Abstract
Objective
This study was performed to examine the clinical outcomes of epidural and intradural decompression for degenerative cervical myelopathy.
Methods
The data for 13 patients who underwent epidural and intradural decompression for treatment of degenerative cervical myelopathy (study group) and 20 patients who underwent only cervical laminoplasty, fusion, and epidural decompression (historical control group) were retrospectively reviewed. The preoperative and postoperative neurological status was evaluated using the Japanese Orthopaedic Association (JOA) score.
Results
All patients’ neurological symptoms were significantly improved at the final follow-up. In the study group, the patients’ mean preoperative JOA score was 8.07 ± 1.80, and the final score improved by 70.88% ± 21.18%. The blood loss and operation time were significantly greater in the study group than control group. The recovery time was shorter in the study group than control group. The improvement rate was not significantly different between the two groups.
Conclusions
A pia mater incision with separation of the arachnoid adhesion can significantly improve the cerebrospinal fluid flow and spinal blood flow in degenerative cervical myelopathy. Arachnoid adhesion can lead to intradural spinal scar compression. The surgical intervention described herein can achieve satisfactory neurological outcomes and shorten the recovery time.
Keywords
Introduction
Degenerative cervical myelopathy (DCM) is a common degenerative spinal cord disease and the main cause of neurological deficits and progressive spinal cord dysfunction in adults, especially those of advanced age.1,2 The most common etiology of DCM is spinal stenosis caused by cervical spondylotic myelopathy, ossification of the posterior longitudinal ligament, ossification of the ligamentum flavum, or degenerative disc disease. 2 Neurological deficits can be resolved in most patients by surgical decompression,1,3,4 including anterior, posterior, and combined anterior-posterior approaches. 1 The mainstays of posterior surgical procedures are cervical laminoplasty and cervical laminectomy with fusion, while the most common anterior surgical procedures are discectomy and corpectomy with fusion. 1 All of these surgical interventions are effective in treating DCM.1,3,4
Many reports have described satisfactory clinical results of surgical intervention for DCM in recent years.1,3,4 Improved cerebrospinal fluid circulation and spinal cord blood flow are critical for postoperative neurological recovery. However, intradural scar compression can reduce the spinal cord blood supply and block cerebrospinal fluid circulation. For example, after routine extradural decompression, some patients exhibit obviously weakened dural nonpulsatile or pulsatile flow because of intradural scar and arachnoid adhesion. Therefore, intradural decompression is needed to improve the cerebrospinal fluid circulation and spinal cord blood flow. Zhu et al. 5 reported the outcomes of 30 patients who underwent neurosurgical management, including separation of the arachnoid adhesion to restore cerebrospinal fluid flow and debridement of the spinal cord necrotic tissue with concomitant intramedullary decompression. All patients showed significant neurological recovery at the 3-month follow-up. 5 Some animal experiments have demonstrated that dural decompression can improve cerebrospinal fluid pressure and spinal cord blood flow and further accelerate neurological recovery after spinal cord injuries.6,7 However, no reports have described the clinical outcomes of intradural decompression followed by epidural decompression using posterior surgical procedures in patients with DCM. Hence, we herein report the clinical outcomes of epidural and intradural decompression for treatment of DCM.
Materials and methods
Patients
All patients provided consent. This study was approved by the Medical Ethics Committee at the First Affiliated Hospital of Jinan University.
We reviewed patients with DCM who were treated in our institution from 2016 to 2018. The exclusion criteria for this study were traumatic cervical spinal cord injury, a history of cervical spine surgery and neurologic disease, and other traumatic diseases. The inclusion criteria were DCM, progressive neurological dysfunction, no history of cervical spine surgery or neurologic diseases, obvious neurological symptoms, and performance of spinal pia mater incision for intradural decompression for treatment of degenerative spinal disease.
All patients in this study had signs of upper motor neuron lesions and symptoms of paralysis and were followed up for at least 18 months. We also enrolled patients who underwent only cervical laminoplasty, fusion, and epidural decompression as the historical control group. All patients in the control group underwent surgical treatment in our hospital before 2016.
Surgical intervention
All patients underwent posterior laminoplasty and fusion. If the patient’s dural flow was nonpulsatile or obviously weakened after routine extradural decompression, the spinal pia mater was incised under microscopy followed by separation of the arachnoid adhesion and shunting of the liquefaction zone to restore cerebrospinal fluid flow and spinal blood flow. The extent of laminoplasty was based on preoperative magnetic resonance imaging. The length of the dural incision was based on the cerebrospinal fluid flow condition and generally included all stenotic levels. After completion of extradural and intradural decompression, the focal zone was rinsed with cold saline solution and the spinal pia mater was closed. A drainage tube was placed before closing the surgical incision with sutures. The blood loss and operation time were recorded.
Postoperative treatment
Steroids were administered for 3 days postoperatively. The patients thereafter received combined therapies including routine infection prevention and administration of neurotrophic drugs and gastric mucosal protection drugs. Additionally, high-pressure oxygen and rehabilitation exercises were recommended to accelerate nerve reconstruction and functional recovery.
Follow-up
The preoperative and postoperative neurological status was evaluated according to the Japanese Orthopaedic Association (JOA) scale. The improvement rate of the JOA score was calculated as follows: (postoperative JOA score − preoperative JOA score)/(17 − preoperative JOA score) × 100%.
Statistical analysis
Data analyses were performed using SPSS version 16.0 for Windows (SPSS Inc., Chicago, IL, USA). A t test was performed to determine the differences between the preoperative and postoperative follow-up JOA scores. An independent-samples t test was performed to investigate the differences in age, blood loss, operation time, preoperative JOA score, postoperative follow-up JOA score, and JOA score improvement rate between the two groups. The time until getting out of bed postoperatively (TOB) and the postoperative hospitalization days (PHD) were reviewed. All values are presented as mean ± standard deviation. A P value of <0.05 was considered statistically significant in all analyses.
Results
The study group comprised 13 patients (12 men and 1 woman) ranging in age from 32 to 65 years (mean age, 51.46 ± 9.46 years) (Table 1). The historical control group comprised 20 patients. The neurological symptoms of all patients in both groups were significantly improved at the final follow-up. The mean preoperative JOA score in the study group was 8.07 ± 1.80, and that at the final follow-up was 14.38 ±2.14 (Table 2); the mean improvement rate of the JOA score was 70.88% ± 21.18%. In the study group, the recovery process plateaued after surgery at a mean of 6.61 ± 4.64 months. The blood loss volume was significantly lower and the operation time was significantly shorter in the study group than in the control group (P = 0.037 and 0.007, respectively). The mean recovery time was also significantly shorter in the study group than in the control group (P = 0.000). However, there was no significant difference in the improvement rate between the two groups. The TOB and PHD were significantly longer in the study group than in the control group (P = 0.000 and 0.003, respectively) (Table 2).
Clinical characteristics in the study group.

JOA, Japanese Orthopaedic Association.
Comparison between the study group and historical control group.
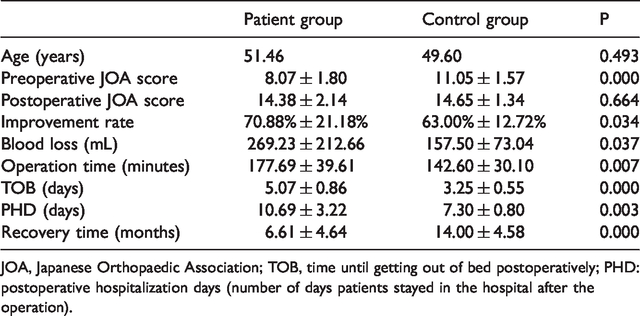
JOA, Japanese Orthopaedic Association; TOB, time until getting out of bed postoperatively; PHD: postoperative hospitalization days (number of days patients stayed in the hospital after the operation).
At the final follow-up, no instrument failure had occurred. One patient had transient postoperative C5 palsy. One patient had a lung infection and two had a urinary tract infection, all of which were cured with sensitive intravenous antibiotics. No pseudarthrosis, cervical kyphotic deformity, or wound infection occurred during the 1-year follow-up. No reoperation was performed. No postoperative neurological deterioration was reported. No patients developed severe vascular or neurological lesions.
Discussion
DCM is the most common cause of spinal cord dysfunction in people of advanced age.1,2 Dysfunction of the spinal cord can cause various clinical neurological symptoms, such as paresthesia, gait disturbance, postural instability, sensory disturbance, and sphincter and balance problems.1,8 To improve DCM, surgical intervention is recommended to relieve the spinal compression, restore the spinal alignment, stabilize the spine, further arrest the neurological deterioration, and accelerate neurological recovery.1,4,8 Surgical decompression procedures are also suggested for DCM and consist of cervical anterior discectomy, corpectomy, posterior laminoplasty, cervical laminectomy and fusion, or combined anterior-posterior approaches. 1 However, the optimal surgical procedure remains controversial. Each procedure has its own advantages and disadvantages in treating DCM.
Since Smith and Robinson 9 first reported anterior cervical discectomy and fusion (ACDF), ACDF has become the most common surgical intervention for treating DCM and radiculopathy. 1 ACDF can directly achieve ventral cervical spinal cord decompression by removing the cervical disc material and fusion by replacing the disc space with an interbody graft. 10 ACDF is regarded as the standard surgical intervention for treating single-level or two-level cervical disc disease. 1 However, ACDF is not suitable for patients with more than three levels of cervical disc disease because of the increased risk of pseudarthrosis caused by the increased number of graft–host interfaces. 11 Corpectomy is an alternative procedure that can reduce the incidence of pseudarthrosis by reducing the number of graft–host interfaces and providing more local autograft. 12 Other than pseudarthrosis, some common complications of cervical anterior discectomy and corpectomy and fusion include recurrent laryngeal nerve palsy, C5 palsy, dysphagia, vertebral artery injury, postoperative hematoma formation, and esophageal injury.1,13–16 Although posterior approaches can avoid some complications that result from anterior approaches, posterior procedures also have limitations. One meta-analysis suggested that anterior procedures result in better postoperative neural function than posterior procedures. 17 In addition, Huang et al. 18 revealed that anterior corpectomy had a higher reoperation rate, longer operation time, and greater blood loss. Liu et al. 19 reported that anterior corpectomy and fusion was the preferred treatment for patients with fewer than three surgical segments, and a posterior procedure was recommended for patients with three or more levels of myelopathy. Different posterior procedures also have their own limitations and advantages. Some researchers have suggested that patients treated with laminoplasty have lower complication rates than those treated with laminectomy and fusion. 20 Liu et al. 21 conducted a meta-analysis showing that laminoplasty had a shorter operation time and lower incidence of C5 palsy than laminectomy and fusion. Chang et al. 22 reported a larger postoperative cervical range of motion in patients treated with laminoplasty than laminectomy. Lau et al. 23 reported that laminectomy and fusion provided greater neurological recovery but results in more blood loss and a higher complication rate than laminoplasty. However, laminectomy without fusion may lead to more severe kyphosis and segmental instability over time than laminoplasty. 24 We recommend designing the surgical plan according to the patient’s individual disease characteristics, such as the number of involved segments, spinal instability, cause of DCM, and preoperative cervical alignment. The surgeon’s experience and proficiency with different surgical procedures are also critical factors that can significantly influence the postoperative clinical outcomes.
In our institution, we prefer laminoplasty and fusion for patients who require surgical intervention of more than two segments. After traditional extradural decompression, we incised the spinal pia mater under microscopy and separated the arachnoid adhesion, then shunted the liquefaction zone if a liquefied cavity was present to restore the cerebrospinal fluid flow and spinal blood flow. Chronic reduction of the intraparenchymal spinal cord blood flow has been shown to result from chronic cervical spinal cord compression and activate some key biological mechanisms, causing further neural degeneration.2,25,26 Chronic intraparenchymal ischemia can induce a unique immune response,2,27,28 resulting in microglia and macrophage accumulation at the site of the compression. 2 Microglia may participate in activating the neural damage process.2,29 The persistent hypoxic and neuroinflammatory response can activate the neuronal cell apoptotic pathway, resulting in progressive neurological dysfunction.2,30–34 Some researchers have observed chronic endothelial cell dysfunction and blood–spinal cord barrier disruption even during the late stages of compression, exaggerating the inflammatory process via peripheral immune cell infiltration.2,25 Our currently ongoing pilot study showed higher levels of interferon γ, interleukin 17F, interleukin 6, and soluble CD40 ligand in the cerebrospinal fluid collected at the site of the myelopathy. Thus, except in areas of cord decompression, improving cerebrospinal fluid circulation and spinal cord blood flow can accelerate postoperative neurological recovery. In theory, a good blood supply and unobstructed cerebrospinal fluid circulation can carry away more inflammatory factors and noxious substances at the site of spinal cord compression, thus reducing the accumulation of inflammatory factors and noxious substances and providing a better microenvironment for neurons. During the surgical intervention in the present study, we found that the dural nonpulsatile or pulsatile flow was obviously weakened in some patients. The cerebrospinal fluid spurted out of the pia mater incision in some patients, and different degrees of arachnoid adhesion were observed at the site of compression (Figure 1). Additionally, the venous circulation of the spinal cord surface was obscure. After the operation, all patients had obvious dural pulsatile flow, and obvious venous dilation was seen on the spinal cord surface (Figure 2). Significant neurological and locomotor recovery was observed at the final follow-up. The final results also showed that the recovery time was shorter in the study group than in the control group. Therefore, intradural scar compression is an important factor that can reduce the spinal cord blood supply and block cerebrospinal fluid circulation, further slowing the recovery process. However, the blood loss and operation time were significantly greater in the study group than in the control group, and the TOB and PHD were significantly longer in the study group than in the control group.

Preoperative arachnoid adhesion. Arachnoid adhesion was noted, and the venous circulation on the surface of the spinal cord was obscure.

Separation of the arachnoid adhesion. After separation of the arachnoid adhesion, venous dilation on the surface of the spinal cord became obvious.
The present study has some limitations. First, this was a small cohort study; a larger cohort study is needed. Second, this was not a randomized controlled study, and the control group was a historical control group. Third, the follow-up time was relatively short; longer-term clinical results are needed. Finally, the surgical indication was weak dural pulsatile flow. This method is not suitable for all patients with cervical spondylotic myelopathy because of the dural incision for intradural decompression.
Conclusion
A pia mater incision with separation of the arachnoid adhesion can significantly improve the cerebrospinal fluid flow and spinal blood flow in patients with DCM. Arachnoid adhesion can lead to intradural spinal scar compression. The surgical intervention described herein can achieve satisfactory neurological outcomes and shorten the recovery time.
