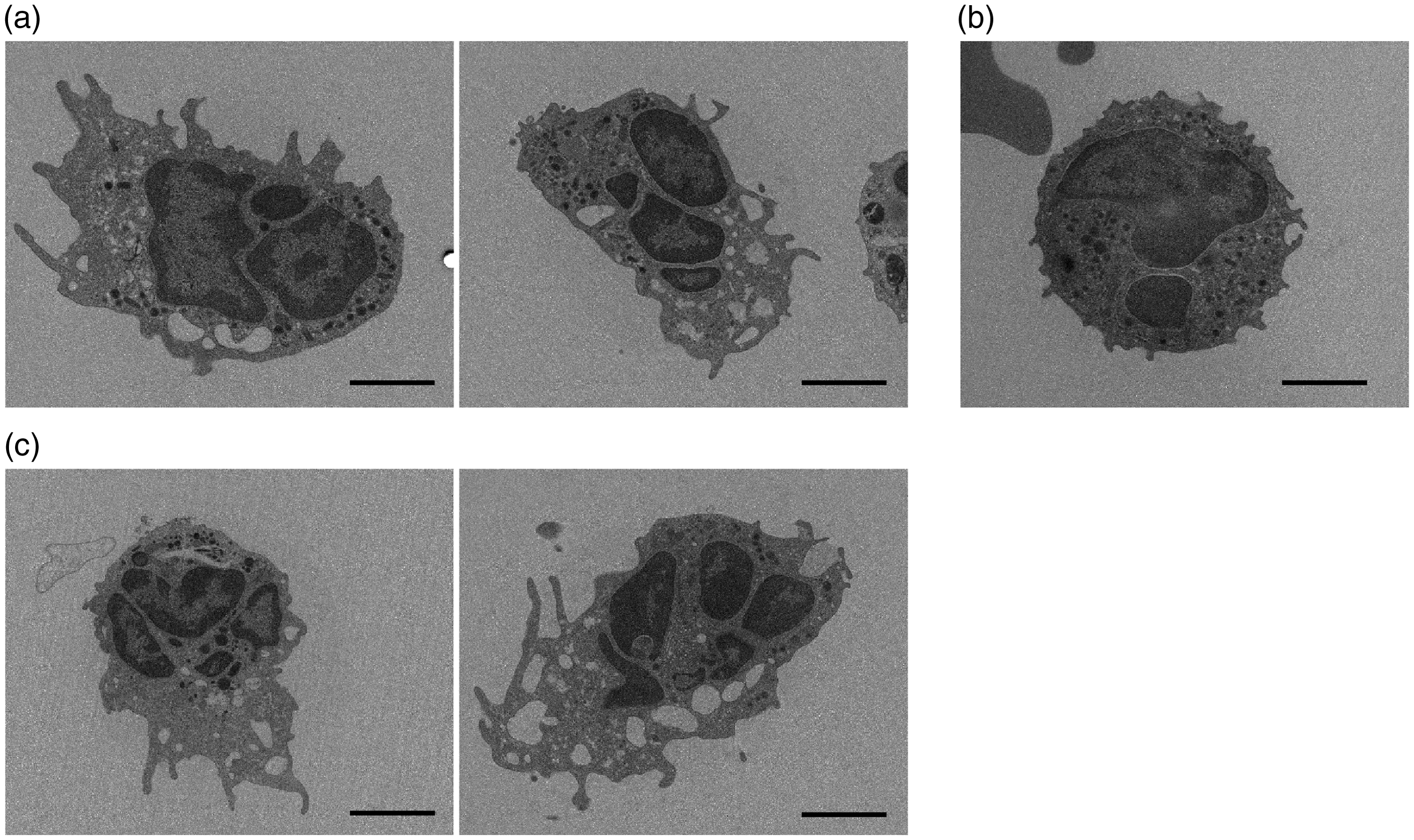
Transmission electron microscopy observation of cells in the bronchiolar lavage fluid (BALF) of acute lung injury (ALI)-induced mice. Neutrophils in the BALF of (a) ALI-induced mice treated with PBS, (b) ALI-induced mice treated with deferasirox, and (c) the control mice. Scale bars = 2 µm.
White blood cell (WBC) count and dsDNA content
WBCs were counted in the BALF using an XN-2000 apparatus. LPS induced a 10-fold increase (

White blood cell (WBC) count and dsDNA content in the bronchiolar lavage fluid (BALF) and whole blood of acute lung injury (ALI)-induced mice. Mice were pre-administered with deferasirox or PBS, and ALI was then induced by the intratracheal administration of lipopolysaccharides. (a) WBC count in the BALF. (b) dsDNA content in the BALF. (c) dsDNA content in the whole blood. Data represent the mean ± SD,
Hematoxylin and eosin staining
The left lung was subjected to hematoxylin and eosin staining before observation. Compared with the saline-administered control mice (Figure 3a), noticeable infiltration of inflammatory cells and exudate was observed in ALI-induced mice that were intratracheally administered with LPS (Figure 3b). However, the infiltration of cells and exudate caused by LPS administration was inhibited by DFS pre-treatment (Figure 3c, d).
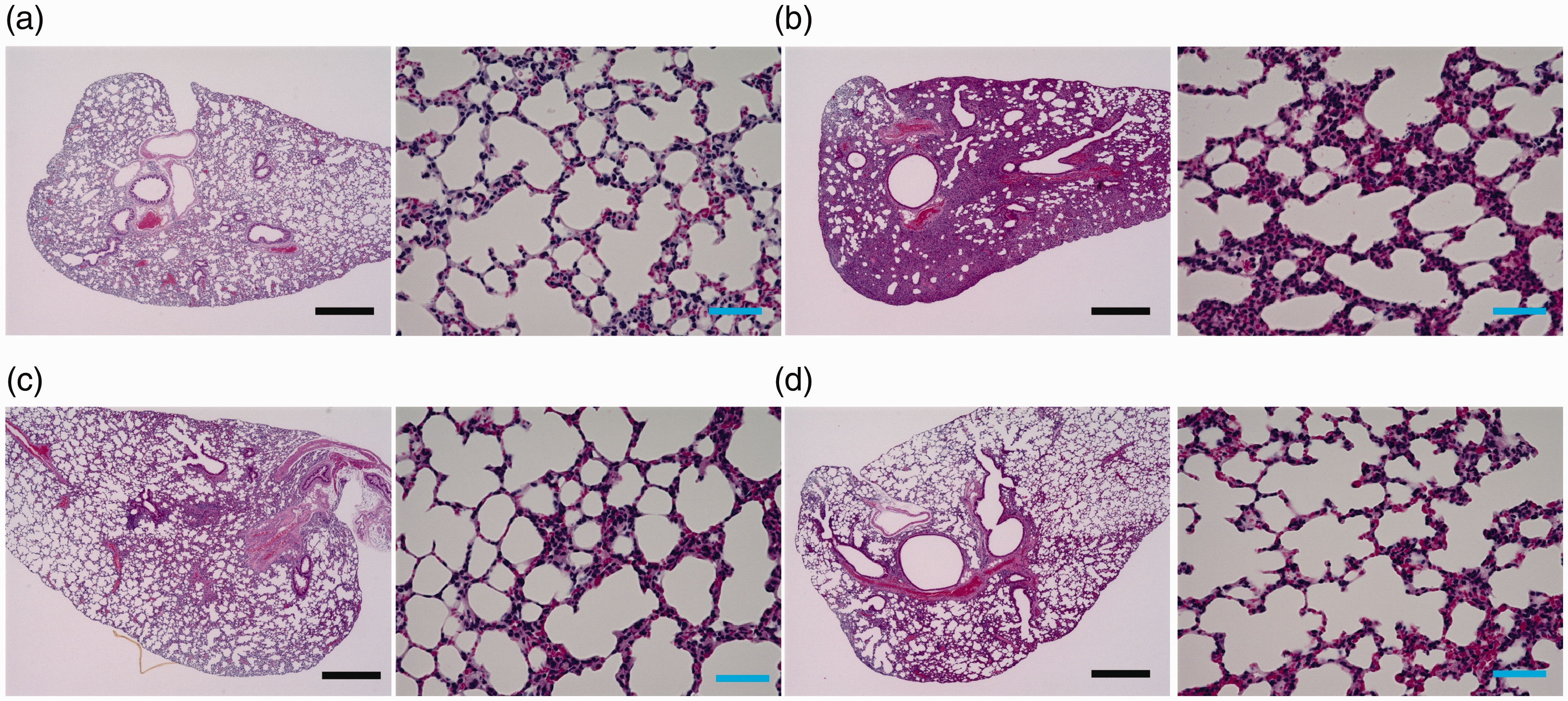
Hematoxylin and eosin staining of lung tissue from acute lung injury (ALI)-induced mice. Mice were pre-administered with deferasirox or PBS, and ALI was then induced by the intratracheal administration of lipopolysaccharides. (a) Control mice treated with PBS only. (b) ALI-induced mice treated with PBS only. (c) Control mice pre-administered with deferasirox. (d) ALI-induced mice pre-administered with deferasirox. Low- and high-magnification images are shown in the left and right panels, respectively. Black scale bars = 500 μm, blue scale bars = 50 μm.
Immunohistochemical staining
Immunohistochemical staining of the lung tissue revealed that the cells were positively stained for citrullinated histones in LPS-treated mice. However, citrullinated histones were not observed before LPS administration or in the mice pretreated with DFS (Figure 4).

Lung tissue from acute lung injury (ALI)-induced mice immunostained for citrullinated histones. Mice were pre-administered with deferasirox (DFS) or PBS, and ALI was then induced by the intratracheal administration of lipopolysaccharides (LPS). Scale bars = 25 μm.
Discussion
Recent reports and reviews have shown that ALI pathogenesis is associated with neutrophil activation, which involves enhanced ROS production, NET formation, elastase release, and neutrophil accumulation in the lungs.21,22
Attempts to alleviate ALI have been reported in animal studies. Glutamine inhibits ROS production by heme oxygenase-1 activation via the enhancement of glutathione synthesis, and reduced ROS generation ameliorates acute respiratory distress syndrome through the inhibition of NET formation and interferon-gamma production. 23 Because NETs directly damage the alveolar epithelium and endothelial cells via the production of toxic histones and myeloperoxidase, 24 Twaddell et al. showed that NETs are effective targets in ALI treatment. 25 In particular, ALI damage is reportedly alleviated by the depletion of neutrophils and/or platelets. 25
We previously reported that DFS inhibits ROS production, vacuolization, and NET formation
Although DFS did not suppress neutrophil vacuolization in our study, it did inhibit neutrophil invasion in the lung tissues. Thus far, whether DFS inhibits chemotaxis has not been demonstrated. However, it is speculated that fewer activated neutrophils and, as shown in a previous
Conclusions
The WBC count in the BALF was significantly decreased by DFS, and NET formation was partially suppressed by DFS. Although DFS did not suppress neutrophil vacuolization, it did inhibit neutrophil invasion in the lung tissue. Thus, our study confirmed that DFS has the potential to prevent diseases related to neutrophil activation including ALI.
