Abstract
Objective
To compare the efficacy of dexmedetomidine and magnesium sulfate as an adjuvant to local anesthetics in spinal anesthesia.
Methods
A search of PubMed, Medline, Embase, the Cochrane Library, and Google Scholar was performed. Randomized controlled trials comparing the efficacy of dexmedetomidine and magnesium sulfate as a local anesthetic adjuvant in spinal anesthesia were identified. The primary outcome was sensory block duration. The mean difference (MD) or odds ratio along with the 95% confidence interval (CI) was used to analyze the outcomes.
Results
Six studies involving 360 patients were included. Intrathecal dexmedetomidine was associated with a significantly longer sensory block duration (MD = −73.62; 95% CI = −101.09 to −46.15), faster onsets of sensory blockade and motor blockade, and a longer motor block duration than intrathecal magnesium sulfate. There was no significant difference between the regarding the rates of hypotension, bradycardia, shivering, and postoperative nausea and vomiting between the groups.
Conclusions
Dexmedetomidine is superior to magnesium sulfate as an adjuvant to local anesthetics in spinal anesthesia because of its more rapid onset and longer duration of spinal block without significant adverse effects.
Keywords
Introduction
Spinal anesthesia is a common and reliable anesthetic technique, but it has the disadvantage of a limited duration of action. Therefore, appropriate adjuvants are added to local anesthetics during spinal anesthesia to enhance the blockage quality and extend the block duration. 1 , 2
N-methyl-
Dexmedetomidine can act as an α2-adrenoreceptor agonist in the peripheral and CNS. This analgesic effect of intrathecal dexmedetomidine occurs through inhibition of the release of C-fiber transmitters and hyperpolarization of the postsynaptic dorsal horn neurons, which can explain the prolonged duration of spinal block when dexmedetomidine was added to intrathecal anesthetics. 7 , 8
Several randomized controlled trials (RCTs) have compared the analgesic effect of intrathecal dexmedetomidine and intrathecal magnesium sulfate in recent years.9–11 We decided to perform a meta-analysis to formally compare the efficacy of these agents as local anesthetic adjuvants in spinal anesthesia using a large sample size.
Methods
This study was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines, 12 and we did not register our study with PROSPERO. A literature search of PubMed, Medline, Embase, the Cochrane Library, and Google Scholar was performed from inception up to December 2019 using the following terms: intrathecal, anesthesia, spinal injections, magnesium, magnesium sulfate, dexmedetomidine, Dex, clinical trial, and randomized controlled trial. There were no language restrictions.
Inclusion criteria
The inclusion criteria were as follows: 1) adult patients who underwent surgery under spinal anesthesia; 2) dexmedetomidine and magnesium were compared as adjuvants to local anesthetics; and 3) the study was an RCT.
Exclusion criteria
Animal studies, conference reports, correspondences, or editorials were excluded.
Data extraction
Two researchers chose eligible clinical trials independently. The titles and abstracts of the initially selected articles were examined. The citations of these articles were also obtained and examined to identify eligible studies. Thereafter, the references of the identified papers were searched manually to find additional eligible papers.
Two authors independently analyzed the quality of the eligible RCTs using the Jadad score (a minimum of 3 points was required) and the Cochrane Collaboration guidelines. 13 , 14 Any disagreement was resolved by a third researcher.
Two authors independently extracted relevant data from the included RCTs. Any disagreement was resolved by a third author. If the included RCTs contained more than two groups, data were extracted only for the intrathecal dexmedetomidine and intrathecal magnesium groups. The primary endpoint was sensory block duration in the intrathecal dexmedetomidine and intrathecal magnesium groups.
Statistical analysis
Statistical analysis was conducted using Review Manager 5.3 (Nordic Cochrane Centre, Copenhagen, Denmark). The mean difference (MD) or odds ratio along with the 95% confidence interval (CI) was used to analyze the outcomes. Leave-one-out sensitivity analysis was performed by iteratively removing one study at a time if a high level of heterogeneity was detected (
Results
The search flowchart was presented in Figure 1. Two hundred fifty-four records were initially identified in the literature search. Of these, 242 studies were excluded after screening the abstracts. The full texts of 12 articles were found and examined, after which six more articles were excluded. Overall, six RCTs involving a total of 360 patients were identified for the final analysis.9–11,15–17 Five studies included three study groups featuring comparisons among dexmedetomidine, magnesium, and saline. 9 , 10 ,15–17 The last study only compared dexmedetomidine and magnesium. 11 The characteristics of the eligible RCTs are displayed in Table 1. All six articles had intermediate to high Jadad scores (Table 1). The risk-of-bias plot was created using Review Manager 5.3 (Figure 2).

Flow chart for selection of the included studies.
Characteristics of the included studies.

Dex, dexmedetomidine; M, magnesium; S, saline; RCT, randomized controlled trial; ASA, American Society of Anesthesiologists. ①: Onset of sensory blockade; ②: Sensory block duration; ③: Onset of motor blockade; ④: Motor block duration; ⑤: Postoperative pain intensity; ⑥: Sedation score; ⑦: Nausea and vomiting; ⑧: Shivering; ⑨: Hypotension and bradycardia; ⑩: Postoperative rescue analgesics.

Risk of bias summary: Authors’ judgment about each risk of bias item for each included study. Green, red, and yellow circles indicate low, high, and unclear risks of bias, respectively. Note: There were no high risks of bias found in these studies.
Sensory block duration
Five studies9–11,
15
,
16
reported the sensory block duration after intrathecal injection. Patients in the intrathecal dexmedetomidine group had a longer sensory block duration (MD = −73.62; 95% CI = −101.09 to −46.15,
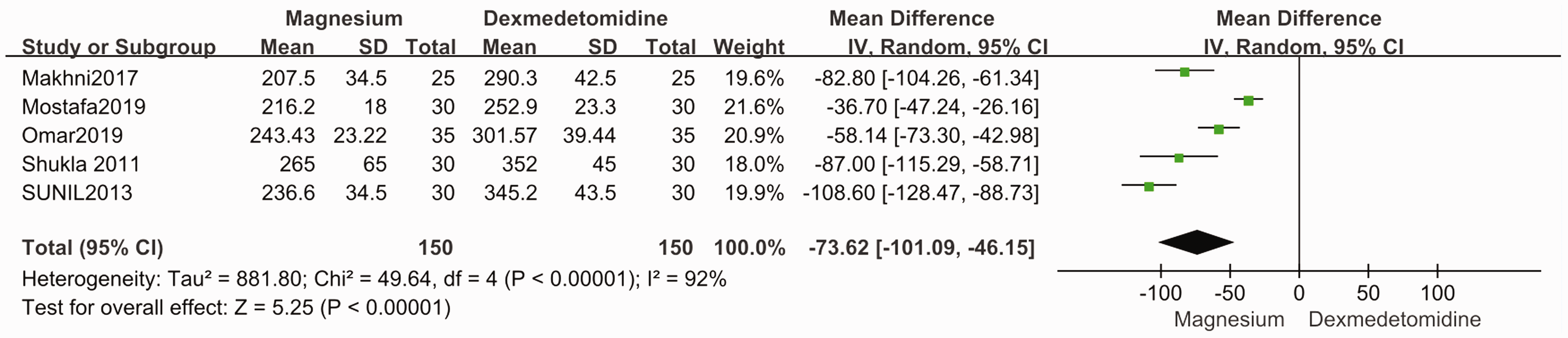
Forest plot of the sensory block duration in minutes. SD, standard deviation; CI, confidence interval; IV, inverse variance.
Sensitivity analysis was conducted to assess the reliability of the result by removing each RCT individually, and the results did not change (Table 2).
Sensitivity analysis of the primary outcome: Sensory block duration.

The results are presented after the indicated study was excluded in the leave-one-out sensitivity analysis. MD, mean difference; CI, confidence interval.
Onset of sensory blockade
All six included studies compared the onset of sensory blockade.9–11,15–17 Patients receiving intrathecal dexmedetomidine had a significantly faster onset of sensory blockade (MD = 2.77; 95% CI = 1.77–3.77,

Forest plot for the onset of sensory block in minutes. SD, standard deviation; CI, confidence interval; IV, inverse variance.
Onset and duration of motor blockade
The onset of motor blockade were assessed in five studies (300 patients),
9
,
11
,15–17 and the motor block duration was reported in four studies involving 240 patients.
9
,
11
,
15
,
16
Patients receiving intrathecal dexmedetomidine had a significantly faster onset of motor blockade (MD = 3.07; 95% CI = 2.02–4.11,

Forest plot for the onset of motor block in minutes. SD, standard deviation; CI, confidence interval; IV, inverse variance.

Forest plot for the motor block duration in minutes. SD, standard deviation; CI, confidence interval; IV, inverse variance.
Side effects
Two studies assessed the rates of hypotension, bradycardia, shivering, and postoperative nausea and vomiting. 9 , 15 No significant differences in the rates of any of these adverse events were noted between the groups (Figure 7).

Forest plot for the incidence of side effects. CI, confidence interval; M-H, Mantel–Haenszel.
Discussion
This study demonstrated that intrathecal dexmedetomidine is associated with longer durations of sensory and motor block and shorter onsets of sensory and motor block than intrathecal magnesium sulfate without an increased risk of adverse effects. Therefore, dexmedetomidine is superior to magnesium sulfate as a local anesthetic adjuvant for spinal anesthesia.
Dexmedetomidine is a highly selective α2-adrenoreceptor agonist. The analgesic effect of intrathecal dexmedetomidine occurs through the inhibition of C-fiber transmitter release and hyperpolarization of postsynaptic dorsal horn neurons. 18 The mechanism by which dexmedetomidine prolongs spinal block as a local anesthetic adjuvant is unclear. The synergistic effect between the local anesthetic and α2-adrenoreceptor agonist may contribute to this phenomenon. Local anesthetics and α2-adrenoreceptor agonists have different mechanisms of analgesia. Local anesthetics block sodium channels, whereas α2-adrenoreceptor agonists bind to pre-synaptic C fibers and postsynaptic dorsal horn neurons to produce analgesic effects.
Magnesium can inhibit calcium influx into cells and antagonize NMDA receptors, which may determine the duration of acute pain. 4 , 5 Kroin et al. 19 reported that intrathecal magnesium enhanced the analgesic effect of opioids for acute pain in rat models.
The results of this meta-analysis did not reveal statistically significant differences between the rates of hypotension, bradycardia, shivering, and postoperative nausea and vomiting between the dexmedetomidine and magnesium groups.
There was a high level of heterogeneity for some outcomes. Therefore, more high-quality RCTs with large sample sizes are needed. However, the results of this meta-analysis were not changed in the leave-one-out sensitivity analysis, illustrating that the meta-analysis results were not driven by any single study.
Conclusion
Dexmedetomidine is superior to magnesium sulfate as a local anesthetic adjuvant in spinal anesthesia because of its more rapid onset and longer duration of spinal block without significant adverse effects in patients.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
