Abstract
Objective
The purpose of this study was to systematically evaluate the association between methyl-CpG binding domain 4, DNA glycosylase (MBD4) Glu346Lys polymorphism and cancer risk.
Methods
A comprehensive document retrieval from the Chinese National Knowledge Infrastructure (CNKI), EMBASE, and PubMed databases was performed through 1 September 2019. The strength of the correlation was assessed using the pooled odds ratio (ORs) and 95% confidence interval (CIs).
Results
Five relevant studies were retrieved following screening, including 1804 cases and 2193 controls. We found no association between MBD4 Glu346Lys polymorphism and cancer risk under all genetic models. Nevertheless, a subgroup analysis based on country showed a strong association in the Chinese population. Under the recessive model, Chinese individuals with the Lys/Lys genotype had a higher risk of cancer (OR = 1.37, 95% CI = 1.11–1.70).
Conclusion
Analysis of the MBD4 Glu346Lys polymorphism in different populations will help to elucidate the pathogenesis of cancer. The polymorphism can be utilized as a biomarker for cancer susceptibility among Chinese people.
Introduction
The human MBD4 gene is located on chromosome 3q21.3 and encodes a nuclear protein that binds to a methyl-CpG domain at the N-terminal and a mismatch-specific glycosylase domain at the C-terminal. 1 The N-terminal domain functions both in specifically binding to methylated DNA and in interacting with co-repressors to repress transcription from methylated gene promoters. The C-terminal domain shares homology with a base excision repair protein and is involved in DNA mismatch repair. MBD4 can remove T and U from G:T and G:U mispairings in CpG methylation regions, which are caused by spontaneous deamination of 5-methylcytosine (5mC) and cytosine (C), respectively.2–4 Based on these functions, MBD4 is of great importance in maintaining genomic stability. Furthermore, MBD4 is involved in several cellular processes such as cell cycle arrest, apoptosis, and protection against oxidative stress.5,6 Inactivation of MBD4 could modify a mismatch repair–deficient cancer phenotype and thus contribute to tumorigenesis. 7 These results suggest that MBD4 is a potential tumor suppressor.
Several studies have concentrated on the role of the MBD4 Glu346Lys polymorphism (rs140693 G>A) in the risk of cancers, including esophageal squamous cell carcinoma (ESCC) and cervical, gastric, lung, and colorectal cancers.8–12 However, the results of these studies are inconclusive and require further clarification. Hence, we conducted a meta-analysis of published articles, which will help researchers more precisely understand the status of the MBD4 polymorphism in cancer risk.
Methods
Literature search
A comprehensive literature search based on the Chinese National Knowledge Infrastructure (CNKI), EMBASE, and PubMed databases was performed through 1 September 2019. To discover all eligible studies assessing the association between the polymorphism of MBD4 Glu346Lys (rs140693 G>A) and the risk of cancer, we used the following keywords: “methyl-CpG binding domain protein 4 or MBD4”, “SNP or variant or polymorphism or rs140693” and “tumor or carcinoma or cancer”. Furthermore, we screened the reference lists of both primary articles and reviews to identify additional eligible studies. At the same time, we implemented the following inclusion criteria to obtain eligible studies: (1) studies on the polymorphism of MBD4 Glu346Lys and the risk of cancer; (2) case-control studies; and (3) studies containing sufficient data on genotype distribution. Exclusion criteria were (1) case-only studies; (2) studies lacking sufficient data; or (3) editorial comment and review.
Data extraction
Two researchers independently extracted the available information from each qualified article. The following data were analyzed and gathered: name of the first author, year of publication, country of origin, type of cancer, numbers of cases and controls, genotype counts of cases and controls, and the P-value for Hardy–Weinberg equilibrium (HWE) in control subjects. If there were disagreements, agreement was reached through discussion and consultation. In addition, the quality of each included study was analyzed according to quality assessment criteria. 13 The quality assessment score ranged from 0 to 15; studies with scores <9 were categorized as low quality and those with scores ≥9 as high quality.
Statistical analysis
The strength of the associations between MBD4 Glu346Lys polymorphism and cancer risk was assessed by calculating the odds ratio (ORs) and 95% confidence interval (CIs). In addition, heterogeneity was estimated based on the Q test and I2. A P-value for heterogeneity (PH) > 0.1 indicated that heterogeneity was absent, in which case, a fixed-effects model was applied. Otherwise, a random-effects model was used. The HWE in control subjects was calculated by Chi-squared test; PHWE < 0.05 suggested that genotype distribution in control subjects deviated from HWE. To determine the influence of each excluded study on the overall OR value, we performed a sensitivity analysis. Publication bias was assessed using funnel plots. All data were analyzed using Review Manager software (version 5.2; Cochrane Collaboration, London, UK).
Results
Main information of included studies
The literature retrieval identified 30 potentially relevant studies. After reading the titles and abstracts, 25 studies were excluded because they were irrelevant to the present topic. Subsequently, the full text of the remaining five studies was reviewed and all were finally included.8–12 The five studies in this meta-analysis included 1804 cases and 2193 controls. The main features of the included articles are listed in Table 1. The year of publication ranged from 2004 to 2012. The genotype distribution in control subjects of included studies conformed to HWE (PHWE > 0.05). Among the included studies, one was focused on cervical cancer, one on gastric and colorectal cancer, one on ESCC, and two on lung cancer. In addition, three of the studies were from China and two from Korea.
Main characteristics of the studies included in the meta-analysis.

PHWE, P-value for Hardy–Weinberg equilibrium in controls; ESCC, esophageal squamous cell carcinoma.
Meta-analysis
The results of the meta-analysis are presented in Table 2, and a representative forest plot is shown in Figure 1. According to the data, the MBD4 Glu346Lys polymorphism was not associated with the risk of cancer under all genetic models. However, a subgroup analysis based on country showed a strong association between MBD4 Glu346Lys and the risk of cancer in the Chinese population. Chinese individuals with the Lys/Lys genotype had a higher risk of cancer under the recessive model (OR = 1.37, 95% CI = 1.11–1.70, P-value for z-test, PZ = 0.004).
Meta-analysis of the association between MBD4 Glu346Lys polymorphism and cancer risk.
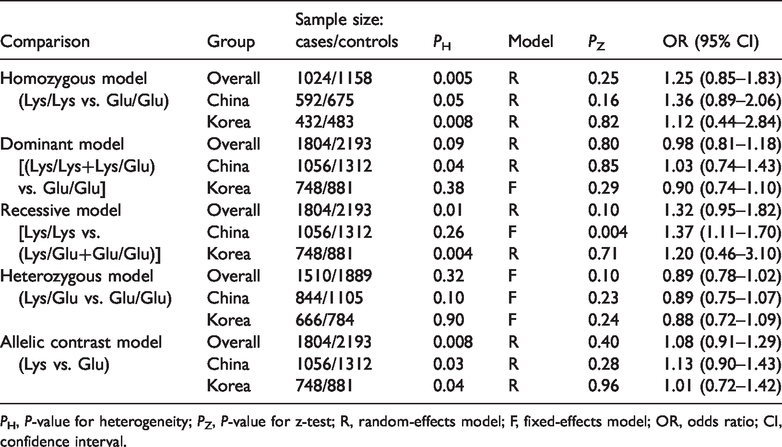
PH, P-value for heterogeneity; PZ, P-value for z-test; R, random-effects model; F, fixed-effects model; OR, odds ratio; CI, confidence interval.

Forest plot of the association between MBD4 Glu346Lys polymorphism and cancer risk under homozygous model in a meta-analysis of five studies. CI, confidence interval; M-H, Mantel–Haenszel method.
Sensitivity analysis and publication bias
After removing the study of Shin et al., 9 we observed a significant association between MBD4 Glu346Lys polymorphism and cancer risk under the homozygous model (Lys/Lys vs. Glu/Glu, OR = 1.44, 95% CI = 1.01–2.04, PZ = 0.04) (Figure 2a) and the recessive model [Lys/Lys vs. (Lys/Glu+Glu/Glu), OR = 1.44, 95% CI =1.18–1.76, PZ = 0.0003] (Figure 2b). After removing the study of Hao et al., 8 we observed a significant association under the heterozygous model (Lys/Glu vs. Glu/Glu, OR =0.85, 95% CI = 0.73–0.99, PZ = 0.04) (Figure 2c). Potential publication bias was assessed using funnel plots and the results are shown in Figure 3. Studies in the funnel plots were distributed symmetrically under all genetic models, indicating no publication bias in the overall meta-analysis.

Sensitivity analysis of the association between MBD4 Glu346Lys polymorphism and cancer risk in a meta-analysis of five studies. CI, confidence interval; M-H, Mantel–Haenszel method.

Funnel plots of the association between MBD4 Glu346Lys polymorphism and cancer risk under different models. OR, odds ratio.
Discussion
Methyl-CpG binding domain protein 4 (MBD4) plays an important role in the development of cancer, and the Glu346Lys polymorphism in MBD4 may be implicated in the risk of cancer. In 2004, Hao et al. first investigated the association of MBD4 Glu346Lys polymorphism with ESCC, and found that genotype distribution of the MBD4 Glu346Lys polymorphism in ESCC patients differed from that in the control group; subjects with the Lys/Lys genotype had a dramatically increased risk of developing ESCC compared with those with the Glu/Glu genotype. 8 Subsequently, Xiong et al. obtained similar results for cervical cancer. 12 In contrast, the study of Shin et al. found that compared with the Glu346Glu genotype, the Lys346Lys genotype of MBD4 was related to a highly reduced risk of lung adenocarcinoma. 9 The study of Miao et al. revealed that the Glu/Lys genotype of MBD4 Glu346Lys was strongly associated with a decreased risk of lung cancer compared with the Glu/Glu wild-type. 10 In addition, the study of Song et al. indicated that MBD4 Glu346Lys was involved in the risk of colorectal cancer but not gastric cancer. 11 These inconsistent results might be attributed to limited sample sizes, different study designs, or potential sampling bias in single-center studies, and must be clarified by using meta-analysis.
Based on published data, we analyzed all relevant data to evaluate the relationship between the MBD4 Glu346Lys polymorphism and the risk of cancer, including 1804 cases and 2193 controls from five studies. The overall results suggested that under all genetic models, the MBD4 Glu346Lys polymorphism was not associated with the risk of cancer, but subgroup analysis based on country showed a significant association between MBD4 Glu346Lys polymorphism and cancer risk in the Chinese population. Chinese individuals with the Lys/Lys genotype were more likely to develop cervical cancer, lung cancer, and ESCC based on data analysis under the recessive model. We speculated that under a certain genetic background, the polymorphism could affect MBD4 protein activity by changing the spatial structure of the protein. According to our information, this is the first meta-analysis on the association of the MBD4 polymorphism with cancer risk, and our results expand the understanding of the role of genetic variants in the onset of cancer. However, there are some limitations in this study that should be addressed. First, due to limited data, other confounding factors that might affect cancer risk were not considered in the meta-analysis. Second, the sensitivity analysis showed that our results were not sufficiently robust, which might be attributed to the small sample size and the limited number of included studies. Our meta-analysis included two studies in a Korean population and three studies in a Chinese population, which increased the sensitivity of removing just one study and introducing bias based on country selection. Hence, further research and analysis is still required.
In summary, our results indicate that MBD4 Glu346Lys polymorphism is associated with cancer risk in a Chinese population, and this polymorphism could be used as a biomarker for cancer susceptibility. However, due to the limitations described above, our results should be interpreted cautiously.
