Abstract
Chronic inflammation and bone defects after total knee arthroplasty are a challenge for the orthopedic surgeon. There have been few reports on application of a three-dimensional (3D) printed porous tantalum prosthesis in such situations. We report an 83-year-old female patient who presented to our clinic with consistent pain of the left knee for 10 years and a severe decline in mobility for 2 years. Chronic inflammation, loosening of a tibial prosthesis with a bone defect, and severe osteoporosis were diagnosed. The patient was treated with computer designed and manufactured, personalized, 3D printed porous pure tantalum pad-assisted left total knee arthroplasty. The surgery went smoothly and the patient achieved a satisfactory recovery after surgery. A 3D printed porous tantalum prosthesis can be used to reconstruct tibial bone defects in patients with chronic inflammation after joint replacement surgeries.
Introduction
Total knee arthroplasty (TKA) has been one of the most successful surgical procedures in the last century. TKA can completely relieve pain of the knee joint, promote maximum recovery of knee function, and restore self-care ability of patients. Therefore, TKA is widely used in clinical practice.1,2 However, TKA can have devastating complications. Infection after TKS is a destructive complication after TKA. TKA causes pain, loss of mobility and increases the cost of treatment. At present, there are many treatment options for infection after TKA, among which revision surgery is the most widely used and has the best effect. 3 Treatment methods of patients with revision after TKA mainly include one- and two-stage revision. One-stage revision is generally used for treating non-infected patients. However, one-stage revision does not provide effective debridement to remove infectious pathogens, and infection is difficult to cure and easily relapses. 4 At present, two-stage revision is the most accepted method of revision for infection after TKS. However, the long interval between the first and second revision surgeries can lead to extensive scarring, disuse atrophy of the bone, and contracture of joint capsule soft tissues, which hinder effective recovery of joint function. 5
In recent years, emergence of advanced implants made of novel materials and manufacturing techniques have made one-stage revision surgery safer and more effective. 6 However, reports on three-dimensional (3D) printed tantalum prostheses in revision surgery of TKA are rare. 7 We report here a patient with inflammation who had a 3D printed porous tantalum prosthesis to reconstruct a tibial bone defect after TKA in a one-stage surgery.
Case report
The study was approved by the ethics committee of the Southwest Hospital of China (registration number: SWH2016ZDCX-2010). All of the procedures were carried out according to the Declaration of Helsinki. Written consent was obtained from the patient to participate in the study and for use of her tissues for further studies. A statement of consent for publication was also obtained from the patient.
An 83-year-old female patient presented to our clinic with the complaint of consistent pain of the left knee for 10 years and a severe decline in mobility for 2 years. The patient suffered from swelling and pain of the left knee joint after accidentally sliding down a ladder 16 years ago. There was no fever, swelling, night sweating, or other discomforts at this time. The pain in the left knee progressed over the years, and the patient was treated for deformity of the left knee secondary to traumatic arthritis with TKA 9 years previously. In this surgery, the patient received a model C femoral prosthesis (Zimmer, Warsaw, IN, USA), Number 2 proximal tibia metal pad with a pad thickness of 12 mm (Zimmer), and no patella prosthesis. Five years previously, the patient suffered from a right femoral intertrochanteric fracture due to a fall and received closed reduction and percutaneous compression plate internal fixation.
Four years previously, the patient started to feel pain in the left knee after walking for more than a kilometer. The pain in the left knee joint was relieved after rest. Furthermore, the patient felt no joint swelling, elevated skin temperature, fear of cold and fever, joint clicking and interlocking, or night convulsions. The patient suffered from a left femoral intertrochanteric fracture after a fall 3 years previously, and was treated with closed reduction and proximal anti-rotation intramedullary nail internal fixation. Two years previously, she started to feel slight collapse of the left knee joint without obvious inducement. This gradually worsened and her mobility further declined. The patient came to our clinic after no pain killers could alleviate her pain in the left knee.
Physical examinations showed normal physiological curvature of the spine, no deformity of both upper limbs, varus deformity of both lower limbs, shortening of the left lower limb by approximately 1 cm compared with the right lower limb, and no obvious swelling of the left and right hips Additionally, there was no obvious atrophy of both quadriceps femoris muscles, slightly weakened muscle strength, and slight swelling of the left knee joint. A patella buoyancy test was negative, a patella grinding test was positive, hyperextension and hyperflexion were positive, lateral patellar tenderness was positive, medial joint space tenderness was positive, and a lateral stress test was negative. The left knee range of motion (flexion and extension) was 15 to 90 degrees. Skin sensation of both lower limbs was normal, distal blood supply, sensation, and activity were normal, and dorsal pedal artery pulsation was normal. No obvious abnormalities were found in other physical examinations.
An X-ray showed that internal fixation was in place after bilateral femoral neck and proximal femur internal fixation. Additionally, there was a tilted left tibial plateau, osteoarthritis of the right knee, and changes in varus alignment in both knees (Figure 1). Three-dimensional computed tomography (CT) imaging showed that after left TKA, the longitudinal line of the artificial tibial plateau was slightly angled with the longitudinal axis of the tibia, and the proximal tibial bone was discontinuous with irregular margins (Figure 2).
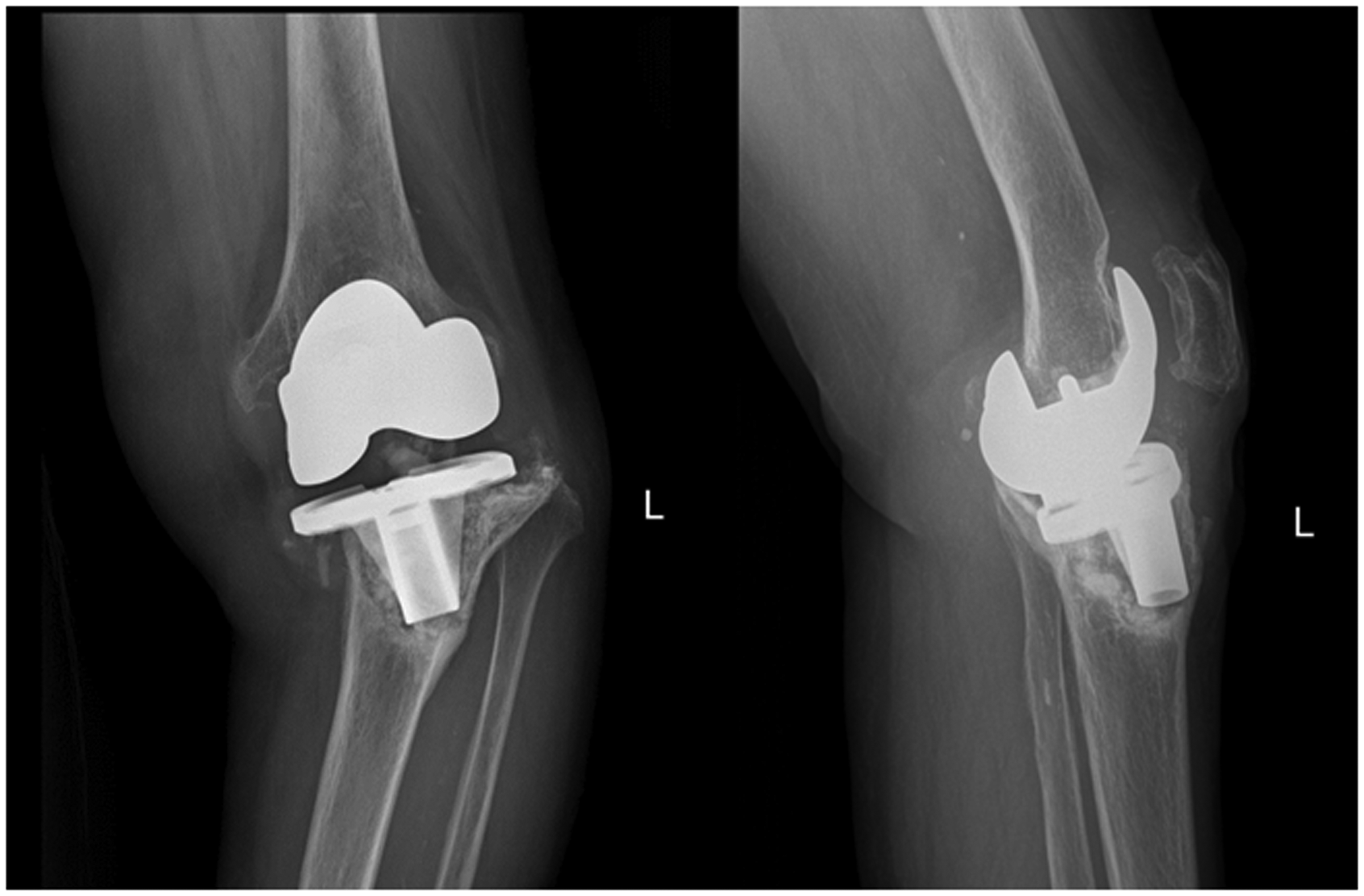
An X-ray shows a tilted left tibial plateau and a change in varus alignment in the left knee.
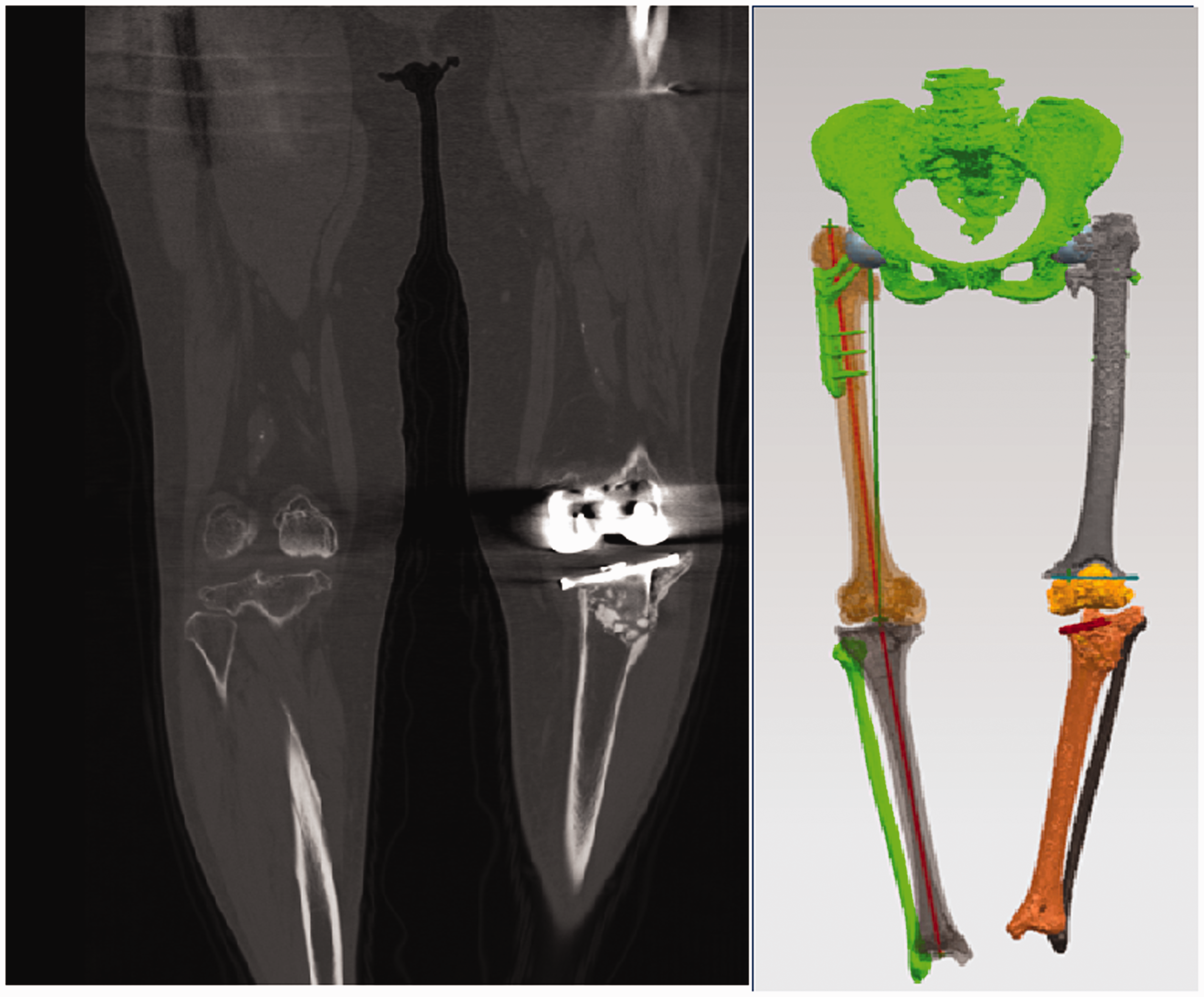
Three-dimensional computed tomography imaging shows that after left TKA, the longitudinal line of the artificial tibial plateau is slightly angled with the longitudinal axis of the tibia, and the proximal tibial bone is discontinuous with irregular margins.
The diagnoses of chronic inflammation, loosening of the tibial prosthesis with a bone defect, and severe osteoporosis were considered and the patient was admitted to our hospital. After admission, no major abnormalities were found in blood and urine routine tests, a liver function test, a coagulation function test, chest X-ray, electrocardiogram, and other preoperative examinations. Cardiovascular and respiratory medicine consultation was requested to exclude surgical contraindications.
The patient’s distal femoral and proximal tibial bones were scanned by a CT scanner (Siemens Medical Solutions, Forchheim, Germany) and the scan data from the Picture Archiving and Communication System were reconstructed by Mimics 17.0 software (Materialise NV, Leuven, Belgium). The extent of the lesion was outlined, and preoperative surgical planning was made using a 3D model. Three-dimensional data, including the surgical plans, were stored in STL format and sent to an engineer for implant printing. The implant was designed by an engineer according to surgical plans, bone quality, and the overall patient’s condition, such as weight and height (Figure 3).

Personalized three-dimensional design of the implant according to computed tomography and magnetic resonance imaging scans of the distal femoral and proximal tibial bones.
Finite element (FE) analysis was carried out evaluate the plausibility of biomechanical stress distribution of the implant. In FE analysis, the tibial pad was divided into 262,217 tetrahedral unit materials. Body weight was set at 60 kg, maximum stress of the tibial pad was 2.061 × 10−1 MPa, and maximum displacement of the tibial pad was 3.551 × 10−2 mm. The femoral pad was divided into 29,339 tetrahedral units. The material properties were based on a porus tantalum foam as reported by Zardiackas et al. 8 The boundary condition was fixed at the interface between the pad and the prosthesis, and the load was based on the patient’s weight. Maximum stress of the femoral pad was 6.531 × 10−1 MPa.
To avoid bone resorption and implant loosening from stress shielding, a porous structure was used in the 3D implant (Figure 4). The design process of the 3D printed tantalum was as follows. 1) The patient’s pelvis, left femur, right femur, and left implanted knee prosthesis were reconstructed using X-ray scans. 2) The left femur and tibial prosthesis were scanned by CT, and the 3D shape of the prosthesis was reconstructed inversely. 3) A 3D file (STL format file) of the patient’s pelvis, left femur, left tibia, and left knee prosthetic implant was imported into UG11.0 software (Unigraphics NX, Siemens PLM Software, Plano, TX, USA). 4) The patient’s pelvis and lower limbs were placed in the standing position, the force line of lower limbs was designed to be 180 degrees after surgery, and the STP format file of the left femoral and tibial prosthesis was imported. The posterior tilt angle, and internal and external rotation of the left tibial prosthesis were adjusted to identify the best area to place the prosthesis. 5) A file of the left femoral prosthesis was imported into UG11.0 software. The angles of internal and external rotation and anteversion of the left femur were adjusted, and the 3D position of the left femoral prosthesis was simulated. 6) An extension rod was placed according to the position of the left femoral and tibial prosthesis. The anatomical structure and spatial shape of the 3D printed porous tantalum were designed according to the spatial shape of the bone defect between the prosthesis and the femur and tibia. The density of the implant was 5.17 g/m3, the diameter of each aperture was 700 μm, and the extent of porosity was 68.98%. The muscle and ligament suturing points were designed according to CT and magnetic resonance imaging scans.

Three-dimensional printed implant adjusted with the 1:1 printed model of the patient’s femoral and tibial bones.
After being cleared for surgery, the patient received computer designed and manufactured, personalized, 3D printed porous pure tantalum pad-assisted TKA under combined epidural and lumbar anesthesia. Intraoperatively, synovial hyperplasia with scar formation, clear brown fluid, and large number of wear debris accumulation were observed in the articular cavity. Joint fluid was sent for bacterial culture and synovial tissue for pathological examination. Joint cavity cleaning, patelloplasty, and prosthesis removal were then performed, and knee prosthesis revision was performed (Figure 5). Commercial implants included a tibial component (Zimmer), LCCK left D femoral component (Zimmer), LCCK CD 3-4 12-mm liner (Zimmer), 59 × 30-mm filler/tibial conical filler (Zimmer), and 40 g of two bags of antibiotic-loaded bone cement.

Intraoperative procedure of three-dimensional printed porous tantalum prosthesis implantation to reconstruct a tibial bone defect in our patient who had inflammation after total knee arthroplasty.
The surgery proceeded smoothly, and the patient recovered quickly after the surgery. Her general condition and vital signs were stable by the time of her discharge. A physical examination showed that her left knee joint was slightly swollen, joint flexion and extension activity were approximately 0 to 90 degrees, blood supply, sensation, and activity of the distal lower limbs were normal, and she could walk under a weight load. She was instructed to increase nutritional intake, continue rehabilitation training, and make regular follow-up visits to our clinic. During 12 months of follow-up, the patient gradually recovered mobility and reported no more discomfort of the lower extremities (Figure 6).

Postoperative X-ray of the patient 1 year after the revision surgery and after total knee arthroplasty of the right knee.
Discussion
Infection is one of the most common and serious complications after TKA. In recent years, use of prophylactic antibiotics and continuous improvement of aseptic operation techniques have led to a decrease in the incidence of infection after TKA. However, TKA still remains a major postoperative complication that has a serious effect on some patients. 9
Revision surgery after TKA can cause further bone defects in the distal femur, proximal tibia, and patella. 10 Because revision of artificial joints can cause further bone defects, a prosthesis should have high biocompatibility, promote bone regeneration, and integrate with regenerated bone. A bone defect due to infection after knee arthroplasty is large and irregular, and the prognosis of revision surgery is poor. In cases of severe osteoporosis, a preexisting bone defect, and inflammation, conventional prostheses may not be sufficiently fixed by bone cement and screws, leading to a poor effect of revision.10,11 Traditional materials for reconstruction of those defects include autogenous and allogeneic bone transplants. However, the source of autogenous bone is limited and it results in harvest site morbidity. Allogeneic bone transplants may fail because of different immunogenicities and a lack of strong structural support.12,13 In patients with severe patellar defects, knee joint revision with traditional materials often results in failure.
Three-dimensional printing technology shapes an implant into any shape to precisely match complex bone defects. Micropores on the surface of a 3D printed prosthesis increase the contact area, and enhance the bond strength and initial stability of the inner plant–bone interface. The pore structure of a 3D printed prosthesis is conducive to soft tissue attachment, has good biomechanical stability, promote adhesion and proliferation of osteoblasts, maintain cell morphology, and achieve better bone growth and long-term stability.14,15 Additionally, 3D printed personalized prostheses have a shorter manufacturing cycle than traditional custom prostheses. Therefore, for revision surgeries with severe bone defects, 3D printed prostheses have advantages in terms of precise matching and stability compared with conventional prostheses. 16
Studies have shown that a tantalum prosthesis with high porosity promotes growth of bone tissue and osteointegration, which increases stability of the prosthesis. 17 In patients with inflammation and a severe patellar defect after TKA, a porous tantalum may be the ideal material in these circumstances. One side of the porous tantalum implant is attached and anchored to the remaining patellar bed or ligament, and the other side is combined with polyethylene via bone cement. This porous tantalum patellar prosthesis can be anchored to the patellar ligament to achieve a more stable union. Kamath et al. 18 treated 23 patients with a tantalum prosthesis and achieved significant improvement as shown by Knee Society Clinical Rating System pain and function sores and Oxford Knee scores. Other studies have reported similar results using tantalum scaffolds for knee arthroplasty.19–21
We found that a personalized 3D printed tantalum implant was effective for treating post-TKA inflammation and a bone defect. Although this is a report of only one case, there have been few reports on application of implants printed from tantalum. 7 Cooperation between doctors and engineers should be strengthened to build a normative framework. The results of the current case indicate that, with refinement of 3D printing techniques for joint prostheses and development of biocompatible tantalum alloys, a personalized prosthesis will provide safer and more effective treatment for patients with osteoporosis and postoperative infection.
Conclusion
Three-dimensional printing of a personalized joint prosthesis accurately reconstructs bone defects. The porous structure of this prosthesis reduces the elastic modulus, increases initial stability, induces long-term bone ingrowth, and achieves good follow-up results.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the National Key Research and Development Plan (2016YFB110-14040), Chongqing Municipal/Technology Platform and Base Construction (International Science and Technology Cooperation Base Construction) (cstc2014gjhz110003), Southwest Hospital Managed Project/Clinical Major New Technology Transformation and Breakthrough Plan (SWH2016ZDCX2010), and Southwest Hospital Managed Project/Platform Construction Special Project (SWH2016PTJS-04).
