Abstract
Objective
This study investigated the correlation between the interferon (IFN)-γ +874T/A polymorphism and hepatitis B virus (HBV) susceptibility using meta-analysis.
Methods
PubMed, EMBASE, Web of Science, CNKI, and China Wanfang databases were searched for case–control studies investigating the IFN-γ +874T/A polymorphism and HBV susceptibility from the time of database establishment to April 2020. Stata 15.0 software was used, and the subgroups of ethnicity and Hardy–Weinberg equilibrium were analyzed.
Results
Thirteen articles were included in this study. Significant differences were seen in the allelic model, dominant model, homozygous model, and heterozygous model, but heterogeneity was high. Analysis of the East Asian population revealed combined odds ratios of the allelic model (T vs. A), dominant model (TT + TA vs. AA), homozygous model (TT vs. AA), and heterozygous model (TA vs. AA) of 0.61, 0.56, 0.50, and 0.59, respectively. The difference was significant and the heterogeneity low. The recessive model showed no significance in the overall comparison, or in East Asian and Caucasian populations.
Conclusions
The IFN-γ +874T/A polymorphism is associated with the risk of HBV, especially in the East Asian population. Individuals with the T allele and TT and TA genotypes have a reduced risk of HBV infection.
Introduction
Global public health currently has many serious problems, including hepatitis B virus (HBV) infections, which are a longstanding issue. HBV infections occur worldwide, although their severity and the likelihood of epidemics vary among regions.1,2 Around 2 billion people have been infected with HBV worldwide, with about 240 million still showing chronic infection. 3 HBV infection mainly causes inflammatory lesions of the liver, which can lead to pathological damage such as liver cirrhosis and hepatocellular carcinoma. Short-term infection can cause self-limited, acute and chronic viral hepatitis, while long-term infection leads to a variety of end-stage liver diseases such as cirrhosis and liver cancer, 4 which place a huge burden on society. 5 During HBV infection, the viral envelope recognizes and binds with specific receptors on the surface of hepatocytes and then enters the cells through endocytosis. The final outcome of HBV infections is affected by viral factors (such as the genotype and copy number), host factors (like age, sex, immune level, and genetic susceptibility), and environmental factors (such as the medical and health level of the exposed population, and vaccination status).6–9
Recent studies have investigated the correlation between cytokine polymorphisms and susceptibility to HBV. The interferon (IFN)-γ gene has been shown to have six polymorphic sites located in the promoter, intron 1, intron 3, and the 3′ untranslated region. IFN-γ expression is highest when there are 12 CA repeat sequences in the promoter region of the first intron compared with other numbers of CA repeats under the same conditions. 10 Additionally, the 5′ IFN-γ A/T single nucleotide polymorphism (SNP) located +874 of the CA sequence repeat is also associated with IFN-γ expression and the number of CA repeats. TT is a high-expression genotype, AT is a medium-expression genotype, and AA a low-expression genotype. 10 The +874 site is located exactly at the binding site of transcription factor nuclear factor (NF)κB and the SNP directly affects its binding ability, thus influencing transcription and IFN-γ expression. 11 HBV infection levels were found to decrease with increasing IFN-γ expression in chimpanzees, 12 so the susceptibility to HBV is thought to be associated with IFN-γ gene SNPs. Yu et al. 13 and Korachi et al. 14 found that the T allele at IFN-γ +874 was a protective factor against HBV infection, Arababadi et al. 15 reported that the T allele was not associated with HBV susceptibility, while Ghasemian et al. 16 showed that it was a risk factor for HBV infection. Because the conclusions of these previous studies are inconsistent, we herein used a meta-analysis to investigate whether the IFN-γ +874 SNP is associated with susceptibility to HBV infection.
Methods
Study selection
We searched PubMed, Excerpta Medica Database, Web of Science, China national knowledge infrastructure, and China Wanfang databases for case–control studies on the correlation between the IFN-γ +874T/A SNP and HBV infection from the start of each database establishment to April 2020. Search terms were as follows: (“Hepatitis B” OR “chronic hepatitis B” OR “hepatitis B virus” OR “HBV”) AND (“Interferon-γ” OR “Interferon-gamma”) AND (“polymorphism” OR “single nucleotide polymorphism” OR “SNP”). Only published full-text studies were included. Publication languages were limited to Chinese and English. If multiple publications on the same subject were identified, the most complete and up-to-date results were used. A reference list of selected articles (including comments) was also reviewed to identify other relevant publications.
Inclusion and exclusion criteria
Inclusion criteria were: (1) A case–control study on the correlation between the IFN-γ +874T/A SNP (rs2430561) and HBV infection; (2) a study of humans; and (3) original data available for extraction or to calculate the IFN-γ +874T/A SNP genotype frequency. Exclusion criteria were: (1) Letters, abstracts, or minutes of scientific meetings addressed to editors; (2) exclusion of unpublished data resources; and (3) a Newcastle–Ottawa Scale (NOS) score <6.
Data extraction
Two researchers evaluated each study to determine whether it should be included in the meta-analysis, and independently accessed the methods. The following information was extracted from each study: first author, year of publication, country, ethnic identity of patient population, number of cases and controls, source of controls, demographic variables, genotypic detection methods, SNP information, genotype frequency, and allele frequency.
Document quality evaluation
According to the NOS, 17 the full text was carefully read and evaluated. Studies scoring below 6 were deemed of low quality, so only studies scoring 6 and above were included. In accordance with unified quality standards, the assessment was conducted independently by two assessors who extracted and cross-checked file data. In the case of discrepancy, issues were resolved through discussion or with the assistance of a third party.
Statistical analysis
Odds ratios (ORs) and 95% confidence intervals (CIs) were used to evaluate the association between the INF-γ +874T/A SNP and HBV infection. Data were analyzed using Stata 15.0 statistical software (Stata Corp., College Station, TX, USA). The Q test was used to verify the heterogeneity of included studies. When I2 ≥ 50% and P ≤ 0.05, it was considered that there was heterogeneity among studies and the random-effects model was used. When I2 < 50% and P > 0.05, heterogeneity between studies was considered to be low and the fixed-effects model was used for data integration. The funnel plot was used to evaluate publication bias, with asymmetry representing possible publication bias. Egger’s test was also used to test publication bias. Subgroup analysis was carried out to determine whether ethnicity and the control group met the Hardy–Weinberg equilibrium (HWE) standard. Sensitivity analysis was conducted to determine the stability of the findings.
Results
Literature retrieval
Thirteen articles met the inclusion criteria.13–16,18–26 The literature selection process is shown in Figure 1. The characteristics of the included studies, genotype distribution frequency, and literature quality scores are shown in Table 1.

Flow diagram of the study selection process.
Characteristics of included studies.
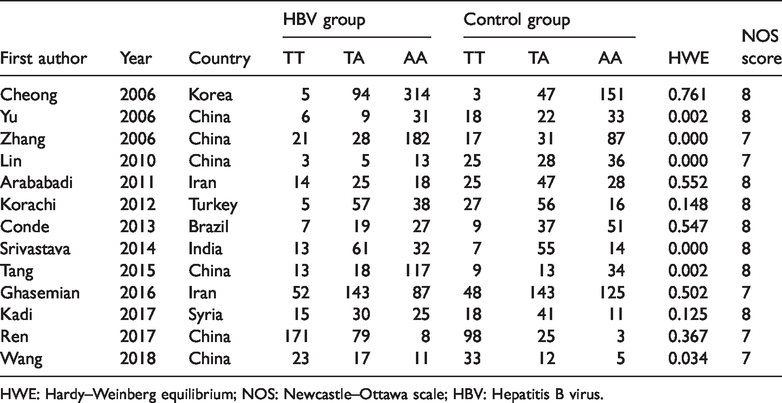
HWE: Hardy–Weinberg equilibrium; NOS: Newcastle–Ottawa scale; HBV: Hepatitis B virus.
Meta-analysis
Allele comparison
The main results of the meta-analysis are shown in Table 2 and Figure 2. Compared with the A allele, the T allele had an I2 = 75.4% (P < 0.05), which showed that heterogeneity among studies was significant so the random-effects model was used. The combined OR was 0.67 (95% CI: 0.53–0.86, P < 0.01), which was statistically significant. According to ethnicity subgroup analysis, when the T allele was compared with the A allele, the combined OR was 0.65 (95% CI: 0.36–1.16) in the Caucasian group, and the difference was not significant; in the East Asian population, the combined OR was 0.61 (95% CI: 0.49–0.76, P < 0.05), the difference was significant and the heterogeneity was low (I2 = 40.2%). Subgroup analysis of the control group meeting HWE showed that heterogeneity did not decrease. The forest plot is shown in Figure 2a, and suggests that the IFN-γ +874T/A SNP was associated with susceptibility to HPV infection, especially in the East Asian population, with the T allele acting as a protective factor against infection. The funnel plot is generally symmetrical (Figure 3a), and Egger’s test showed that P < 0.05, indicating partial publication bias.
Meta-analysis findings for the IFN-γ +874T/A SNP and HBV infection.

OR: odds ratio; CI: confidence interval; FEM: fixed-effects model; REM: random-effects model; HWE: Hardy–Weinberg equilibrium; NA: not applicable.

Forest plot for the association between the IFN-γ +874T/A SNP and HBV infection. (a) Allelic model; (b) Dominant model; (c) Recessive model; (d) Homozygous model; (e) Heterozygote model.
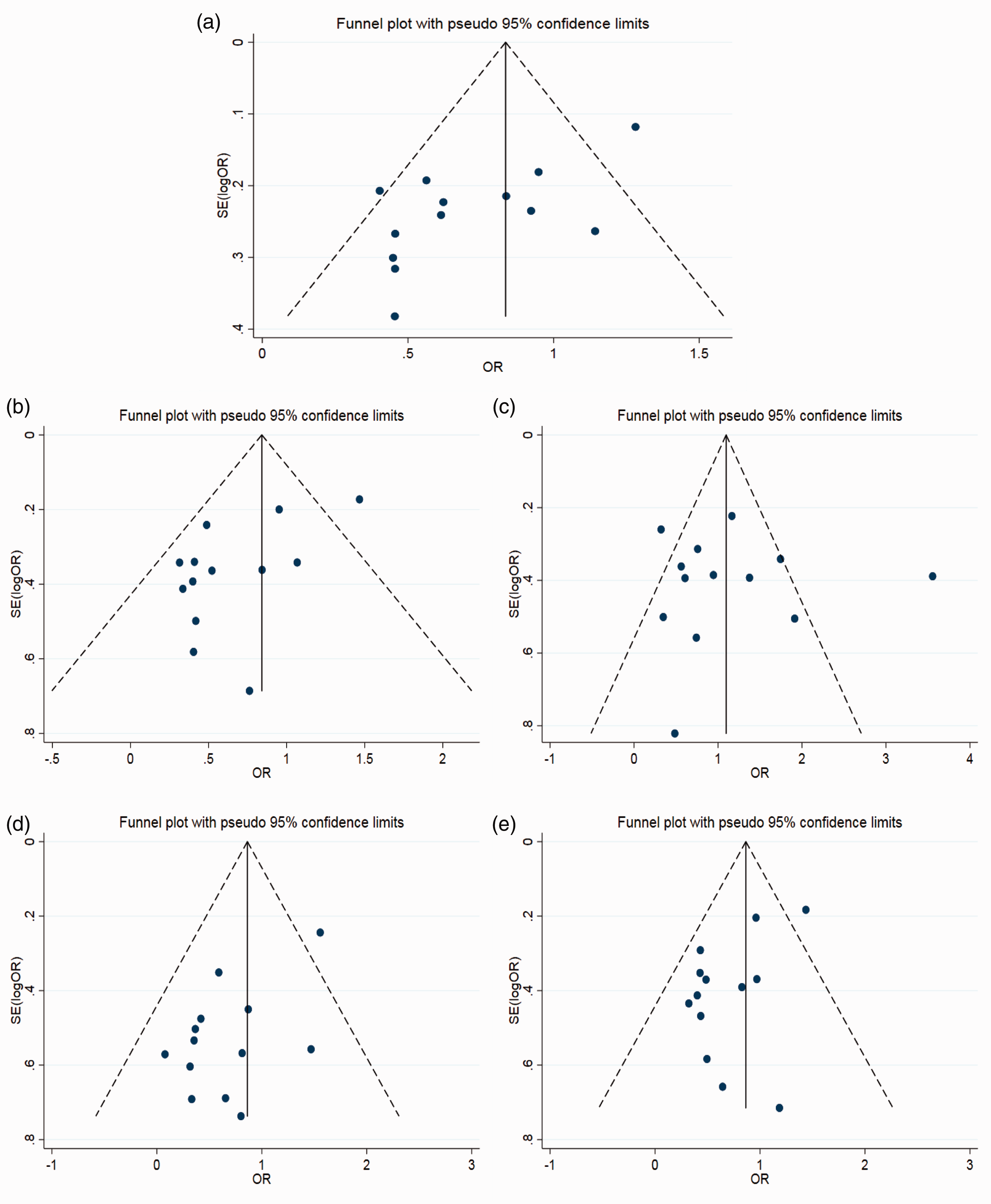
Funnel plot for the assessment of publication bias. (a) Allelic model; (b) Dominant model; c: Recessive model; (d) Homozygous model; (e) Heterozygote model.
Dominant model
Compared with genotype AA, genotypes TT + TA had an I2 = 70.2% (P < 0.05), indicating heterogeneity among studies so the random-effects model was used. The combined OR was 0.60 (95% CI: 0.43–0.84, P < 0.05), which was statistically significant. According to ethnicity subgroup analysis, the combined OR was 0.49 (95% CI: 0.22–1.10) in the Caucasian group and the difference was not significant; in the East Asian population, the combined OR was 0.56 (95% CI: 0.42–0.74, P <0.05), the difference was significant, and the heterogeneity significantly decreased (I2 = 27.5%). The forest plot is shown in Figure 2b, and subgroup analysis of the control group meeting HWE showed that the heterogeneity did not decrease. The funnel plot is generally symmetrical (Figure 3b). Egger’s test showed that P < 0.05, indicating partial publication bias.
Recessive model
Compared with genotype AA, genotypes TT + TA had an I2 = 70.2% (P < 0.05), indicating heterogeneity among studies so the random-effects model was used. The combined OR was 0.90 (95% CI: 0.16–1.33), which was not significant. According to ethnicity subgroup analysis, the combined OR was 0.99 (95% CI: 0.61–1.60) in the Caucasian group, and the difference was not significant; in the East Asian population, the combined OR was 0.73 (95% CI: 0.39–1.35), and the difference was also not significant. The forest plot is shown in Figure 2c, and subgroup analysis of the control group meeting HWE showed that the heterogeneity did not decrease. The funnel plot is generally symmetrical (Figure 3c). Egger’s test showed that P > 0.05, indicating that publication bias was well controlled.
Homozygous model
Compared with genotype AA, genotypes TT + TA had an I2 = 65.9% (P < 0.05), indicating heterogeneity among studies so the random-effects model was used. The combined OR was 0.55 (95% CI: 0.34–0.87, P < 0.05), which was statistically significant. According to ethnicity subgroup analysis, the combined OR was 0.35 (95% CI: 0.07–1.73) in the Caucasian group, and the difference was not significant; in the East Asian population, the combined OR was 0.50 (95% CI: 0.35–0.73, P < 0.05), the difference was significant, and there was evidence of heterogeneity (I2 = 0%). The forest plot is shown in Figure 2d, and subgroup analysis of the control group meeting HWE showed that the heterogeneity did not decrease. The funnel plot is generally symmetrical (Figure 3d). Egger’s test showed that P < 0.05, indicating partial publication bias.
Heterozygote model
Compared with genotype AA, genotype TA had an I2 = 60.6% (P < 0.05), indicating heterogeneity among studies so the random-effects model was used. The combined OR was 0.64 (95% CI: 0.47–0.88, P < 0.05), which was statistically significant. According to ethnicity subgroup analysis, the combined OR was 0.52 (95% CI: 0.28–0.99, P < 0.05) in the Caucasian group, and the difference was significant; in the East Asian population, the combined OR was 0.59 (95% CI: 0.43–0.81, P < 0.05), the difference was significant, and heterogeneity decreased significantly (I2 = 24.1%). The forest plot is shown in Figure 2e. The funnel plot is generally symmetrical (Figure 3e). Egger’s test showed that P < 0.05, indicating partial publication bias.
Sensitivity analysis
Because of the large heterogeneity of the overall comparison, few studies were included on West Asian and Caucasian populations, while the East Asian population had low heterogeneity. Therefore, our study only focuses on the sensitivity analysis of genetic models of the East Asian population which are shown in Figure 4. Each study was excluded one at a time, and the meta-analysis was applied again to determine the stability of the results. There was no significant change in the combined effect of each genetic model, indicating that HWE was not the main source of heterogeneity and that the conclusion between the IFN-γ +874T/A SNP and patients with HBV infection in East Asia is stable.
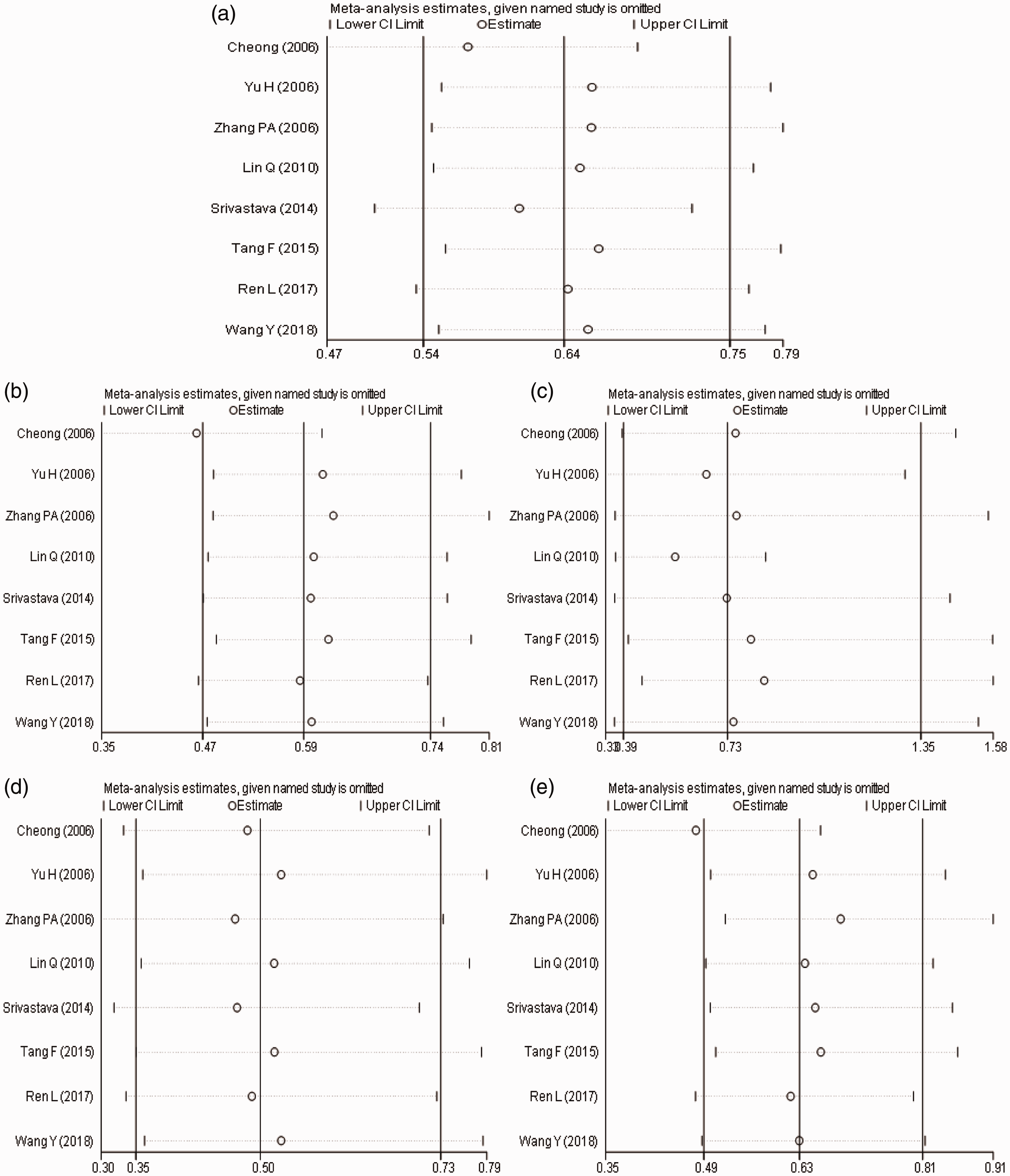
Sensitivity analysis of the East Asian population. (a) Allelic model; (b) Dominant model; (c) Recessive model; (d) Homozygous model; (e) Heterozygote model.
Discussion
The main purpose of this meta-analysis was to investigate the correlation between the IFN-γ +874T/A SNP (rs2430561) and the risk of HBV infection. We found that rs2430561 was closely associated with the risk of HBV infection, suggesting that it may be related to increased susceptibility to HBV infection.
IFN-γ is mainly secreted by T lymphocytes and natural killer (NK) cells after activation, and can also be produced by T helper type 1 cells, macrophages, and hepatocytes. It has multiple functions and adaptively regulates antiviral responses. 27 Ma et al. 28 studied changes in CD8+ memory T cell subsets in different immune states during the natural clearance of chronic HBV infections, and found that IFN-γ expression of the HBsAg seroconversion group was significantly higher than in the HBeAg seroconversion group. Gao et al. 29 and others found that IFN-γ gene SNPs may also interact with polymorphisms in interleukin (IL)-4, IL-10, and other genes to resist HBV infection. IFN-γ may also inhibit HBV-induced NF-κB activation by inducing the nuclear localization of kinase by NF-κB. 30 Tjwa et al. 31 reported that the functional interaction between IFN-γ-activated monocytes and NK cells in patients with chronic hepatitis B infection was weakened by the impairment of monocyte dendritic cell function, and could be partially restored by antiviral therapy. Enhancing this interaction would increase innate and adaptive T cell responses, which may be an important step in achieving effective antiviral immunity.
Our meta-analysis identified a strong correlation between IFN-γ +874T/A and susceptibility to HBV infection, with significance detected in the allelic model, dominant model, homozygous model, and heterozygous model, but not in the recessive model. However, heterogeneity was large in these models, the funnel chart was generally symmetrical, and partial publication bias was evident. Therefore, we conducted a subgroup analysis of ethnicity and HWE to identify the source of heterogeneity. In the East Asian population, heterogeneity was small and the difference was significant in the allelic model, dominant model, homozygous model, and heterozygous model; however, heterogeneity was large and the difference was not significant in the recessive model. Further sensitivity analysis of the genetic models for the East Asian population confirmed these findings. In the Caucasian population, only the heterozygous model (TA vs. AA) showed significance. However, because only three studies of Caucasian populations were included in the meta-analysis and heterogeneity was large, these conclusions should be interpreted with caution. Additionally, because only two studies have investigated the correlation between IFN-γ +874T/A and susceptibility to HBV infection in the West Asian population so the sample size is too small to reach a conclusion, we have not performed any more in-depth analysis of these. Excluding the control group that did not meet HWE, the repeat meta-analysis showed that the heterogeneity of each genetic model increased, so HWE was not the main source of heterogeneity. Our findings are consistent with the meta-analysis conducted by Sun et al. 32 in Asian populations which showed that the IFN-γ +874T/A T allele was a protective factor against chronic hepatitis B infection, with significant differences seen in dominant, homozygous, and heterozygous genetic models.
The present study has a number of limitations. First, except for the recessive genetic model, partial publication bias was evident in the comparison of other genetic models. This may reflect the exclusion of unpublished data and studies published in languages other than English and Chinese. Second, almost all of the included studies were conducted in Asia, mostly East Asia, with only two in West Asia and only three in Caucasians. Therefore, any conclusions for West Asian and Caucasian populations should be interpreted with caution. Third, control genotypes in some included studies did not meet the requirements of HWE. Finally, we did not study gene–gene or gene–environment interactions that may have affected the relationship between the IFN-γ SNP and the risk of HPV infection. Nevertheless, our findings for the East Asian population are relatively stable and reveal a strong correlation low heterogeneity.
Our conclusions clearly show that the IFN-γ +874T/A SNP was associated with the risk of HBV infection, especially in East Asian populations. Although no correlation was found in the recessive model, correlations were detected in other genetic models. The T allele and genotypes TT, TT + TA, and TA could reduce the risk of HBV infection in East Asian populations. These findings should be confirmed in further studies.
Footnotes
Availability of data
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
