Abstract
Objective
We evaluated the effect of chronic use of statins based on C-reactive protein (CRP) levels and hospital length of stay (LOS) in patients admitted with community-acquired pneumonia (CAP).
Methods
We conducted a retrospective study over 12 months at a teaching hospital in Lebanon comparing patients with CAP taking chronic statins with patients not taking statins. Included patients with CAP were older than age 18 years and had two CRP level measures during hospitalization. CURB-65 criteria were used to assess the severity of pneumonia. A decrease in CRP levels on days 1 and 3, LOS, and normalization of fever were used to assess the response to antibiotics.
Results
Sixty-one patients were taking statins and 90 patients were not taking statins. Patients on statins had significantly more comorbid conditions; both groups had comparable CURB-65 scores. In both groups, no statistically significant difference was seen for the decrease in CRP level on days 1 and 3 and LOS. No difference in days to normalization of fever was detected in either group.
Conclusion
No association was found between the chronic use of statins and CRP levels, LOS, or days to fever normalization in patients with CAP.
Keywords
Introduction
Community-acquired pneumonia (CAP) is a leading cause of morbidity and mortality worldwide. 1 CAP is considered the most common infection leading to hospitalization. 1 Since the introduction of antibiotics in the 1950s, the overall mortality owing to CAP has decreased substantially, but it has remained constant over the past 50 years. 2 Although an effective inflammatory and immune response is essential in eradicating microorganisms, an excessive increase in proinflammatory markers and immunological response, as well as endothelial dysfunction, are predictive factors of poor outcomes and treatment failure in patients diagnosed with CAP. 3 The systemic elevation of cytokine levels in specific circumstances may lead to acute lung injury, acute respiratory distress syndrome, or septic shock. Competitive inhibitors of 3-hydroxy-3-methylglutaryl coenzyme A reductase, or statins, are lipid-lowering agents widely prescribed for the prevention and treatment of cardiovascular disease. The pleiotropic effects of statins, which include anti-inflammatory, anti-oxidative, and immunomodulatory regulation effects, suggest the possibility of their use in infectious diseases. 4 These effects are attributed to decreased production of isoprenoid molecules owing to the inhibition of mevalonic acid synthesis. Consequently, intracellular signaling molecules such as Ras, Rap, Rho, and Rab cannot act as messengers, resulting in a moderated cellular inflammatory response. 4 The decrease in the production of inflammatory cytokines and C-reactive protein (CRP) levels, together with the effects on neutrophil activity and chemotaxis and improvement in vascular function, may explain the potential beneficial effect of statins in CAP.4–6 Statins can also decrease systemic levels of tissue necrosis factor (TNF) and interleukin (IL)-6, which may have additive beneficial anti-inflammatory effects. 7 In addition; the effects of statins are dose dependent, so the optimal intensity must be determined. Recently, several studies have evaluated the postulated favorable effect of statin use in CAP, albeit with conflicting results; therefore, further studies are needed to evaluate the use of statins in CAP.8–10
The objective of this study was to evaluate antibiotic response based on CRP levels, hospital length of stay (LOS), and days to normalization of fever in patients admitted with CAP, and based on chronic statin therapy among those patients receiving treatment for CAP without being on statin therapy.
Patients and methods
General study design
We conducted a retrospective observational study, including patients receiving chronic statins in comparison with those who were not on statins. This study was completed over 12 months (June 2016 through June 2017) at the Lebanese American University Medical Center–Rizk Hospital, a tertiary care teaching hospital in Beirut. A list of all patients with a diagnosis of CAP during 2014 and 2015 was retrieved from the medical records department. The patient list was cross-checked with the laboratory to identify patients with CAP who had a minimum of two CRP levels measured during hospitalization. The endpoints were CRP levels for patients with chronic use of statins and those who did not use statins. We evaluated LOS in addition to CRP levels.
Data collection process
Patients were included in the study if they: 1) were inpatients older than 18 years of age, admitted with a CAP diagnosis during 2014 and 2015, 2) had two CRP levels measured on different days during their hospitalization, the first one preferably on the day of admission and the second within 72 hours. Patients were excluded if they did not meet the inclusion criteria. Therefore, included patients were those who met the inclusion criteria whether they were admitted to a medical ward or intensive care unit.
We collected data of patient demographics, LOS, medical history, and comorbidities including history of drug resistance, surgical history, and current infection information including culture results, CURB-65 (confusion of new onset, blood urea nitrogen greater than 7 mmol/L or 19 mg/dL, respiratory rate of 30 breaths per minute or greater, systolic blood pressure less than 90 mmHg or diastolic blood pressure 60 mmHg or less, and age of 65 or older) score, antibiotics used including macrolides, chronic intake of statins, current medication intake, CRP level, erythrocyte sedimentation rate, white blood cell differentials, liver function tests when available, and temperature. Temperature normalization was recorded, defined as temperature ≤37.1°C. 11
Ethics approval
This study was approved by the institutional review board of the Lebanese American University. This was a retrospective study in which no patient informed consent was needed.
Sample size calculation and statistical analysis
For a two-tailed study, with alpha = 0.05 and a power of 80%, 116 patients (58 in each group) were needed to detect a difference in CRP. 12
An individual who was independently trained in the data collection tool completed data entry. Descriptive statistics were calculated for all study variables, including the mean and standard deviation for continuous measures, and frequency and percentage for categorical variables. The Cox regression method was used to calculate the days to fever normalization in both groups; CURB-65 and macrolides were taken as confounders. IBM SPSS version 22.0 was used for all statistical analysis (IBM Corp., Armonk, NY, USA).
Results
A total of 151 patients were included in the final analysis, 61 in the group taking statins and 90 in the group not taking statins. Mean age was 74 years in the statins group and 71.4 years in the non-statins group.
There were 61 (40%) female patients. Patients’ baseline characteristics with respect to age, weight, and height did not differ between groups. Comorbidities such as diabetes, dyslipidemia, hypertension, and renal insufficiency requiring dialysis were more prevalent in patients chronically taking statins than in those who were not taking statins (Table 1). However, patients in both groups had similar proportions of other comorbidities such as congestive heart failure, chronic obstructive pulmonary disease, asthma, or gastroesophageal reflux disease, in addition to similar pneumonia severity based on the CURB-65 criteria. It is worth highlighting that macrolide intake was significantly different between those who took statins and those who did not (p = 0.04) (Table 2). The mean hospital LOS was 8.82 ± 6.65 days for patients who were on statins and 8.44 ± 4.05 days for those who were not on statins. In terms of CRP levels, based on a Sig (two-tailed) independent sample test where an equal variance was assumed, there was no significant difference in CRP levels on either day 1 or day 3 in patients receiving or not receiving statins. On day 1, patients receiving statins had a mean CRP of 21.10 ± 29.98 mg/L in comparison with patients not receiving statins (mean 17.48 ± 13.40, 95% confidence interval (CI): −10.72 to 3.47). On day 3, the mean CRP level decreased in both groups in comparison with day 1; however, there was no significant difference between patients taking and those not taking statins (6.51 ± 5.85 mg/L versus 8.48 ± 15.93 mg/L, 95% CI: −2.24 to 6.18).
Patient characteristics and comorbidities for those taking statins and not taking statins.
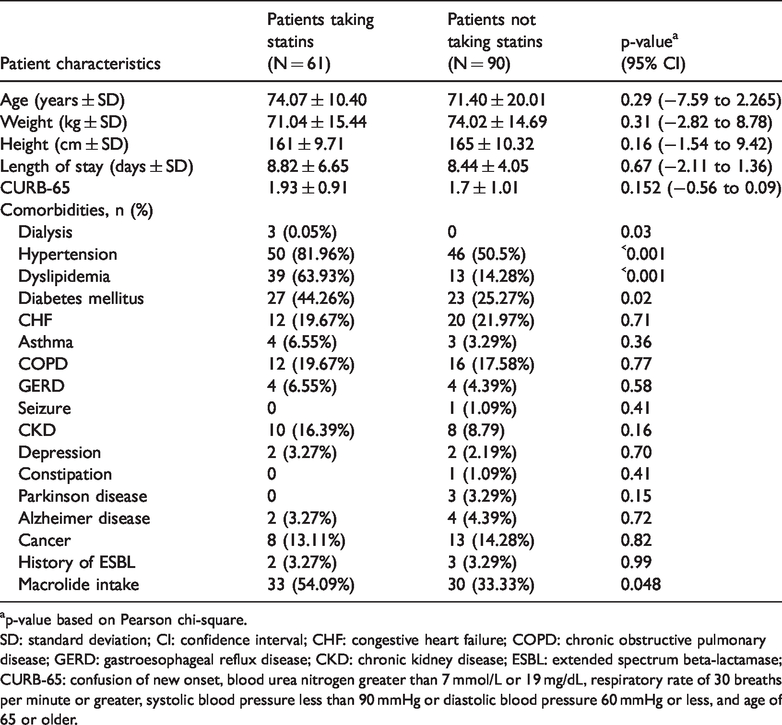
ap-value based on Pearson chi-square.
SD: standard deviation; CI: confidence interval; CHF: congestive heart failure; COPD: chronic obstructive pulmonary disease; GERD: gastroesophageal reflux disease; CKD: chronic kidney disease; ESBL: extended spectrum beta-lactamase; CURB-65: confusion of new onset, blood urea nitrogen greater than 7 mmol/L or 19 mg/dL, respiratory rate of 30 breaths per minute or greater, systolic blood pressure less than 90 mmHg or diastolic blood pressure 60 mmHg or less, and age of 65 or older.
CURB-65 criteria for patients taking statins and not taking statins.

ap-value based on Pearson chi-square.
RR: respiratory rate; SBP: systolic blood pressure; DBP: diastolic blood pressure, BUN: blood urea nitrogen; CURB-65: confusion of new onset, blood urea nitrogen greater than 7 mmol/L or 19 mg/dL, respiratory rate of 30 breaths per minute or greater, systolic blood pressure less than 90 mmHg or diastolic blood pressure 60 mmHg or less, and age of 65 or older.
CURB-65 and macrolides were used as confounders. There was no significant difference between the two groups; however, CURB-65 was significant (p = 0.008) (Table 3).
Days to normalization, Cox regression enter method.

CURB-65: confusion of new onset, blood urea nitrogen greater than 7 mmol/L or 19 mg/dL, respiratory rate of 30 breaths per minute or greater, systolic blood pressure less than 90 mmHg or diastolic blood pressure 60 mmHg or less, and age of 65 or older.
Discussion
In this retrospective study, we did not identify a positive impact on either inflammatory response measured using CRP or days to normalization of fever and LOS between patients taking and those not taking statins. These findings are controversial in the related published studies. The measured clinical outcomes have been either 30-day mortality, LOS, inflammatory mediators, intensive care unit admission owing to pneumonia, or a combination of outcomes. For instance, a nested study within EPIC, a larger multicenter, prospective surveillance study assessing incidence and etiology of CAP, evaluated the effect of statins on hospital LOS and in-hospital mortality among patients admitted for CAP. 10 Similar to our study, statins had to be continued throughout hospitalization for patients to be included. That study did not find statistical significance regarding LOS or mortality rates. 10 Two additional observational studies showed that statins did not reduce either the risk of CAP in immunocompetent older patients or the mortality rate or intensive care admissions owing to pneumonia. 13 , 14 Another retrospective cohort study showed that patients who were started on statins prior to admission for CAP had a substantial reduction in 30-day mortality, by 64%. 9 However, it is important to note that this study was conducted at a Veterans Affairs hospital with mostly male participants and missing data stated as a limitation. 9 Furthermore, a systematic review and meta-analysis concluded that statin use is associated with a favorable effect in both the prevention and treatment of CAP. 15 The risk of CAP development and mortality was significantly decreased. However, the meta-analysis had limitations related to the observational design of the included studies, their heterogeneity, and publication bias.
The effect of statins on clinical outcomes in patients with CAP was measured in a prospective observational study. 16 The investigators found a significant decrease in 30-day mortality and a decrease in the development of complicated pneumonia among patients receiving statins. 16 Moreover, the effect of simvastatin on inflammatory cytokines (TNF-α, IL-6, and IL-10) in CAP was studied in a randomized double-blind placebo-controlled trial. 12 Patients were randomly assigned to receive either simvastatin 20 mg or placebo in the first 24 hours of admission and continued for 4 days during hospitalization. No detection of a significant decrease in the level of inflammatory cytokines was noted, and there was no reduction in the time to clinical improvement among older patients hospitalized for CAP. 12 In addition, atorvastatin was prospectively compared with placebo among patients in intensive care with pneumonia, among other infections treated. The researchers looked at the concentrations of inflammatory markers, specifically IL-6 and CRP level. No significant difference between atorvastatin and placebo was detected. 17 Furthermore, a recent nested-control study using a large research database in Taiwan looked at patients admitted with myocardial infarction in whom statin therapy was initiated. The results showed that patients in the statins group had a significantly decreased risk of pneumonia; patients who were not receiving prior statin therapy had the greatest benefit. 18
It is important to note that no standard statin therapy was used in the studies mentioned above to compare the anti-inflammatory effect of statins on the endpoints, including inflammatory cytokines. In our study, the dose of statins differed among patients. Statins used by patients were pravastatin, simvastatin, atorvastatin, rosuvastatin, and pitavastatin. Most patients taking atorvastatin received a dose of 10 to 20 mg whereas those on pravastatin received a dose of 20 to 40 mg and those on rosuvastatin received a dose of 5 to 10 mg; all doses were in the clinically recommended range. A randomized double-blind placebo-controlled trial assessing the effects of statin therapy on inflammatory cytokines in patients with bacterial infection showed a reduction in the inflammatory cytokine levels. The statin regimen was 40 mg simvastatin followed by 20 mg. 19 Further, as previously mentioned, a considerable number of patients in both groups were taking macrolides. Macrolides exert anti-inflammatory activity inhibiting the production of inflammatory cytokines such as IL-8, altering leukocyte function, apoptosis and mucus production. 20 Although there was a statistically significant difference between the number of patients who were taking macrolides in the two groups, this did not impact CRP normalization, which could be owing to the study being underpowered.
The limitations of this study are as follows. First, this was a retrospective observational study in which randomization was not possible; the investigators used inflammatory indicators that were previously measured, such as CRP. Second, CRP levels were not taken at standardized times, as in a prospective trial. Third, the dose of statins was not standardized; patients were on different medically appropriate doses. Such variation in doses does not allow for assessment of the anti-inflammatory role of statins based on the agent and strength used. However, this study was not powered to detect the anti-inflammatory effect of different statin doses. Finally, this study was underpowered to assess the clinical endpoints.
Conclusion
We did not find an association between the chronic use of statins as anti-inflammatory agents and CRP levels, days to normalization of fever, and hospital LOS in patients admitted with CAP.
