Abstract
Amniotic fluid embolism (AFE) is a rare disease, but it is a potentially fatal condition, as well as a leading cause of unpredictable maternal death in developed countries. The typical clinical symptoms of AFE include hypoxia, hypotension, and coagulopathy, and with rapid progression, it may cause cardiac arrest and death. We report a case of sudden hemodynamic instability due to amniotic fluid embolism, which was successfully treated with veno-arterial extracorporeal membrane oxygenation (ECMO). A 39-year-old woman was scheduled for delivery at 38 weeks and 2 days of gestation. During labor, sudden generalized tonic-clonic seizures developed, followed by dyspnea and cyanosis. Despite immediate cesarean section being performed, she became hypoxemic and experienced cardiovascular collapse. ECMO was promptly applied, and the patient became stable 8 days after cesarean section. ECMO appears to be an appropriate treatment option for catastrophic amniotic fluid embolism.
Keywords
Introduction
Amniotic fluid embolism (AFE) has an incidence of approximately 1/40,000 cases, with a mortality of almost 60%. 1 Although AFE is a rare disease, it is a potentially fatal condition, as well as a leading cause of unpredictable maternal death in developed countries. 2 AFE occurs when a destroyed maternal–fetal barrier during delivery leads to maternal exposure to fetal tissues, which then causes clinical symptoms. The typical clinical symptoms of AFE include hypoxia, hypotension, and coagulopathy, and with rapid progression, it may cause cardiac arrest and death. 3 AFE is occasionally diagnosed under clinical suspicion, and the patient may recover within several hours if immediate emergency treatment is performed upon the onset of transient hemodynamic decompensation. 4 Maintaining hemodynamic support is essential in interventions of AFE and hemodynamic support focuses on augmentation of blood pressure and oxygenation support. Prior research on advanced hemodynamic support for AFE is scarce. There have been reports on the use of extracorporeal membrane oxygenation (ECMO), intra-aortic balloon counterpulsation, and continuous hemodiafiltration with corresponding survival rates, but interventions are still dependent on experimental approaches.4,5
ECMO was first used in the 1970s for the long-term support of severe respiratory failure. Widespread use of ECMO was not considered until ECMO was found to clearly lower mortality and the incidence of disability compared with conventional ventilator support in a relatively recent clinical trial on conventional ventilatory support versus ECMO for severe adult respiratory failure (CESAR). 6 Therefore, the potential utility of ECMO in a variety of clinical environments is still not well known. We report a case of successful treatment of sudden hemodynamic instability in a patient with AFE using veno-arterial ECMO.
Case report
A 39-year-old woman at 38 weeks and 2 days of gestation (G4P2012) visited a local clinic for delivery. The woman had one miscarriage previously and two normal vaginal deliveries. The patient received periodic prenatal care and all prenatal test findings were normal. Her vital signs were normal, with a blood pressure (BP) of 128/75 mmHg, pulse rate (PR) of 82 beats/minute, oxygen saturation of 100%, and body temperature of 36.7°C, with no allergic history.
The patient went into labor after admission and induced delivery was begun with oxytocin at 1.2 units/hour. After 4 hours since beginning induced delivery, an analgesic epidural catheter was placed, through which ropivacaine 6 mg/mL and fentanyl 4.46 μmol/L were continuously infused at 3 mL/hour. The cervix was dilated to 4 cm after 6 hours since beginning induced delivery. While attempting to perform a non-stress test to check on the status of the fetus, the mother showed a mental change with generalized tonic-clonic seizures that persisted for approximately 10 minutes. Generalized cyanosis with reduced oxygen saturation was observed later. Intubation was performed immediately and the patient was transferred to our emergency center.
Upon arrival at the emergency center, the patient’s vital signs were a BP of 108/70 mmHg, PR of 111 beats/minute, and oxygen saturation of 88%. She was in a semicoma, with a pupil size of 4 mm on the right and 3 mm on the left, and the fetal heart rate was 50 beats/minute. A blood test showed a prothrombin time (PT) of ≥120 seconds, activated partial thromboplastin time (aPTT) of ≥180 seconds, and fibrinogen level of <1.764 mmol/L. Additionally, the platelet count was 35 × 10³/μL, antithrombin III was 79%, and D-dimer level was ≥109.52 nmol/L, which suggested severe disseminated intravascular coagulation (DIC). Arterial blood gas analysis showed severe lactic acidosis, with a blood pH of 7.040, PaCO2 of 31.0 mmHg, PaO2 of 127.0 mmHg, bicarbonate level of 8.4 mEq/L, base excess of −21.0 mEq/L, and lactate level of 1.44 mmol/L. We considered that the patient had AFE and decided to perform emergency cesarean section. Therefore, the patient was immediately taken to an operating room.
Vital signs immediately after arrival at the operating room were as follows: BP, 105/77 mmHg; PR, 124 beats/minute; oxygen saturation, 89%; and body temperature, 36.5°C. Anesthesia was induced with propofol 100 mg and rocuronium 40 mg while supplying oxygen at 100%, and the patient’s endotracheal tube was connected to a ventilator. Anesthesia was maintained with a mixture of oxygen, air, and sevoflurane 1.5 vol% at a fraction of inspired oxygen of 0.5 and total flow of 4 L/minute. Intraoperative mechanical ventilation was set to a pressure control ventilation–volume guaranteed mode with a tidal volume of 400 mL, peak airway pressure of 25 mmHg, and respiratory rate of 14 beats/minute. Immediately after induction of anesthesia, arterial cannulation was performed on the left brachial artery for invasive arterial BP monitoring, and a central venous catheter was inserted into the right subclavian vein. Arterial blood gas analysis performed immediately after beginning surgery showed a blood pH of 7.130, bicarbonate level of 16.3 mEq/L, base excess of −12.1 mEq/L, and lactate level of 0.95 mmol/L. At this time, a neonate was born through cesarean section. The 1-minute and 5-minute Apgar scores of the neonate were 1 and 2, respectively. A pediatrician performed cardiopulmonary resuscitation (CPR) and intubated the neonate immediately after birth, and the neonate was transferred to the neonatal intensive care unit (ICU).
After delivery, 80 mL of sodium bicarbonate 8.4% was administered to the mother. Her BP dropped to 85/50 mmHg, PR was increased to 133 beats/min, and oxygen saturation was 90%. Dopamine 20 μg/kg/minute and dobutamine 25 μg/kg/minute were continuously administered to maintain BP. However, BP further dropped to 57/39 mmHg 10 minutes later. Therefore, we began red blood cell (RBC) and fresh frozen plasma (FFP) transfusion while additionally administering norepinephrine 1 μg/kg/minute. Portable echocardiography results suggested left heart failure, with a left ventricular ejection fraction of 23%. Additionally, oxygen saturation dropped to 73% and BP to 35/25 mmHg. Therefore, we performed veno-arterial ECMO insertion into the right femoral artery and vein using the Seldinger technique to maintain oxygenation and BP while infusing vasopressin at 0.10 units/minute.
After ECMO insertion, BP rose to 79/57 mmHg, but bleeding still persisted. Therefore, cesarean hysterectomy was performed with additional insertion of a central venous catheter into the right internal jugular vein. During this process, RBC, FFP, and platelet concentrate (PC) were continuously transfused. After cesarean hysterectomy, bleeding stopped and BP rose to 111/85 mmHg. Dopamine and dobutamine were stopped, and norepinephrine was reduced to 0.3 μg/kg/minute and vasopressin to 0.05 units/minute. After 10 minutes, BP was stabilized and vasopressin was also stopped with norepinephrine reduced to 0.1 μg/kg/minute. Surgery lasted for 3 hours and 15 minutes, and the mother’s vital signs were as follows: BP, 111/88 mmHg; PR, 122 beats/minute; oxygen saturation, 98%; and body temperature, 35.9°C. During surgery, approximately 3000 mL of blood was lost, and 17 units of RBCs, 15 units of FFP, and 20 units of PC were transfused. The patient was transferred to the ICU.
A cardiac marker test, which was performed upon arrival at the ICU, showed an elevation of the creatine kinase-muscle/brain level to 101.40 ng/mL and troponin-T level to 3.310 ng/mL. The ECMO flow rate was 3.47 L/minute and mean arterial pressure was 78 mmHg. Further, the patient also had a weak pulse, which suggested weakening of cardiac contractility. While maintaining norepinephrine at 0.05 μg/kg/minute, isosorbide dinitrate was administered at 0.5 mg/kg/minute to treat acute heart failure. An electrocardiogram that was taken 1 hour after ICU admission showed ventricular tachycardia for approximately 3 seconds and an intermittent ventricular premature complex. Therefore, amiodarone was added at 20 mg/hour. Eight hours after ICU admission, the patient recovered consciousness and was able to open her eyes and nod her head upon being called. From postoperative days (PODs) 0 to 2, 15 units of RBCs, 10 units of FFP, 10 units of PC, 1 unit of apheresis platelets, and 10 units of cryoprecipitate were additionally transfused at the ICU.
On POD 3, the patient had a BP of 128/75 mmHg and PR of 95 beats/minute. Therefore, norepinephrine was stopped. However, acute bleeding of approximately 150 cc/hour occurred through the closed wound drainage system (MEDVAC; Hyupsung Medical Co., Ltd., Seoul, Korea) that was placed in the patient’s surgical site. Therefore, the patient was taken to the operating room to evacuate the hematoma that was observed in the muscle layer and perform suturing and electrocauterization for the wide-ranging bleeding in the vagina and bladder area. Upon arrival at the operating room, the patient’s BP was 134/86 mmHg, PR was 89 beats/minute, oxygen saturation was 99%, and body temperature was 37.7°C. Anesthesia was induced with propofol 60 mg and rocuronium 40 mg, and the patient’s endotracheal tube was connected to a ventilator. Anesthesia was maintained with a mixture of oxygen, air, and sevoflurane 1.5 vol% at a fraction of inspired oxygen of 0.5 and total flow of 4 L/minute. Intraoperative mechanical ventilation was set to the pressure control ventilation–volume guaranteed mode with a tidal volume of 400 mL, peak airway pressure of 25 mmHg, and respiratory rate of 14 beats/minute. In addition to continuous infusion of isosorbide dinitrate at 1 mg/kg/minute, 4 units of RBCs, 3 units of FFP, and 10 units of PC were transfused. Surgery lasted for 1 hour, after which the patient was transferred to the ICU.
On POD 4, the patient complained of numbness and pain in the legs. Portable transthoracic echocardiography was performed, which showed no regional wall motion abnormality, but cardiac contractility appeared to be improved with a left ventricular ejection fraction of 57%. Therefore, the flow rate for ECMO was reduced to 1.95 L/minute. The platelet count was 89 × 10³/μL. Therefore, 10 units of PC were transfused. Blood test results showed a normal PT (14.6 seconds), an aPTT of 40.5 seconds, and an international normalized ratio of 1.20. Arterial blood gas analysis results were also normal, with a blood pH of 7.490, PaCO2 of 32.0 mmHg, PaO2 of 114.0 mmHg, bicarbonate level of 25.1 mEq/L, base excess of 2.2 mEq/L, and lactate level of 0.11 mmol/L. The rate of ECMO was continuously reduced until successful ECMO removal on POD 5. During ECMO removal, 3 units of RBCs and 2 units of FFP were additionally transfused.
Following ECMO removal, furosemide 20 mg was intravenously injected to lower pleural effusion. Extubation was performed on POD 7 because the patient’s cardiac function improved. A chest X-ray radiograph taken on POD 8 showed reduced pleural effusion (Figure 1).
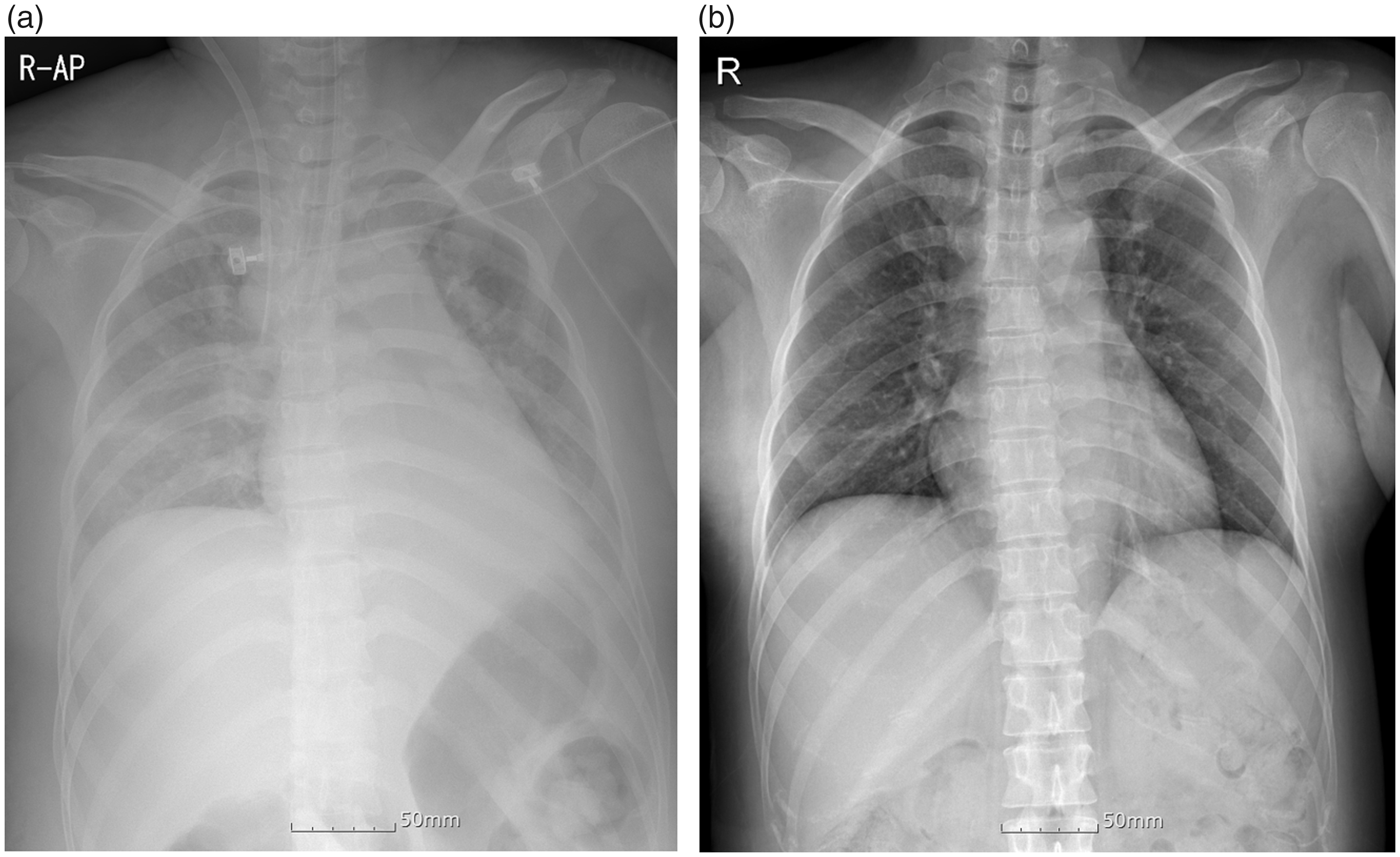
a: Chest radiograph at the time of arrival at the emergency center. b: A chest radiograph shows reduced pleural effusion at 8 days postoperatively.
The patient showed signs of improvement and was transferred to the general ward on POD 10. Despite normal vital signs and mental status, the patient experienced neuropathic pain and symptoms of foot drop in the right leg where ECMO was inserted. The patient was provided gabapentin 300 mg twice a day and a nerve conduction study and electromyography were performed in cooperation with rehabilitation medicine. These examinations suggested an injury to the bilateral sciatic nerve and the lumbosacral plexus with a greater severity on the right side than on the left. Pregabalin 75 mg twice a day was additionally prescribed because of worsening neuropathic pain and numbness in the legs. Daily personal training, which included standing and walking for a bedridden patient, was started for improvement of nerve injury. Additional pain was controlled with lumbar epidural steroid injection, and right tibial nerve block was injected in the right ankle, which had the most severe pain and foot drop symptoms. On POD 22, the patient was able to stand on her own by grabbing onto a pole, and by POD 24, she was able to take small steps while relying on a quad cane. On POD 41, the patient remains hospitalized and is receiving rehabilitation. While the motor grade for the left lower extremity was improved to grade 4, that for the right lower extremity was grade 1 and that for the right ankle was grade 0. This finding indicated major nerve injury complications in the right lower extremity where ECMO was inserted.
This case study was approved by the Institutional Review Board of Eulji University Medical Center (EMC 2019-08-015) and conformed with the EQUATOR Network guidelines. Written informed consent for publication was obtained from the patient’s husband and parents.
Discussion
With the exception of bleeding, AFE is one of the most common causes of peripartum cardiac arrest, with a high mortality of approximately 60%. 2 Although the pathophysiology of AFE has not yet been clearly described, the most widely accepted hypothesis is that fetal tissue in the amniotic fluid enters the maternal circulation and causes pulmonary vascular occlusion, thereby inducing serious collapse of the maternal cardiovascular system. Therefore, when fetal tissue enters the maternal circulation, a complex sequence of hemodynamic alteration is induced, along with abnormal activation and mechanical obstruction of the proinflammatory mediator system. Pulmonary vasoconstriction and hypertension lead to left ventricular failure, followed by acute right ventricular failure, thereby activating a coagulation cascade. 7 This phenomenon is triggered by damage to the maternal–fetal barrier, and the common sites of injury are the endocervical veins, uterine trauma sites, and placental attachment site. 8
Clark et al. 9 suggested that pulmonary vascular occlusion caused by embolism is not the major mechanism of AFE because pulmonary vascular occlusion is not observed in most cases, clinical symptoms vary widely, and inducing similar symptoms in animal studies is difficult. Alternatively, Clark et al. speculated that AFE is a secondary damage of systemic inflammatory response syndrome caused by fetal tissue acting as an antigen in the maternal circulation. Therefore, these authors speculated that AFE and anaphylaxis have high similarity and thus have a fundamentally similar pathophysiology. Clark et al. suggested that AFE should be renamed anaphylactoid syndrome of pregnancy. Further, another report confirmed that distinguishing AFE from anaphylaxis is extremely difficult, particularly among survivors. 10 For this reason, AFE is managed similarly to anaphylaxis.
The risk factors of AFE include pregnancy after the age of 35 years, cesarean delivery, placenta previa or abruption, preeclampsia, and multiple pregnancies. 1 However, regardless of the presence of these risk factors, predicting AFE is challenging. Therefore, AFE must be considered in the differential diagnosis for pregnant patients or recent postpartum patients with sudden cardiopulmonary collapse.
AFE can occur at any point, including during labor or delivery, immediately postpartum, or within 48 hours postpartum. A previous report showed that AFE occurred after miscarriage, abortion, amnioinfusion during delivery, amniocentesis, abdominal blunt injury, surgical trauma, and removal of cervical cerclage, irrespective of labor. 11 The typical symptoms of AFE include acute cardiopulmonary collapse, hypotension, arrhythmia, cyanosis, dyspnea, pulmonary edema, and adult respiratory distress syndrome. Among these symptoms, hypotension, respiratory distress syndrome, and cyanosis are the most common symptoms that occur in nearly all patients. Maternal death is generally caused by bleeding due to cardiac arrest, coagulopathy, respiratory distress syndrome, or multiple organ failure that suddenly recurs after the patient survives once from acute onset of symptoms. 1 DIC is present in 80% of all patients with AFE 9 and is generally caused by hemorrhagic complications. AFE is diagnosed on the basis of clinical symptoms, including the classic triad of severe hypoxic respiratory failure, coagulopathy, and cardiovascular collapse accompanied by bilateral pleural effusion before or after labor. In cardiovascular collapse with coagulopathy, subclinical or nonspecific symptoms, such as anxiety, agitation, restlessness, chest pain, feelings of distress, and shortness of breath, may also be present. 4 To ensure early diagnosis, physicians should always be aware of the possibility of AFE and consistently suspecting it. Maternal prognosis could be substantially improved with rapid detection of symptoms and prompt resuscitation.
In the present case, the patient was a 39-year-old woman, and was thus considered to have a high-risk pregnancy. The patient displayed a mental change in consciousness (stupor) with generalized tonic-clonic seizures, which consequently induced hypotension, respiratory failure, and cyanosis. Blood work that was performed at the emergency center indicated severe DIC and lactic acidosis. Moreover, an initial chest X-ray radiograph showed bilateral pleural effusion, all of which are typical symptoms of AFE.
Conservative treatment, such as oxygen saturation, cardiac output, maintenance of BP, and correction of coagulopathy, should be the primary approach for AFE. Laboratory investigations should be repeated several times during the first few hours because coagulopathy can occur in the early stage. 12 If possible, treatment should be performed at the ICU, and immediate CPR should be performed immediately upon cardiac arrest. Treatment at the ICU is important for postoperative management of left heart failure, adult respiratory distress syndrome, and DIC. Before delivery, emergency cesarean section must be considered to enhance the prognosis of the mother and fetus because an enlarged uterus with a full-term fetus can cause aortocaval compression. This in turn obstructs venous return to the heart and exacerbates hypotension.
If a patient with AFE loses consciousness or enters a hypoxic state, ventilation should be performed with a 100% oxygen mask, immediately followed by intubation and mechanical ventilation. If possible, a central venous catheter should be placed, and volume resuscitation, vasopressors, and inotropic support must be used for cardiovascular support. If a pulmonary artery catheter is inserted, cardiac output, central venous pressure, pulmonary artery wedge pressure, systemic vascular resistance, and pulmonary artery pressure can be measured and used as the criteria for hemodynamic management. Insertion of this catheter also makes arterial blood gas analysis easier. Further, transthoracic echocardiography is useful for examining cardiac function. If the patient shows severe hypotension or severely reduced cardiac function, dopamine or dobutamine should be administered. Prevention and treatment of coagulopathy require rapid and continuous administration of FFP, PC, and cryoprecipitate. Additionally, in a poor hemostatic state, timely initiation of a massive RBC transfusion protocol is crucial. We summarized published case reports on AFE into a tabular form (Table 1).4,5,12–16
Seven case reports of amniotic fluid embolism that complicated medical termination of pregnancy.

HR: heart rate (beats/minute), BP: blood pressure (mmHg), C/Sec: cesarean section, ECMO: extracorporeal membrane oxygenation, M: mother, F: fetus, DIC: disseminated intravascular coagulation, CPR: cardiopulmonary resuscitation, IABP: intra-aortic balloon pump, PT: prothrombin time (seconds), INR: international normalized ratio, Fib: fibrinogen (g/L), ATIII: antithrombin III, RBC: red blood cell, FFP: fresh frozen plasma, PC: platelet concentration, IV: intravenous, SBP: systolic blood pressure, Hb: hemoglobin (g/dL), aPTT: activated partial thromboplastin time (seconds), Plt: platelets, CVVHD: continuous veno-venous hemodialysis.
In our case, the patient required more aggressive treatment compared with conservative treatment upon arriving at the emergency center because she already had an altered mental status and cardiovascular collapse, with 85% to 95% oxygen saturation. An emergency cesarean section was performed because the enlarged uterus needed to be emptied to save the fetus in distress while resolving aortocaval compression. For hemodynamic restoration, a central venous catheter was inserted and pre-load was supplemented using crystalloid. Additionally, we included a mixture of various vasopressors. Initial treatment using an inotropic agent was begun by infusing dopamine and dobutamine. Norepinephrine was added to further stabilize BP and PR via positive inotropic and chronotropic action. Despite such measures, systolic BP remained at approximately 80 mmHg. Adding vasopressin at this point recovered systolic BP to be maintained at approximately 110 mmHg.
Algorithms or guidelines for managing anaphylaxis, including use of vasopressin, are still lacking. Hussain et al. 17 reported a case of a 24-year-old woman who developed serious anaphylactic shock during induction of anesthesia for laparoscopic cholecystectomy. The anaphylactic shock in this case was not responsive to epinephrine and to a large amount of the pure alpha-agonists phenylephrine and norepinephrine. However, the patient recovered a normal circulation and BP with administration of an intravenous bolus of 2 units of vasopressin.
ECMO insertion may be considered if a patient does not show improvement and continues to show severe hemodynamic instability, despite aggressive treatment. ECMO is generally used to resuscitate patients in refractory cardiogenic shock. 5 This is a complex technique, which is used as an adjuvant to sustain life in patients who show severe cardiopulmonary collapse, but are considered potentially reversible. 18 Because this technique oxygenates blood outside the body, it obviates the need for gas exchange in the lungs and provides cardiovascular support when required. 18 Indications for ECMO for cardiac support are a low cardiac output with a cardiac index of <2 L/minute/m2, despite administration of appropriate intravenous volume and use of high-dose inotropic agents or hypotension with a systolic BP of ≤90 mmHg. 19 In an emergency situation, femoral access is the most preferred for insertion of veno-arterial ECMO because it is not only less invasive, but also enables the quickest access for ECMO insertion. 19 In the present case, the patient was indicated for ECMO because she showed severe hypotension with systolic pressure dropping to 35 mmHg, despite appropriate intravenous volume with massive blood transfusion and use of high-dose inotropic agents. Therefore, ECMO insertion was immediately performed following emergency cesarean section.
After ECMO insertion, patients need to be examined to determine whether an appropriate volume status is maintained, which can be assessed using urine output. The first 24 to 48 hours after ECMO insertion is considered an oliguric phase, and there is little urine output. This is because the inserted ECMO circuit induces an acute inflammatory reaction, which causes intravascular volume depletion, thereby leading to oliguria and acute tubular necrosis. After 48 hours, the diuretic phase begins and urine output can be measured, which is considered the earliest sign of recovery. If the patient’s urine output cannot be measured even after 48 hours, diuretics need to be administered to reduce edema. If renal failure is anticipated, continuous renal replacement therapy is required. 19
Prolonged ECMO increases the risk of complications, such as hemolysis, renal function impairment, intracranial hemorrhage, sepsis, and anterior compartment syndrome with limb ischemia. 20 Therefore, patients should be weaned from ECMO as early as possible to avoid these complications. Moreover, the activated clotting time must be well controlled between 180 and 240 seconds to successfully maintain ECMO while preventing circuit thrombosis and systemic thromboembolism. Additionally, the use of anticoagulant agents, such as heparin, is essential. However, continuous use of anticoagulant agents while maintaining ECMO is a clinical dilemma because systemic heparinization caused by anticoagulant agents may increase the incidence of hemorrhage in patients with coagulopathy. The incidence of hemorrhage is close to 34% in veno-arterial ECMO, and surgery may be required if severe hemorrhage occurs. 19 In the present case, the patient was transferred to the ICU following ECMO insertion, and the patient was thought to be maintaining an appropriate volume status on the basis of a urine output of 60 cc or higher per hour from 30 hours after insertion of ECMO. However, on POD 3, acute bleeding of approximately 150 cc/hour was observed, and evacuation of the hematoma, suturing, and electrocauterization were performed. This situation was thought to be due to poor hemostasis as a result of the use of heparin. There are few case reports on the use of ECMO for treatment for AFE.5,21 Once anaphylactic shock occurs, promptly achieving maternal cardiopulmonary stabilization is the primary goal. Hsieh et al. 5 and Reyftmann et al. 21 successfully managed seriously ill patients with ECMO, and these patients were able to be weaned from ECMO after 40 hours and 150 hours, respectively.
Acute compartment syndrome can occur as a result of limb ischemia or reperfusion while ECMO is inserted. The anterior compartment of the thigh is enclosed by a rigid wall, and when pressure in the anterior compartment exceeds intra-arterial pressure, it may cause venous stasis and arterial occlusion. This then leads to muscle swelling and necrosis of muscles and nerves, ultimately causing limb ischemia. 22 The severity of nerve damage can vary depending on the duration of ischemia and the degree of occlusion. More than 30% of adults with a history of ECMO treatment develop neurological complications. 23 The most commonly known complication is sensory deficits in the right lateral cutaneous nerve of the thigh. 24 This may be relevant to our patient because she was cannulated through the right femoral vein.
In our case, ultrasonic-guided ECMO insertion was performed through the right femoral artery and vein, and catheters were placed in the arterial cannula and the venous cannula,. Heparin was used for anticoagulation. The patient was weaned from ECMO after 5 days because vital signs returned to normal. However, ECMO-induced complications, including muscle weakness and foot drop symptoms, were observed from the right leg where ECMO was inserted. On the basis of rehabilitation consultation and results of a strength test, bilateral lumbosacral plexus injury was suspected, and the right lumbosacral plexus injury appeared more severe than the left. The patient was carefully observed because the complication of nerve injury from ECMO insertion is rare. Pain management and rehabilitation through personal training were emphasized. The patient was able to walk with the help of a quad cane, and the motor score for the left lower extremity improved to grade 4. However, the motor grade still remains at 0 for the right ankle and foot and complications are still present at the time of writing this report.
In conclusion, AFE is an unpredictable, life-threatening disease. Although the prognosis of AFE is poor because of acute cardiac arrest and severe DIC, patients with AFE may recover in a few days with appropriate initial treatment. Aggressive treatment, such as ECMO, plays a pivotal role in saving the lives of the mother and newborn. Prompt ECMO insertion can be considered as an appropriate treatment before organ failure occurs in patients with AFE who do not respond to conservative treatment. Further, an expert team, including an obstetrician, anesthesiologist, and thoracic surgeon, should work together to manage AFE and lower ECMO-related complications to achieve the best prognosis in terms of reduced mortality.
Supplemental Material
IMR903640 Supplemental Material - Supplemental material for Successful resuscitation by using extracorporeal membrane oxygenation in a patient with amniotic fluid embolism: a case report
Supplemental material, IMR903640 Supplemental Material for Successful resuscitation by using extracorporeal membrane oxygenation in a patient with amniotic fluid embolism: a case report by Jae Won Kim, Jin Hwan Kim, Tae Woo Kim, Keon Hee Ryu, Sun Gyoo Park, Chang Young Jeong, Jin Ho Choi and Dong Ho Park in Journal of International Medical Research
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
