Abstract
Objective
This study aimed to describe drug utilisation in a large Chinese neonatal unit and to compare the findings with those from other countries.
Methods
Data were collected from electronic medical records. Prescription drugs were defined as the number of unique medication names for each patient. Medicine doses were defined as the total number of doses of all medicines administered. Information was collected regarding drugs prescribed to inpatients between March 1 and April 1 2018 in the neonatal intensive care unit and the general neonatal ward of West China Second University Hospital.
Results
The 319 neonates received 1276 prescription drugs and 11,410 medicine doses involving 81 drugs. Vitamin K1, hepatitis B vaccine, and cefoperazone-sulbactam were the three most frequently prescribed drugs. Antimicrobials were the most frequently used group of medicines, with cefoperazone-sulbactam and piperacillin-tazobactam the most frequently used in an off-label manner. Domperidone and simethicone were both widely used.
Conclusions
The most commonly prescribed antimicrobials differed greatly from those reported for other countries. The evidence base for the use of some medicines is poor, and is indicative of irrational prescribing.
Introduction
Drug utilisation research is important in determining whether medicines are being used in a rational manner. According to the World Health Organisation, rational prescribing is “when patients receive the appropriate medicines, in doses that meet their own individual requirements, for an adequate period of time, and at the lowest cost both to them and the community”. 1 Irrational drug use is a major problem worldwide. 2 The World Health Organisation estimates that more than half of all medicines used are prescribed, dispensed, or sold inappropriately. These problems are particularly apparent in neonatal practice, and there is a need for pharmacoepidemiological research to describe the scale of the problem in this patient population and identify potential solutions. 3
Most pharmacoepidemiological studies in paediatric populations are performed in outpatient settings,4–7 with few studies conducted in neonates. 2 With advances in neonatology, more vulnerable infants are surviving. A greater number of extremely preterm neonates may lead to changes in the pattern of drug use. Many medicines used in neonates are used in an off-label manner. 7 Additionally, for many medicines, evidence related to their use is lacking, which may be of greater importance than approval status. 8 Increasing our knowledge of the use of medicines in neonates can help in setting priorities for research in this population. 9
In China, about 17 million neonates are born every year, and about 1.2 million of these cases are preterm neonates. Neonatal mortality accounts for more than half of child deaths. 10 Improvements in medical services for the large number of neonates are therefore needed urgently. 11 Medicine administration plays an important part in neonatal care, and a lack of appropriate medicines is a healthcare challenge. However, overuse may also be inappropriate; for example, overuse of antibiotics increases bacterial resistance and the risk of drug toxicity. 12
The present study was conducted to establish the use of medicines in neonates in China to support prioritisation of future research in relation to the rational use of medicines in this patient population. In this study, drug use information was collected at the neonatal unit of West China Second University Hospital, one of the largest tertiary neonatal medical centres in China. This neonatal unit is the largest referral centre for neonates with severe diseases in West China. Furthermore, this unit cooperates with the pharmacy department and is involved in national discussions on Chinese neonatal medication guidelines.
Methods
Study design and objectives
This was a cross-sectional study conducted at one of the largest neonatal units in China. Our unit has 80 beds, including 30 neonatal intensive care unit (NICU) cots and 50 cots on a general ward. Approximately 4000 neonates with internal (medical) conditions or conditions requiring surgery are admitted annually. Surgical cases include neonates who require both general and cardiac surgery. Based on previous studies 2 and on the number of admissions to the unit, we estimated that data collection over a 1-month period would provide information on medicine use in 300 neonates. From March 1 to April 1 2018, data were collected for inpatient cases who were hospitalised for more than 24 hours in the neonatal intensive care unit or on the general neonatal ward of West China Second University Hospital. Patients were divided into four groups by gestational age: extremely preterm (<28 weeks), very preterm (28–31+6 weeks), moderate or late preterm (32–36+6 weeks), and term (≥37 weeks) infants.
Data collection
All data were collected anonymously from electronic medical records. Each infant had a unique hospital number corresponding to his or her identity. Gender, birth weight, gestational age, mode of delivery, discharge outcome, and medication course-related data including dosage, route of administration, and drug use frequency were recorded. The prescribed drugs were counted by nurses using the electronic database of physician prescriptions. We described medication use by two different methods. First, prescription drugs were defined as the sum number of unique medications (one medicine accounted as 1 for each individual, regardless of frequency and duration of use) per patient as in previous studies.13,14 Second, medicine doses were defined as the total number of doses actually administered (frequency × duration). For example, one infant received two medicines (A and B) during hospitalisation. Medication A was prescribed twice a day for 2 days and medication B was prescribed once a day for 3 days. The number of doses in this case would be (2 × 2) + (1 × 3) = 7. The following medications were excluded: intravenous solutions (including 0.9% sodium chloride, 5% or 10% glucose injection, and sterile solution for injection), blood products (except albumin), 1% silver nitrate eye drops, parenteral nutrition, heparin used for venous access, oxygen, and electrolytes (such as calcium gluconate, sodium bicarbonate, magnesium sulphate and potassium chloride), as in previous studies. 13
Information on drug instructions contained within the Summary of Product Characteristics (SPC) was collected by the authors. Medications were considered off-label if they were not used in accordance with the following information of the SPC: (1) therapeutic indication, (2) therapeutic indication for use in subsets, (3) appropriate strength (dosage by age), (4) pharmaceutical form, or (5) route of administration. 15 Drugs were classified manually by two authors (YY and LC), and the classifications were subsequently confirmed by two other authors (TX and JS). Medications were evaluated to determine whether they were used off-label based on the following definition: used for a different indication, age, dose, formulation, or route of administration to that stated in the drug instructions.
Statistical analyses
Descriptive statistical analysis was performed using SPSS for Windows, version 22.0 (SPSS Inc., Chicago, IL, USA) and Microsoft Excel, Version 2010. Categorical variables were summarised using frequencies and percentages and continuous variables were analysed as means, medians, standard deviations (SD), and interquartile ranges (IQR). Mean and SD were used for normal distribution and median and IQR were used for skewed distribution.
Results
Characteristics of participants
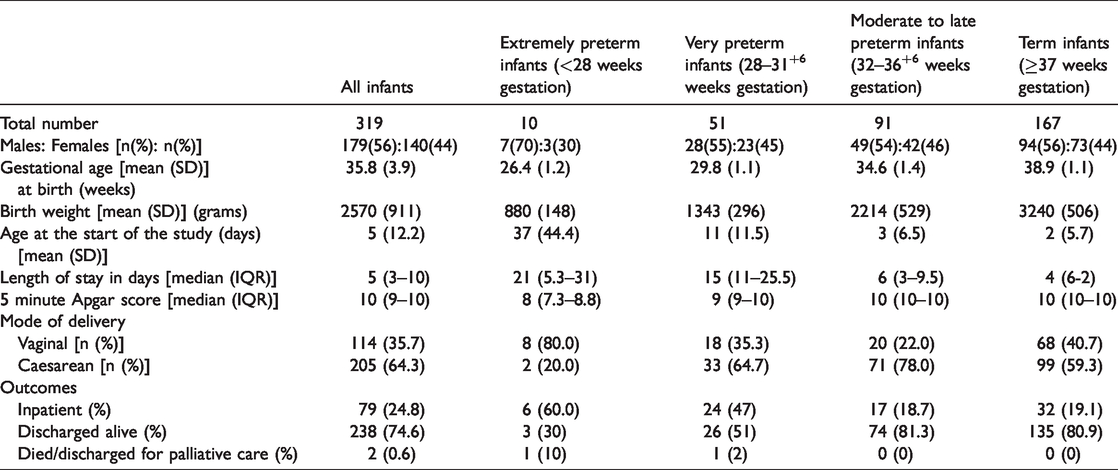
A total of 321 neonates were admitted as inpatients during the study period. Two infants were excluded because of admission < 24 hours and the remaining 319 infants (males, 56%) were included in this study. The mean (SD) gestational age was 35.8 (3.9) weeks and the mean (SD) birth weight was 2570 (911) g (Table 1). Approximately 47% patients were preterm, of whom 60% were moderate to late preterm infants (32–36 weeks gestation). The median (IQR) hospital stay was 5 (3–10) days. Hospital stay decreased with increasing gestational age, with the most premature infants spending the longest time in hospital. Almost two thirds of the infants were delivered by caesarean section. Two patients died or were discharged for palliative care during the study period.
Characteristics of participants.
Utilisation of medicines
The neonates received a total of 1276 prescribed drugs and 11,410 doses of 81 medicines (Table 2). The median (IQR) number of drugs per patient was 3 (1–5.5). Very preterm infants received the largest number of drugs (7 per patient), while term infants received the least number of drugs (2 per patient). The median (IQR) number of doses administered per patient was 13 (1–40), and very preterm infants received the highest number (83 per patient). Intravenous administration was the most commonly used dosing route, accounting for 62% of medicine doses. Other methods of administration were used less frequently: oral 26%, intratracheal 6%, topical 5%, and intramuscular 1%. All intramuscular medications were for hepatitis B vaccine injection.
Number of medicines and prescriptions in the study.

Vitamin K1, hepatitis B vaccine, and cefoperazone-sulbactam, were the three most frequently prescribed drugs (Table 3). The most frequently prescribed drugs varied in different groups. Caffeine and levothyroxine were used most frequently in preterm infants. Among the commonly prescribed drugs, cefoperazone-sulbactam, piperacillin-tazobactam, meropenem, domperidone, mezlocillin-sulbactam, and phosphocreatine were used off-label, and most of these drugs were antimicrobials.
Ten most frequently prescribed drugs.

*Off-label use.
Approximately half of the infants received off-label treatment in this study. The main reason for off-label drug use was age, i.e., lack of information in neonates, accounting for 97% of cases of off-label prescription drug use. For example, meropenem is labelled only for infants aged 3 months and over and domperidone is labelled only for children aged 1 year and over. The other reason for off-label use, accounting for 3% of cases, was indication use. For example, naloxone, which is labelled for respiratory depression caused by opioid drugs, was used for apnoea in prematurity to stimulate spontaneous breathing as a second-line medication.
Cefoperazone-sulbactam, piperacillin-tazobactam, fluconazole, ampicillin, and meropenem were the five most frequently used antimicrobials (Table 4), and their use differed among the four groups. In term infants, mezlocillin-sulbactam was commonly used, whereas it was rarely used in preterm infants. In the three preterm groups, cefoperazone-sulbactam was the most frequently used antimicrobial. Interestingly, vancomycin and tobramycin (eye drops) were only commonly used in extremely preterm infants but not in the other three groups (Table 4).
Five most frequent antimicrobials.

*Off-label use.
Discussion
The aim of the present study was to examine drug utilisation in a Chinese neonatal unit and compare this information with that from neonatal units in other countries. The number of drugs administered for each neonate differs between countries. Previous studies have reported a median of 3 to 11 prescription drugs for each infant.2,16,17 In our study, the median (IQR) number of prescriptions was 3 (1–5.5), which is fewer than that reported by units in Europe, India, and Australia.16–19 This difference may be attributable to differences in study populations and in the medicines included in the prescription count. Studies restricted to preterm and smaller infants are likely to report higher numbers of prescription drugs per patient. More premature and smaller infants may be sicker and require more medications. Over half of the participants in the present study were born at term. Some infants were admitted due to hyperbilirubinemia requiring phototherapy, as many community hospitals in China lack phototherapy facilities; medication is frequently not required in this patient group. Participants in another subgroup in the present study, the extremely preterm infants, had been hospitalised for long periods (the median age at the start of the study was 18.5 days) and were already in a stable condition at the beginning of the study, meaning that there may have been fewer reasons for these participants to receive medications. We excluded medicines for neonatal nutrition, although other studies such as that by Neubert et al. 16 included all nutritional products and reported 8 drugs per patient.
Antimicrobials are the most commonly used medicines in neonatal wards. 20 Notably, significant differences in antimicrobial use exist between our research and other studies. Overall, it appears that the antimicrobials used in neonatal units are similar among the majority of high-income countries, with prevalent use of gentamicin, ampicillin, and vancomycin.14,20,21 However, in the present study, none of the above antibiotics were commonly used, with cefoperazone-sulbactam and piperacillin-tazobactam instead found to be most frequently prescribed. Ampicillin resistance is a significant health issue in China. 22 Furthermore, Chinese guidelines prohibit gentamicin use in neonates due to the risk of hearing loss. 23 Vancomycin, used for the treatment of serious infections caused by Gram-positive bacteria, is ranked among the top five most commonly used antimicrobials in several studies.14,24 In our unit, vancomycin was primarily used to treat sepsis in extremely preterm infants. Coagulase negative staphylococcus, Escherichia coli, Klebsiella pneumoniae, and Pseudomonas aeruginosa are the most common pathogens leading to neonatal infections in Chinese neonatal units, although bacterial spectra differ by region.25–27 These bacteria are sensitive to cefoperazone-sulbactam and piperacillin-tazobactam in China,25,27 providing a plausible rationale for the common use of those two antibiotics.
Neither cefoperazone-sulbactam nor piperacillin-tazobactam has been adequately studied in neonates. Cefoperazone is a third-generation cephalosporin with poor stability to beta-lactamase. Sulbactam, a beta-lactamase inhibitor with relatively weak antibacterial activity, has a strong and irreversible inhibitory effect on the beta-lactamase produced by Staphylococcus aureus and most Gram-negative bacteria. The combination of cefoperazone and sulbactam has a stronger antibacterial effect and broader antibiotic spectrum than cefoperazone alone. 28 At present, no studies have evaluated the recommended dose or frequency of administration for cefoperazone-sulbactam in neonates. Piperacillin-tazobactam, a combination of the semisynthetic piperacillin and the beta-lactamase inhibitor tazobactam, is used to treat moderate to severe hospital-acquired infections. Piperacillin-tazobactam possesses good antimicrobial activity against some beta-lactamase-producing bacteria resistant to piperacillin. Piperacillin-tazobactam has been approved for use in children over 2 months of age.29,30 Only one study to date has provided pharmacokinetic information on piperacillin-tazobactam in infants younger than 2 months. 30 Although cefoperazone-sulbactam and piperacillin-tazobactam seem to have little toxicity, their safety profile in neonates remains unknown. Some studies have reported that cefoperazone and piperacillin may disrupt coagulation function, leading to increased risk of bleeding in neonates.29,31 Meropenem was also found to be commonly used in our study, accounting for 9% of prescriptions. In contrast, meropenem ranked number 52 in the USA among all commonly used medications, with only 0.7% exposure. 14 Meropenem has strong stability to the hydrolysis of most beta-lactamases, thus exhibiting a broad antibacterial spectrum and strong antibacterial activity. 32 Therefore, meropenem is often used as first-line treatment for severe hospital infections, multidrug-resistant bacterial infections, and enzyme-producing bacterial infections in the neonatal unit. Several studies have evaluated the pharmacokinetics, pharmacodynamics, and safety of meropenem in neonates.33,34 The findings of these studies indicate that meropenem is well tolerated in neonates, with minimal toxicity.33,34 In addition to safety concerns related to the use of off-label antimicrobials, the risk of drug resistance resulting from frequent use of broad-spectrum antimicrobials such as cefoperazone-sulbactam, piperacillin-tazobactam, and meropenem is a significant issue. Although these antibiotics seem to be effective in treating certain diseases, over-reliance on these broad-spectrum, potent antibiotics may be counterproductive and may lead to the induction of drug-resistance bacteria. These broad-spectrum antibiotics should therefore be used with caution.
Preterm neonates, neonates with intravenous indwelling catheters, and neonates receiving broad-spectrum antibiotics (especially third- or fourth-generation cephalosporins) are susceptible to candida infection. 35 Fluconazole is recommended routinely for neonates with suspected candida infection. 36 In our neonatal unit, fluconazole is typically used as a preventive treatment for preterm and term infants and is ranked as the third most prescribed medicine. In other studies, fluconazole was primarily used in extremely low birth weight infants at high risk of invasive candidiasis.14,16 In recent years, an increasing trend for fluconazole administration has been observed, which may be attributable to improved understanding of its safety and efficacy.14,16
Domperidone, simethicone, and levothyroxine are rarely prescribed in the neonatal units of other countries. In the present study, domperidone was used to treat neonatal gastroesophageal reflux (GER), despite a lack of evidence to support its use in this patient population. Furthermore, domperidone is associated with an increased risk of adverse events such as ventricular arrhythmias and unpredictable prolonged QT intervals. 37 Although proton pump inhibitors (PPIs) remain the optimal choice for treating GER in adults and older infants, their efficacy and safety in neonates remains uncertain. 38 Studies have reported that PPIs may increase the risk of respiratory tract infection, necrotising enterocolitis, and osteopenia in neonates and may therefore not represent the best first choice for the treatment of GER in neonates.3,37,38 Well-designed studies are needed to explore the best choice of drug (if any) for the treatment of neonatal GER.
Simethicone was commonly used for abdominal distension in this study, and is believed to reduce the formation of gas in the intestine and potentially relieve intestinal flatulence. 39 However, there is a lack of good quality evidence for the efficacy of simethicone in infantile colic or other abdominal conditions. 40 Simethicone use in the present study might therefore represent an example of irrational prescribing.
The use of levothyroxine for preterm infants remains controversial. In our unit, levothyroxine is generally used for preventing hypothyroidism in preterm infants. However, the benefits of routine levothyroxine administration on neurodevelopmental outcomes have been questioned, and it is possible that low thyroxine rather than insufficient thyroid hormone are associated with neurodevelopment.41,42
One limitation of the present study was its single-unit design conducted over a relatively short period. However, as one of the largest neonatal units directly affiliated to the national health commission of China, our unit is likely to be representative with respect to medicine utilisation. We have developed a standard handbook for neonatal medicine utilisation based on label information and the Chinese pharmacopoeia. This handbook has been generally adopted by other neonatal units in West China. The abundance of patients in our unit guaranteed the representativeness of the sample in a relatively short period (1 month), and our findings were similar to those from other neonatal drug utilisation studies published in Chinese units.43,44 Vitamin AD and vitamin K1 were two of the most frequently used medicines in an affiliated hospital of a medical college in Beijing. 43 In these previous studies, β-lactam antibiotics were generally accepted as first-line antibiotics and piperacillin-tazobactam was extensively used. Aminoglycosides were not used at either of these hospitals.43,44 To date, the impact of drug utilisation on populations with Chinese characteristics compared with other populations remains unknown. Future research should therefore consider the possible drug resistance, side effects, and pharmacoeconomic evaluation related to drug utilisation at neonatal units in China. Such studies may potentially contribute a change in the Chinese guidelines, such as an update to the current prohibition of gentamicin use in neonates.
The incidence of off-label medication use in neonatal units varies significantly between countries. Outside of China, 23% to 63% of all drug use in neonatal units has been reported as off-label, and the proportion of off-label prescription drugs in neonatal units ranges from 34% to 90%.2,13,16,21 In the present study, 81 products were used, of which 17 (21%) were prescribed in an off-label manner. Off-label drugs were administered to 53% of the neonates included in this study. Other studies have reported this proportion to be as high as 71% to 100%.2,13,16,21 However, it is difficult to directly compare the results of off-label drug utilisation because regulatory approvals, definition of off-label use, and study methodology are heterogeneous. For example, caffeine is classified as an unlicensed drug in Slovakia, 40 while in China caffeine is licensed for use in neonates. More important than labelling and off-label use, however, is whether there is a clinical evidence base for the safety and efficacy of a given medicine in neonates.
It is difficult to accurately determine the extent of irrational prescribing in neonates, and no tool exists to detect the practice in this patient population. 3 Multiple tools are available for use in older patients, but there is only one for use in paediatric patients – Pediatrics: Omission of Prescriptions and Inappropriate Prescriptions (POPI). 45 This tool is not designed to evaluate drug use in neonates, however. Although intramuscular administration of medicines is a significant problem in many countries because of complications such as sciatic nerve damage 46 and concerns regarding pain, this route of administration was found to be appropriate in the present study.
Irrational prescribing of drugs may place neonates at substantial risk of unintended complications. Unfortunately, this situation is unlikely to improve in the near future, as rational prescribing is not a key area of research, especially in neonates. There remains an urgent need to build research capacity and activities in pharmacokinetic/pharmacodynamic modelling for neonates. In recent years, new research methods have been applied to neonatal clinical medication research such as allometric scaling, population pharmacokinetics, and physiological pharmacokinetics. Preliminary neonatal pharmacokinetic/pharmacodynamic data can be obtained by drawing on available clinical research data from adults or older infants and referring to the state of organ maturity and physiological characteristics of neonates. Such preliminary data combined with further clinical validation could provide information on clinical drug medication in neonates.47,48
Conclusion
The most commonly prescribed antimicrobials differed between the Chinese neonatal unit in the present study and neonatal units in other countries. The choice of antibiotics identified in our study was somewhat unusual and may warrant further investigation. However, the pattern observed must be judged in view of the local pathogen burden, sensitivity, and risk of side effects in the Chinese context, particularly when intravenous use of aminoglycosides is restricted. A major concern, as highlighted by other studies, is non-evidence-based and potentially harmful irrational prescribing of medications such as the novel broad-spectrum antibiotics, simethicone and domperidone. A concerted international effort to develop rational prescribing in neonates is therefore needed.
Footnotes
Consent
Data collection was anonymous and informed consent was not required because of the retrospective nature of the study.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethical approval
Formal ethical approval is not required because this is a retrospective study and all data were collected anonymously.
Funding
This study was funded by the Deep Underground Space Medical Center (no. DUGM201809), the National Science Foundation of China (nos. 81330016, 81630038, 81771634, 81300525, and 81300524), the National Key R&D Program of China (2017YFA0104200), the Grants from Ministry of Education of China (IRT0935), the Grants from Science and Technology Bureau of Sichuan Province (2016TD0002), and the Grant of Clinical Discipline Program (Neonatology) from the Ministry of Health of China (1311200003303).
