Abstract
Pulmonary epithelioid hemangioendothelioma (PEH) is a rare tumor of low to intermediate malignancy, which originates from vascular endothelial cells. Most patients with PEH are asymptomatic and the tumor occurs most frequently in women. Typical radiologic images of patients with PEH are multiple irregular nodules with punctate calcification and pleural indentation. Here, we describe a 54-year-old woman who presented with multiple bilateral nodules of different sizes and well-defined borders, as well as lung markings, without punctate calcification or pleural indentation. These atypical computed tomography images resulted in misdiagnosis as metastatic lung cancer. Right upper lobe wedge resection was performed; intraoperative frozen pathologic examination suggested that the tumor was benign. However, immunohistochemical analysis revealed the presence of PEH. Subsequently, the patient chose watchful waiting, rather than chemotherapy. This rare case of PEH with atypical computed tomography findings, which was misdiagnosed as metastatic lung cancer, demonstrates that intraoperative frozen analysis is unreliable; thus, histopathological analysis is necessary.
Keywords
Introduction
Pulmonary epithelioid hemangioendothelioma (PEH) is a rare low-grade malignant neoplasm of vascular origin.1–4 Most patients with PEH are asymptomatic and the tumor occurs most frequently in women. 5 Typical computed tomography (CT) findings include multiple irregular nodules with punctate calcification and pleural indentation. 6 However, it is difficult to make a correct diagnosis based on clinical and radiologic features alone. To adequately distinguish PEH, histopathological analysis is necessary; positive immunohistochemistry staining is the gold standard for diagnosis.7–9 In the present study, we describe a patient with PEH that was misdiagnosed as metastatic lung cancer based on the presence of multiple bilateral pulmonary nodules with clear boundaries.
Case report
A 54-year-old nonsmoking woman without any symptoms or history of lung diseases visited our hospital for a routine CT scan. The results of a recent physical examination had been unremarkable and serum levels of routine neoplastic markers (i.e., alpha-fetoprotein, carcinoembryonic antigen, carbohydrate antigens [CA19-9, CA125, CA50, CA153], and neuron-specific enolase) were within normal ranges. Chest CT images showed multiple small nodules and well-defined borders on bilateral lungs, without punctate calcification or pleural indentation (Figure 1). Metastatic lung cancer was suspected, based on the chest CT images. While common primary tumors with high incidence of lung metastasis (e.g., thyroid carcinoma, renal carcinoma, breast cancer, or liver cancer) were excluded based on laboratory and imaging examination, the origin and malignancy status could not be determined based on percutaneous biopsy. The patient underwent right upper lobe wedge resection by video-assisted thoracoscopic surgery; intraoperative frozen pathologic examination suggested that the tumor was benign. However, immunohistochemical analysis revealed the presence of PEH (Figure 2). The patient selected watchful waiting, rather than chemotherapy. The disease did not show significant progression at the 3-month follow-up visit (Figure 3). Written informed consent was obtained from the patient for the publication of this case report. The local ethics review board did not require review of the case.
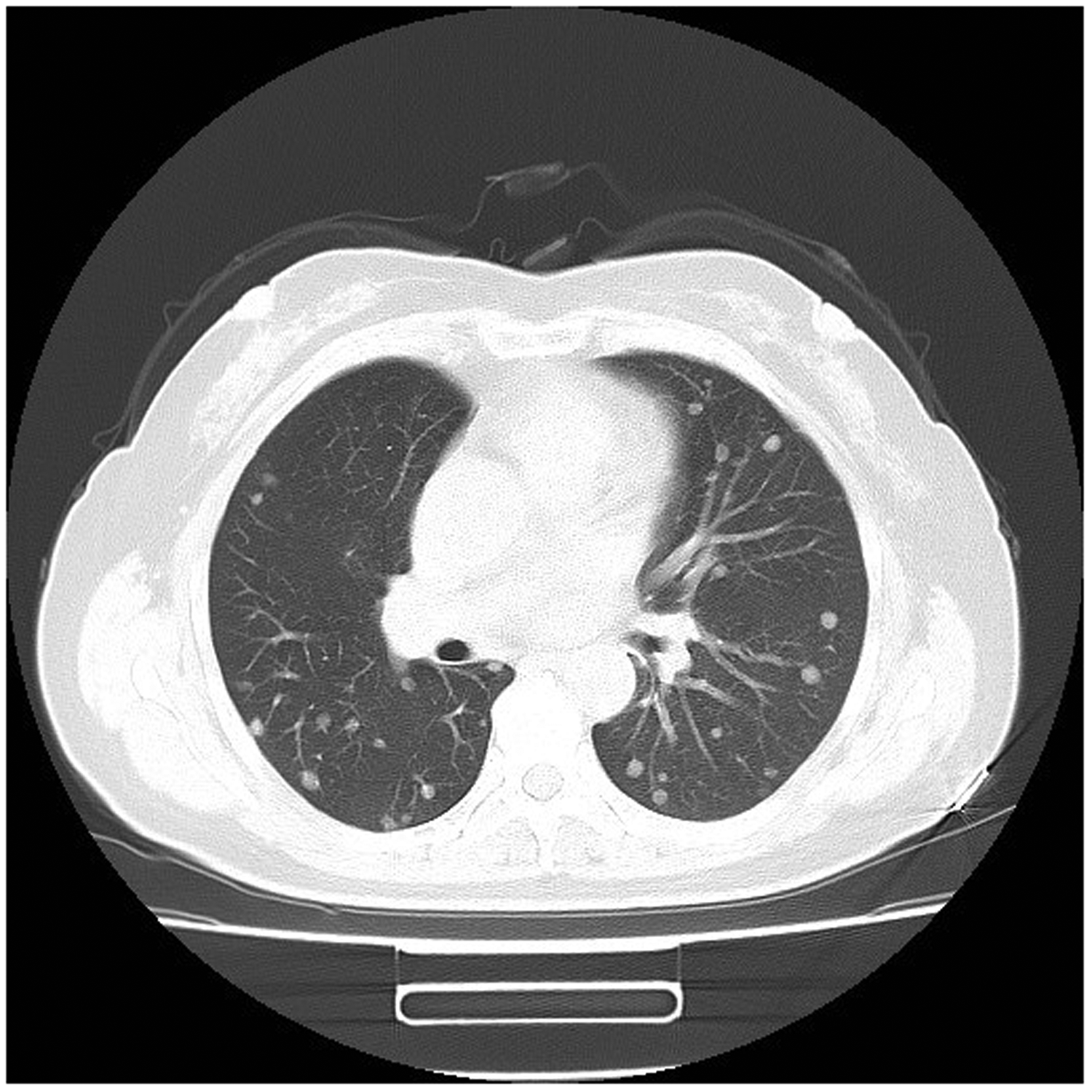
Thoracic computed tomography revealed multiple, well-defined, small, round, and noncalcified pulmonary nodules in bilateral lungs, which were adjacent to bronchioles and medium/small vessels.
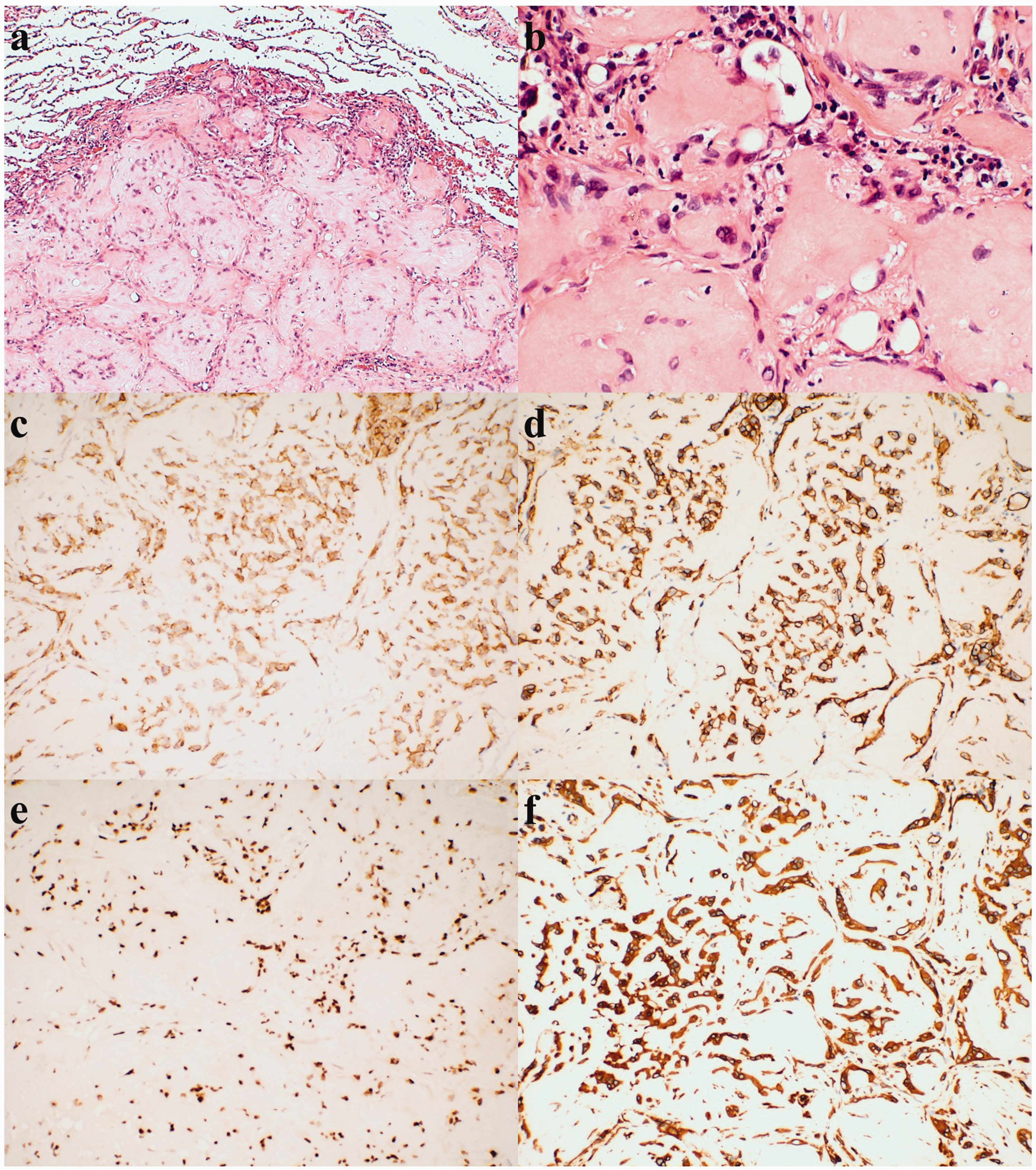
Pathological findings: (a) Neoplastic cells were well-demarcated and rich in eosinophilic cytoplasm, with characteristic intracytoplasmic vacuoles in tumor cells (hematoxylin and eosin, ×10). (b) The boundaries of neoplastic cells were unclear and red blood cells were observed in cytoplasm (hematoxylin and eosin, ×40). (c–f) Immunohistochemical staining showed that tumor cells were positive for CD31 (c), CD34 (d), Fli-20 (e), and vimentin (f) (×20).

Thoracic computed tomography revealed postoperative changes in the upper lobe of the right lung. Scar shadow was visible around the anastomosis. Compared with preoperative CT, the remaining pulmonary nodules exhibited minimal changes.
Discussion
PEH was originally described as an intravascular bronchioalveolar tumor of the lung in 1975. 1 Weiss and Enzinger introduced the term “epithelioid hemangioendothelioma” (EHE) in 1982. 2 The 2015 WHO classification described EHE as a low to intermediate grade malignant vascular tumor. 3 However, EHE has a low prevalence and mainly affects the liver and the incidence of PEH was much less than liver EHE. 4 PEH is a rare tumor that mainly affects young people with a median age of 36 years. 10 The tumor preferentially occurs in women at a rate twofold greater than that in men. 5 Patients with PEH are often diagnosed occasionally during physical examination, without a history of previous symptoms. For symptomatic patients, chest pain, cough, weight loss, hemoptysis, pleural effusion, or dyspnea commonly observed. Significant risk factors for PEH include male sex, chest pain, cough, hemoptysis, multiple unilateral nodules, pleural effusion, and metastases to multiple sites.11,12
Some clinicians have proposed three major CT imaging patterns in patients with PEH: multiple pulmonary nodules, multiple pulmonary reticulonodular opacities, and diffuse infiltrative pleural thickening.13–15 Liu and colleagues analyzed the CT findings of patients with PEH; 6 they found that typical PEH consisted of multiple irregular nodules with punctate calcification and pleural indentation. Our patient exhibited uncommon radiologic features, with multiple bilateral nodules of different sizes and clear and well-defined borders, as well as lung markings. These features are typical in metastatic lung cancer; however, immunohistochemical analysis confirmed the diagnosis of PEH.
Thus far, approximately 160 cases of PEH have been reported worldwide.16,17 The typical microscopic appearance of PEH includes hyalinization, hypercellular nodule periphery, necrosis, hypocellularity, or central calcification with mild or moderate atypia. Vascular-endothelial markers of PEH include CD31, CD34, Fli-1, Fli-20, and Ulex-1; CD31 is relatively specific and sensitive. Other cell markers, such as vimentin, CK, and EMA, are also present in some PEH tumors.7–9 In our patient, immunohistochemical staining assays for CD31, CD34, Fli-20, and vimentin were all positive, which indicated the presence of PEH.
No standard treatment and consensus guidelines for PEH are currently available because of its low incidence. Furthermore, because of the unpredictable nature of PEH, tumor resection should be considered in patients who can tolerate surgery. In patients who undergo surgery, 5-year overall survival is approximately 60%. 18 Chemotherapy is preferred for patients with disseminated disease; 19 however, radiotherapy has been proven ineffective. 20 Spontaneous partial regression has been observed in asymptomatic patients. 11 Therefore, watchful waiting may be appropriate for patients with asymptomatic PEH. 21
In conclusion, we have described a rare case of PEH with atypical CT features, which was misdiagnosed as metastatic lung cancer. For patients with PEH, intraoperative frozen pathologic examination is unreliable and histopathological analysis is necessary. The present report may improve the clinical understanding of PEH.
