Abstract
Intravascular large B-cell lymphoma (IVLBCL) is a rare (<1%), typically aggressive extranodal variant of mature non-Hodgkin B-cell lymphoma. IVLBCL is characterized by malignant lymphoid cells lodged within blood vessels, particularly capillary channels. Herein, we present a case of a 50-year-old man with a history of myeloradiculitis (∼1 year) and paraparesis requiring hospitalization. During the course of his hospital stay, computed tomography (CT), magnetic resonance imaging, CT-positron emission tomography, and biopsy failed to establish a diagnosis. The patient died 2 months later from bilateral pneumonia. Postmortem examination was undertaken to determine the cause of death. Histologic sections of the patient’s brain, heart, lung, and liver showed aggregates of highly atypical cells bearing enlarged, pleomorphic, and hyperchromatic nuclei. Strong intravascular positivity for CD45 and CD20 markers indicated the cells were of B-cell origin, supporting a diagnosis of IVLBCL.
Keywords
Introduction
The latest World Health Organization disease classification revisions define intravascular large B-cell lymphoma (IVLBCL) as a rare subtype of diffuse large B-cell lymphoma. This malignancy may be difficult to diagnose clinically, sometimes requiring autopsy. 1 Patients tend to present with a confusing symptom complex, signaling vascular occlusive organ dysfunction, or with systemic manifestations such as unexplained fever.2,3
The current case had a complex clinical presentation and diagnostic difficulties. An accurate diagnosis was only finally achieved following postmortem examination, based on immunohistochemical staining.
Case report
The patient was a 50-year-old Caucasian man with a history of myeloradiculitis (∼1 year) who was ultimately hospitalized for paraparesis. No specific lesions or other pathology were detected by total-body computed tomography (CT). A whole-body 18-fluorodeoxyglucose positron emission tomography (PET) scan indicated greater uptake by the adrenal glands and in an area of mesenteric fat (Figure 1a). Serologic tests for infection and autoimmune markers were negative. During the first hospitalization, blood biochemistry analysis showed a lactate dehydrogenase level of 465 UI/L (normal value < 250 UI/L), C-reactive protein level of 35.9 mg/L (normal value <50 mg/L), and beta-2 globulin 6.2% (normal value 3.1% to 6.0%). Magnetic resonance imaging (MRI) of the lumbar spine (with and without gadolinium contrast) showed swelling of the conus medullaris (T11–L1), T2-weighted hyperintensities of the anterior and posterior cord, and vasogenic edema (Figure 1b, 1c).
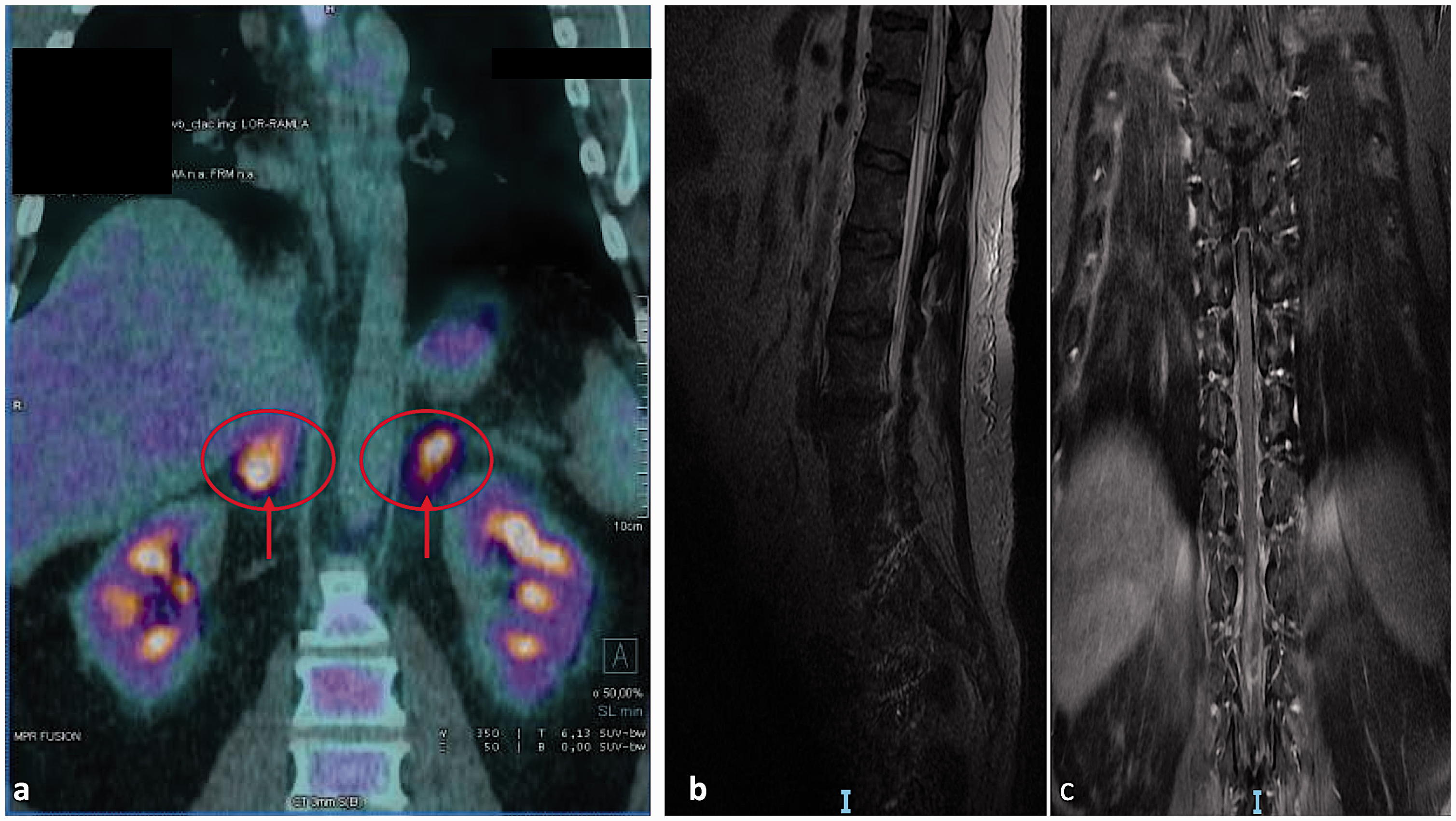
(a) 18-Fluorodeoxyglucose positron emission tomography scan indicated (red arrows and ring) greater uptake by adrenal glands. (b, c) Magnetic resonance imaging of lumbar spine showed swelling of the conus medullaris (T11–L1), T2-weighted hyperintensities of anterior and posterior cord, and vasogenic edema.
Bone marrow biopsy was obtained but was unrevealing, and flow cytometry produced no immunophenotypic signs of clonal cell expansion. Biopsy of the spinal cord disclosed tissue necrosis, with macrophage infiltrates (CD68+) and reactive gliosis, but chromogenic reactivity for microtubule-associated protein 2 was lacking. A dermatologic consultant found no suspicious melanocytic lesions.
The patient developed a pulmonary thromboembolism during hospitalization and his condition progressively worsened. He died of bilateral pneumonia 2 months after the development of thromboembolism.
Postmortem examination was subsequently conducted to establish the cause of death. Gross inspection found no focal changes in the spinal cord, only a palpable loss of consistency. Histologic sections of vital organs (brain, heart, lung, and liver) revealed intravascular accumulations of highly atypical cells with enlarged, pleomorphic, and hyperchromatic nuclei, chiefly confined to the blood vessels, but also scattered focally in the heart and liver interstitium. Endothelial lymphocytic infiltrates were also observed (Figure 2a).

Microscopic features of intravascular large B-cell lymphoma. (a) Pleomorphic, highly atypical cells within blood vessels in the brain and endothelial lymphocytic infiltrate (red arrows) (hematoxylin and eosin, ×40). (b, c) Strong intravascular positivity for CD20 and CD45 (brown reactions) (×40); (d) CD3 positivity (red arrows) of endothelial infiltrate (×40); (e) slight intravascular positivity for PD-L1 (red arrows) (×40); and (f–h) negative reactions for CK7, TTF-1, and MCK (×40).
We performed immunohistochemical staining with a panel of antibodies targeting cytokeratins (CK7, CK20), lymphocyte antigens (CD3, CD20, CD45), programmed death-ligand 1 (PD-L1), muscle creatinine kinase (MCK), and thyroid transcription factor 1 (TTF1). Strong intravascular CD20 and CD45 positivity indicated the cells were of B-cell origin, supporting a diagnosis of IVLBCL (Figures 2b, 2c). There was slight intravascular PD-L1 positivity, and evident endothelial CD3 positivity (Figures 2d, 2e). All other markers (CK7, CK20, MCK, and TTF1) were negative (Figures 2f–2h).
Discussion
IVLBCL is characterized by malignant lymphoid proliferations within blood vessels, particularly capillaries. This uncommon (<1%) extranodal form of mature non-Hodgkin B-cell lymphoma tends to behave aggressively and can involve any organ in the body. Although previous reports have indicated that the usual manifestations involve neurologic signs and symptoms,2,3 the neuropathic expressions may vary, including mental status alterations, brief ischemic attacks, deficient motor functions, and paraparesis. 4 Most patients (76%) are prone to central nervous system (CNS) problems as the disease progresses, often (38%) originating in the spinal cord and nerve roots, whereas some patients (25%) show CNS involvement at presentation.5–9
Despite the known potential for false negative results, contrast-enhanced whole-body CT and whole-brain MRI scans, cerebrospinal fluid cytology, peripheral blood smears, bone marrow biopsy, and biochemical blood testing are the recommended diagnostic methods for IVLBCL. Postmortem immunohistochemical staining should target CD20, CD45, and PD-L1 markers. 10
The current case highlights the limitations of standard diagnostic imaging (CT, MRI, and CT-PET) in this particular setting. Testing biopsy samples using a battery of key immunohistochemical stains may thus be the best diagnostic strategy in patients with suspected IVLBCL.11,12
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethical approval
This case dealt with a death of judicial interest, for which ethical approval and consent were requested and obtained from the Judicial Authority.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Informed consent
This case dealt with a death of judicial interest, for which ethical approval and consent were requested and obtained from the Judicial Authority.
