Abstract
Objective
Several investigations have shown that the level of fibrinogen (Fg) is associated with the susceptibility to dementia and is usually affected by polymorphisms in its gene. Our study aimed to investigate the relationship between Fg level and the fibrinogen β (
Methods
The study included 64 AD patients, 72 VaD patients, and 56 healthy controls (HC). Plasma Fg level was detected by turbidimetry. Among the participants, 64 AD patients, 52 VaD patients, and 56 HC underwent
Results
Plasma Fg was significantly higher in AD and VaD groups than in HC. The AD and VaD patients with
Conclusions
Plasma Fg level was significantly correlated with AD and VaD and related to the −148C/T polymorphism of
Introduction
As the global population ages, the incidence of dementia is increasing each year.
1
Dementia seriously affects patients’ quality of life and has become a focus of domestic and international research. Alzheimer’s disease (AD) and vascular dementia (VaD) are the two most common types of dementia.
2
Previous epidemiological, molecular biology, pathology, and imaging studies have provided evidence indicating that vascular risk factors play an important role in the pathogenesis of AD and VaD. People with higher fibrinogen (Fg) levels often have an increased risk of cardiovascular disease and stroke. Fibrinogen, as a vascular risk factor, can be used as a biomarker for ischemic atherothrombotic stroke, coronary heart disease, peripheral arterial disease, and atherosclerosis.3–7 Evidence from a wide range of sources has suggested that people with higher Fg levels also have an increased risk of dementia,8,9 and Fg level is positively correlated with the risk of AD and VaD.
8
Therefore, Fg, as a common vascular risk factor, may contribute to VaD and AD, and both forms of dementia seem to have similar pathogenesis.10,11 In addition, polymorphisms in the fibrinogen β-chain gene (
VaD is often characterized by vascular pathology, a procoagulant state, and chronic inflammation. The mechanisms underlying these abnormalities in AD are not clear. 13 The level of Fg is significantly higher in AD patients compared with controls in experimental animal models. 14 Depletion of factor XII (FXII) in AD mice inhibits the cleavage of high-molecular-weight kininogen in plasma, resulting in reduced neuroinflammation, fibrinogen deposition, and neurodegeneration in the brain. Moreover, FXII-depleted AD mice show better cognitive function than untreated AD mice, which indicates that Fg is involved in the occurrence of AD. 15 A significant association between the level of Fg and VaD has been found. 16 Besides its prothrombotic effect, Fg seems to have other destructive roles in developing microvascular dysfunctions, including changes in vascular reactivity and permeability. Increased permeability of brain microvessels has a profound effect because it may lead to cerebrovascular remodeling and result in memory reduction. 17 Therefore, Fg may be a risk factor for both AD and VaD. The purpose of this study was to evaluate the relationship between Fg, and its regulatory gene, with AD and VaD.
Materials and methods
Patients
A total of 136 patients, including 64 AD patients and 72 VaD patients, were recruited from the Neurological Outpatient Department in our hospital from December 2005 to December 2007 and enrolled in the study. In addition, 56 healthy controls (HC) without cerebrovascular disease or cognitive dysfunction were randomly selected during the same period. Table 1 shows the basic information of the enrolled patients and HC. Among the participants, 172 (64 AD patients, 52 VaD patients, and 56 HC) underwent
General information of the enrolled subjects.

AD, Alzheimer’s disease; VaD, vascular dementia; HC, healthy controls; MMSE, mini-mental state examination; CDR, Clinical Dementia Rating scale; CDT, clock drawing test.
The study was approved by the Ethics Committee for Medical Research at Tianjin Medical University General Hospital. All patients agreed to provide blood for genetic testing and signed written informed consent.
The inclusion criteria were as follows: (1) patients who completed a detailed clinical questionnaire on histories of cerebrovascular diseases, hypertension, diabetes, coronary heart disease, smoking, drinking, family history, eating habits; and (2) underwent a physical examination (comprehensive neurological examination) combined with clinical application to mini-mental state examination (MMSE) and clock drawing test (CDT) to screen those with cognitive dysfunction. Patients with dementia had an MMSE score between 4 and 24. Severity of dementia was determined by the Clinical Dementia Rating (CDR) scale. The diagnostic criteria for AD were in line with the NINCDS-ADRDA criteria and DSM-IV-R criteria. The diagnostic criteria for VaD5,18 included the State of California Alzheimer’s Disease Diagnostic and Treatment Centers criteria for probable and possible ischemic vascular dementia, the International Classification of Diseases, Tenth Edition (ICD-10) criteria for VaD, the National Institute of Neurological Disorders and Stroke, and the Association Internationale pour la Recherche et l’Enseignement en Neurosciences criteria for probable and possible VaD. All these criteria were in accordance with the VaD diagnostic criteria of the Chinese Medical Association Branch of Neurology.
Patients with cerebrovascular disease, other neurodegenerative dementia, normotensive hydrocephalus, epilepsy, infection, depression, systemic metabolic disease, family history of dementia, undernourished patients, and patients who did not cooperate with blood collection were excluded. All participants underwent brain examination and were excluded if they had history of head injury; tumor; liver or kidney dysfunction; thyroid deficiency; chronic malnutrition; recent use of folic acid and B vitamins; chronic alcoholism; substance abuse; disturbance of consciousness; mental disorders (Hamilton Depression Rating Scale ≥15); or other diseases that can cause dementia.
Methods
Plasma fibrinogen was measured using the Clauss fibrinogen assay. In brief, 2 mL of venous blood was withdrawn from the cubital vein of participants in the early morning. The blood was placed in a tube containing 0.2 mL of sodium citrate for 1 hour, centrifuged at 1006.2 ×
For
Statistical methods
Statistical analysis was performed using SPSS version 11.5 (IBM Corp., Armonk, NY, USA). Genotype frequencies were estimated by gene counting. The genotype distributions in all groups were compatible with Hardy–Weinberg expectations. Measurement data were presented as mean ± standard deviations and compared using Student’s
Results
Compared with individuals in the HC group, AD and VaD patients showed significant differences in MMSE, CDR, and CDT scores (
The plasma Fg levels in AD, VaD, and HC groups were 298.82 ± 50.90, 317.97 ± 97.19, and 259.71 ± 48.20 mg/dL (Figure 1), respectively. Regression analysis showed that after controlling for age, sex, diabetes, hypertension, smoking, drinking, cholesterol, diet, coronary heart disease, gene, and other factors, plasma Fg levels were significantly different among the three groups (

Plasma Fg levels in AD, VaD, and HC groups. Fg, fibrinogen; AD, Alzheimer’s disease; VaD, vascular dementia; HC, healthy controls. The lower and upper ends of the box mark the 25th and 75th percentiles or the location of the first and third quartiles, and the solid band indicates the 50th percentile or the median. The whiskers represent the range of values.
Results of regression analysis.

AD, Alzheimer’s disease; VaD, vascular dementia; HC, healthy controls; TC, total cholesterol; CAD, coronary heart disease.
Analysis of CDR, MMSE, and Fg levels of patients showed no correlation in the dementia group (patients with AD and VaD) (Table 3).
The correlation between CDR, MMSE, and Fg level in AD and VaD groups.

CDR, Clinical Dementia Rating scale; MMSE, mini-mental state examination; AD, Alzheimer’s disease; VaD, vascular dementia; Fg, fibrinogen.
Amplification of the
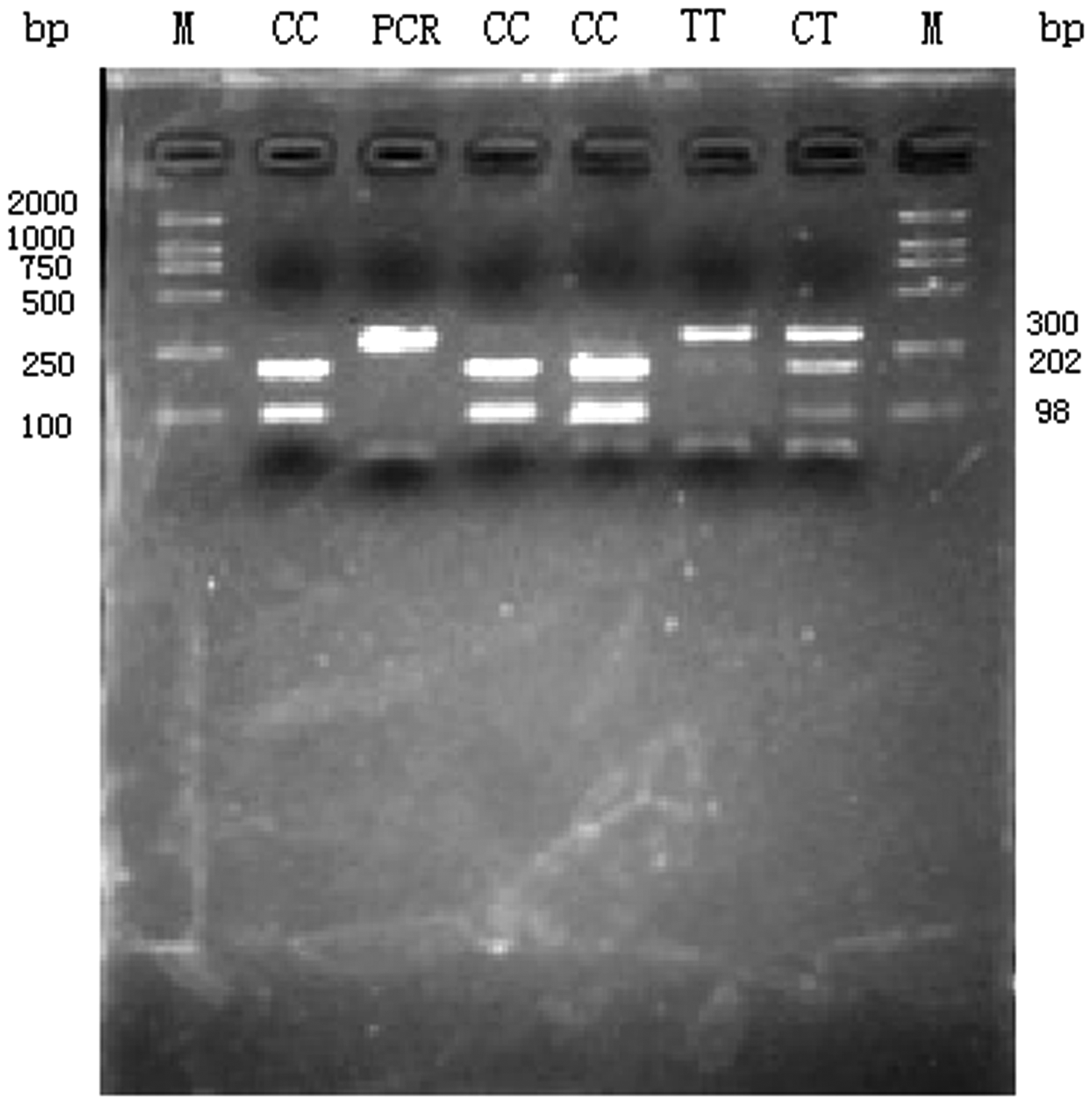
Agarose gel analysis of amplification products of

Sequence diagram of TT genotype of

Sequence diagram of CC genotype of
Figure 5 shows the frequency distribution of

Distribution of
Comparison of frequencies of CC/CT+TT genotypes and C/T allele between different groups (χ2,

AD, Alzheimer’s disease; VaD, vascular dementia; HC, healthy controls.
Figure 6 shows that Fg levels of the three different genotype carriers had homogeneity of variance in the AD and VaD groups (

Comparison of gene polymorphism of
Discussion
Evidence suggests that vascular risk factors play important roles in the development of dementia, and plasma fibrinogen is associated with VaD. 16 AD is the most common form of dementia and there is no effective treatment for AD. In addition to pathologic characteristics, AD also has a vascular component. Substantial evidence has shown that thrombosis and fibrinogen play an important role in AD.19–21 Fourteen days after a stroke, the level of Fg gradually approaches that of healthy subjects. 22 In all of the patients involved in this study, the time elapsed since stroke was more than 3 months. Therefore, the increase in Fg caused by stroke itself had little impact on results in this study. 22 Our results showed that plasma Fg level was significantly higher in the AD and VaD groups than in the HC group. It was not significantly different between patients in the AD group and those in the VaD group. This finding indicated that a high plasma Fg level is strongly related to the onset of dementia. 23 Chung et al. 24 compared Fg levels in 86 patients diagnosed with stroke and their T2-weighted magnetic resonance images (MRI), and found that high Fg level was related to the severity of white matter damage. A study in Austria also suggested that high Fg level was independently associated with white matter density and lacunar infarcts in head MRI. 25 We hypothesized that high plasma Fg level may play a common role in the occurrence of AD and VaD as a vascular risk factor.
The biosynthesis of Fg is influenced by genetic factors and various environmental factors.26,27 Humphries et al.
28
first reported a correlation between Fg gene polymorphisms and Fg level in 1987. Schmidt et al.
29
studied 399 individuals aged 45 to 75 years (Austrian Stroke Prevention Study) by computed tomography and showed that subjects with the TT genotype had a 6.17-fold higher risk of carotid artery atherosclerosis than subjects with the CC or CT genotype. They inferred that the
The mechanism by which the
In this study, the plasma Fg level was significantly higher in the AD and VaD groups than in the HC group. Within the dementia group (AD and VaD), patients with the TT genotype at
This study showed that Fg, as a risk factor for dementia, may participate in the pathogenesis of dementia through its vascular mechanism. An increased level of circulating Fg indicates poorer subsequent cognitive ability and is associated with age-related cognitive decline in several domains, including general ability.
38
The
Conclusions
Plasma Fg level was significantly correlated with AD and VaD but not with the severity of dementia. Plasma Fg level was associated with the polymorphism of
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The study was supported by the Special Research Fund for Central Universities, Peking Union Medical College (No. 3332018181), Tianjin Natural Science Foundation (No. 19JCYBJC26100); The Science &Technology Development Fund of Tianjin Education Commission for Higher Education (No. 2017ZD10).
