Abstract
In the operating room, unanticipated difficult intubation can occur and anesthesiologists can experience challenging situations. Undiagnosed tracheal stenosis caused by congenital factors, trauma, tumors, or post-intubation injury, can make advancing the endotracheal tube difficult. We present an adult patient in whom we were unable to pass an endotracheal tube into the trachea. This was caused by undiagnosed congenital mid-tracheal stenosis with complete tracheal rings. When faced with an unanticipated difficult airway, the anesthesiologist needs to comprehend the results of preoperative evaluations. If an unusual situation (e.g., congenital tracheal stenosis) occurs, active cooperation with other departments should be considered.
Keywords
Introduction
The difficulty of tracheal intubation in the operating room is relatively low, with a rate of difficulty ranging from 1.1% to 3.8%, compared with the emergency department, which has a rate ranging from 3.0% to 5.3%. 1 A recent study reported that tracheal intubation in the operating room had an decreased incidence of difficult intubation compared with that in the intensive care unit (9% vs 16%) in the same patient. 2 During elective surgery, anesthesiologists perform intubation under controlled conditions. Nevertheless, unanticipated difficult intubation can occur and anesthesiologists can experience embarrassing and challenging situations.
Undiagnosed tracheal stenosis caused by congenital factors, trauma, tumors, or post-intubation injury3,4 can cause difficulty in advancing the endotracheal tube. Depending on the location and severity of the stenosis, and the type of surgery, there are multiple options for maintenance of the airway.
We present an adult patient in whom we were unable to pass an endotracheal tube into the trachea. This was caused by undiagnosed congenital mid-tracheal stenosis with complete tracheal rings.
Case report
A 52-year-old woman (weight, 59 kg; height, 162 cm) was scheduled for left microvascular decompression because of involuntary left facial movement. Her medical history showed good functional capacity without respiratory difficulties. She took 150 µg levothyroxine for hypothyroidism. There was no other medical history, including tracheal intubation, and no abnormal findings of routine airway evaluation, including Mallampati class. Preoperative evaluation, including a chest radiograph (Figure 1), electrocardiogram, spirometry, and routine laboratory results, were normal.

Preoperative chest X-ray shows tracheal stenosis.
Routine monitoring equipment, including an electrocardiogram, a pulse oximetry device, and a noninvasive blood pressure monitor, were attached to the patient. After preoxygenation with 100% oxygen, general anesthesia was induced using a target-controlling infusion system (Orchestra; Fresenius Vial, Brezins, France) with propofol and remifentanil. After confirming loss of consciousness of the patient using the eyelid reflex, 50 mg rocuronium was injected. There were no difficulties with manual mask ventilation. The vocal cords were exposed by conventional direct laryngoscopy and the patient appeared to be Cormack–Lehane grade 1. A 6.5-mm inner diameter (ID) reinforced endotracheal tube (8.9-mm outer diameter [OD]; Lo-Counter Oral/Nasal Tracheal Tube cuffed, Shiley™; Covidien, Bedford, MA, USA) easily passed by the vocal cords. However, the tube could not be advanced further because of resistance. The tube was withdrawn and a smaller tube (6.0-mm ID and 8.4-mm OD) was attempted. This tube also could not be advanced passed the same point. The anesthesiologist then asked for assistance. Another anesthesiologist made a third attempt with a 6.0-mm ID tube, but the intubation failed. Additional attempts were considered inappropriate because there was the possibility of vocal cord and tracheal edema. The operation was postponed for further evaluation. The procedure time was 15 minutes. Therefore, the anesthesiologist awakened the patient with 600 mg (10 mg/kg) sugammadex (Bridion; Merck Sharp & Dohme, Kenilworth, NJ, USA). Dexamethasone (5 mg) was injected for the edema. A neurosurgeon consulted an otolaryngologist for evaluation of the airway. Flexible endoscopy by the otolaryngologist showed no tracheal abnormalities. The patient and the neurosurgeon requested anesthesia on the afternoon of the same day with the suspicion of a transient tracheal spasm.
A second anesthesia was induced with propofol and cisatracurium (Nimbex; Mitsubishi Tanabe Pharma Co., Osaka, Japan) because of the use of sugammadex 6 hours previously. There were no difficulties with manual mask ventilation. Before attempting tracheal intubation, flexible fiberoptic bronchoscopy (FOB) was considered to identify the reason for the resistance. FOB showed a reddish lesion that was suspected from the previous intubation trial, but unlike the evaluation by the otolaryngologist, it also showed a stenotic portion in the mid-trachea (Figure 2). The patient was awakened using 3 mg neostigmine and 0.6 mg glycopyrrolate with train-of-four neuromuscular monitoring. She was observed in the post-anesthesia care unit for 90 minutes because of concerns of recurarization.
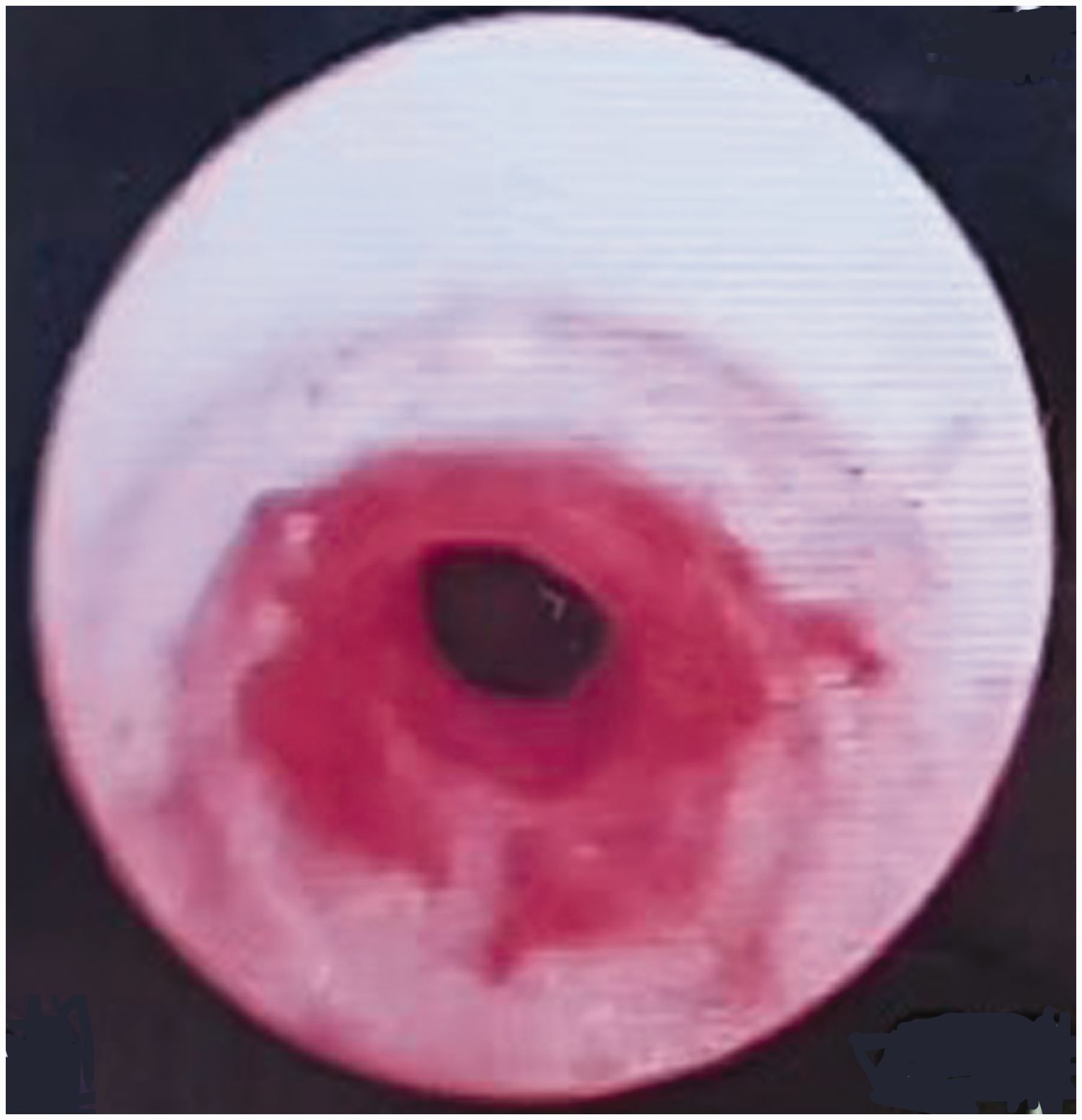
Intraoperative bronchoscopy shows a reddish lesion and a stenotic portion of the mid-trachea.
In the evening, neck computed tomography (CT) was performed, but a radiographic reading by a radiologist was non-specific. The next day, she visited a pulmonologist and was recommended to have a virtual chest CT scan, which was performed using a 64-detector CT scanner (LightSpeed VCT; GE Healthcare, Milwaukee, WI, USA). Chest CT showed segmental congenital tracheal stenosis at 2 cm above the carina. The lesion was 8.27 mm at the shortest diameter and 3.9 cm in length. Sagittal reformation images at 2 mm in thickness showed smooth undulation involving the posterior wall of the trachea, which represented a cartilaginous ring that replaced the membranous portion. Virtual CT-bronchoscopy showed a complete tracheal ring. The proximal and distal parts of the trachea were normal and there were no cardiopulmonary abnormalities (Figure 3a–c). After evaluation by a pulmonologist, she was finally diagnosed with congenital mid-tracheal stenosis with complete tracheal rings. However, the patient did not require additional treatment because she had no respiratory symptoms and the degree of tracheal narrowing was tolerable. After a conversation with a pulmonologist and neurosurgeon, we planned to place a 6.5-mm ID reinforced endotracheal tube above the stenotic site if there was sufficient space between the vocal cords and the tracheal cuff (distance from the cuff to the tip was 8.5 cm). The second option was placing a smaller endotracheal tube through the stenotic site using FOB.

Virtual chest computed tomographic and bronchoscopic images. (a) Three-dimensional volume rendering image shows tracheal stenosis. (b) Sagittal reconstructed computed tomographic image shows cartilaginous undulation in the posterior tracheal wall. (c) Virtual computed tomography-bronchoscopy shows complete tracheal rings involving the posterior wall at the level of stenosis (arrows). (d) Conventional bronchoscopy shows complete tracheal rings.
A third anesthesia was induced. A 6.5-mm ID reinforced endotracheal tube was successfully placed under FOB guidance into the normal portion of the trachea immediately above the stenotic site (Figure 3d). We fixed the tube 20 cm from the incisor. After the patient was placed in the lateral park bench position, the anesthesiologist confirmed the appropriate position of the tracheal tube with a bronchoscope. Throughout the operation, all respiratory parameters were well controlled under volume-guaranteed (tidal volume: 370 mL), pressure-controlled ventilation with a positive end-expiratory pressure of 5 cm H2O. During the operation, her maximal peak airway pressure was 24 cm H2O and arterial blood gas analyses were in the normal range. After the surgery, the patient was transferred to the surgical intensive care unit for neurological evaluation and postoperative care after confirming full recovery of spontaneous respiration. She did not suffer from any respiratory symptoms during the hospital stay.
Ethical permission was approved by the Institutional Review Board of Chonnam National University Hwasun Hospital (CNUHH-2019-117). Written informed consent was obtained from the patient for publication of this case report.
Discussion
Congenital tracheal stenosis is a rare, commonly diagnosed condition in infancy, and is associated with cardiopulmonary abnormality. 5 Stenotic airway lesions are commonly composed of complete tracheal rings of cartilage, which contribute to the severity of luminal narrowing. Cantrell and Guild classified this anomaly into three morphological types: generalized hypoplasia, funnel-like stenosis, and segmental stenosis. 6 In segmental stenosis, a 2 to 5-cm segment of the trachea narrows in an hourglass fashion. In our case, the patient had a 3.9-cm segment and a narrow trachea in the mid-low portion with complete tracheal rings, which hindered the advancement of the tracheal tube.
Stenosis was confirmed via virtual chest CT and flexible endoscopy in our patient. Flexible endoscopy is the invasive gold standard procedure for diagnosing endoluminal lesions. However, the availability of noninvasive virtual chest CT (virtual CT bronchoscopy) is increasing, and it has a diagnostic sensitivity of 94% to 100% for identifying airway stenosis.7,8 Three-dimensional reconstruction of super high-resolution helical CT images is used to delineate tracheal anatomy, and is particularly useful for detecting stenosis that is located in the central airway.
In our case, preoperative chest radiography and chest CT were re-examined after the failed intubation and they showed tracheal narrowing compared with the virtual chest CT. We may not have detected the tracheal narrowing because distinguishing stenosis in the T3 and T4 spinous processes and laminar density without significant clinical symptoms or impressions is challenging. We mostly relied on radiological interpretations, focusing on lung parenchyma abnormalities, unlike pediatric patients who often have a congenital anomaly. Inadequacy of clinical information contributes to errors by radiologists. 9 Therefore, anesthesiologists should review the preoperative radiological findings independently. As in the present patient, non-symptomatic mild tracheal stenosis is not easy to identify.
In the operating room, anesthesiologists rarely confront difficult airways. According to the Practice Guidelines of the American Society of Anesthesiologists Task Force, 10 an airway is considered to be difficult when a conventionally trained anesthesiologist experiences difficulty with facemask ventilation of the upper airway, difficulty with tracheal intubation, or both. Tracheal intubation is considered difficult when it requires multiple attempts in the presence or absence of tracheal pathology. Difficulty in advancing the tube into the trachea after it has passed between the vocal cords is not common. This problem may be due to tracheal stenosis resulting from thyroid or mediastinal tumors, previous tracheostomy, prolonged intubation, traumatic lesions, or a congenital anomaly.3,4
According to the algorithm from the Practice Guidelines of the American Society of Anesthesiologists Task Force, unconscious patients who ventilate well with a mask are followed through the nonemergency pathway. In our case, we did not want to attempt intubation more than three times. We also did not consider the feasibility of other options, such as placing a supraglottic airway device, because of the planned surgical position. At our institution, neurosurgeons prefer the park bench position, which involves neck flexion and rotation, 11 for microvascular decompression. Therefore, tube kinking, elevated peak airway pressure, inadequate ventilation, airway swelling, and airway obstruction are possible. 12 Consequently, an armored (reinforced) endotracheal tube is used to prevent these complications.
The endotracheal tip must be placed at the appropriate depth. Placement of this tube too deep causes atelectasis of the contralateral lung, hyperinflation of the intubated lung, and arterial desaturation. 13 By contrast, placement that is too shallow can impinge the vocal cords, produce trauma, and cause unanticipated extubation according to the head position. 14 To ensure correct placement of the tube, various techniques have been used, such as a chest X-ray, FOB, auscultation, ultrasound, and capnography. 15 However, there are limitations when using these methods intraoperatively.
Diagnosing and treating a patient with a contemporary multidisciplinary management approach is important. In our case, after the initial intubation failure, an otolaryngologist requested evaluation of the airway to determine if there was any tracheal abnormality. The otolaryngologist found an upper airway–larynx subglottic lesion, which is a common cause of difficult passage of an endotracheal tube. If the anesthesiologist or otolaryngologist had performed a tracheal examination with FOB before the end of the first anesthesia, or if the anesthesiologist had communicated in detail with the otolaryngologist about difficult endotracheal intubation, the tracheal stenosis would have been diagnosed earlier. In this scenario, an unnecessary second anesthesia would not have occurred. Direct visualization of the stenosed tracheal segment enables accurate assessment. 5 A multidisciplinary team discussed the patient’s radiological examination together to confirm the appropriate depth of intubation for the third attempt.
When an unanticipated difficult airway occurs, anesthesiologists need to comprehend the results of preoperative evaluations. If an unusual situation, such as congenital tracheal stenosis occurs, active cooperation with other departments should be considered.
