Abstract
Objectives
To evaluate the Th1/Th2 cell profile in spleens of cirrhotic and hypersplenic rats by investigating the expression of Th1-associated chemokine receptors CXCR3, CCR5 and Th2-associated chemokine receptor CCR3.
Methods
Experimental liver cirrhosis and hypersplenism were induced in rats by the intragastric administration of carbon tetrachloride (CCl4; 40% solution [0.3 ml/100g, twice/week for 8 weeks]) and confirmed by pathology and hemogram. Presence of the three chemokine receptors was investigated by real-time polymerase chain reaction (RT-PCR), immunohistochemical staining, and western blot analysis.
Results
By comparison with control animals (n=10), RT-PCR demonstrated that CXCR3 and CCR5-mRNA levels were significantly elevated in the hypersplenic rats (n=26) and CCR3-mRNA levels were lower. Immunohistochemical staining showed that by comparison with controls, the mean density of the Th1-associated CXCR3 and CCR5 receptors was significantly increased but there was no difference between groups in Th2-associated CCR3 receptors. Western blot analysis showed that by comparison with controls, hypersplenic rats had higher levels of CXCR3 and CCR5 protein but lower levels of CCR3 protein.
Conclusions
The abnormal expression of Th1-associated chemokine receptors in spleens of rats with cirrhosis and hypersplenism induced by CCL4 suggests that a functional imbalance between Th1/Th2 cells may play a role in the pathogenesis of hypersplenism.
Introduction
Hypersplenism, or enlargement of the spleen, is frequently accompanied by peripheral blood cytopenia, particularly thrombocytopenia. 1 Many factors have been implicated in the development of hypersplenism and primarily involve retention in the spleen, phagocytosis and autoimmunity. 2 However, its exact pathogenesis is unclear and further investigations are required to provide better management strategies for the treatment of patients with this condition. 2
As the largest secondary lymphoid organ in the body, the spleen is the main site of T lymphocyte activation. 3 CD4+ T helper cells regulate cellular and humoral immune responses to a wide range of pathogens and are involved in many diseases processes. 4 In addition, the CD4 + T helper cell subsets, Th1 and Th2, stimulate the functional activity of major components of cell-mediated immunity including cytotoxic cells, natural killer cells and activated macrophages. 5 Indeed, an imbalance between Th1 and Th2 is thought to play an important role in the pathogenesis of autoimmune disorders. 5 Interestingly, studies have shown that Th1/Th2 type cytokines of the spleen were imbalanced in patients with hepatic carcinoma combined with liver cirrhosis. 6
Th1 lymphocytes express the chemokine receptors CXCR3 and CCR5 whereas Th2 lymphocytes express CCR3 receptors.7,8 Combined with chemoattractant cytokines, these receptors play a fundamental role in many physiological and pathological processes such as directing and controlling migration activation, differentiation, and trafficking.9,10 The use of the receptors CXCR3, CCR5 and CCR3 as markers to differentiate Th1/Th2 cells from CD4+cells has been reported in studies of autoimmune diseases.7,8 However, the status of these chemokine receptors in the spleens of patients with cirrhosis and hypersplenism remains unknown. The purpose of this present study was to investigate the expression of Th1-associated chemokine receptors CXCR3, CCR5 and Th2-associated chemokine receptor CCR3 in spleens of cirrhotic and hypersplenic rats and evaluate the balance between Th1/Th2 cells.
Materials and methods
Rat model of cirrhosis and hypersplenism
Male Sprague Dawley rats (180–250g) approximately 50 days old were used in the experiments. The rats were housed in laboratory cages in a controlled, pathogen-free environment (25 ± 2°C) and kept under a 12/12 h dark/light cycle with free access to food and water. Prior to the experiments the rats were allowed to adapt to their new environment for one week. All procedures involving laboratory animals were performed in accordance with the guidelines prepared by the Research Centre for Drug Safety Evaluation of Hainan Province and every effort was made to minimize any suffering. The study was approved by the Medical Ethics Committee of Hainan General Hospital [2015] No.56.
The rats were separated into a control group (i.e., fed and watered normally) and a hypersplenic model group. The hypersplenic group received an intragastric administration of 40% carbon tetrachloride (CCL4) solution in a peanut oil solution 3.0 ml/kg twice per week and had access to 15% alcohol solution (made from wine) instead of water. The amount of intragastric administration was controlled by weight changes which were measured weekly. For example, if there was a 5% change then the original dose was maintained, if the change was >5% the original dose was increased by 0.5 ml/kg, and if the change was <5% then the original dose was reduced by 0.5 ml/kg. A high yield of micronodular cirrhosis was obtained after approximately 8 weeks. All animals were euthanized by sodium pentobarbital injection. Orbital blood and the liver and spleen were harvested for analyses.
Laboratory analysis
Blood samples from the animals were collected in sodium citrate tubes for routine examination. The gel-activated tubes were allowed to clot and then centrifuged at 4000 rpm for 10 min at 4°C. Serum samples were assessed for alanine aminotransferase (ALT), aspartate aminotransferase (AST) and total protein (TP) content.
Fresh liver and spleen samples were fixed in 10% neutral formalin for 24 h before being embedded in paraffin. The fixed tissues were then cut into sections with a thickness of 5 μ m and mounted onto slides which were heated at 60°C overnight. Haematoxylin and eosin (H&E) staining and Masson trichrome staining were performed according to standard procedures. The diagnosis of cirrhosis and congestive splenomegaly was performed by two independent pathologists from Hainan General Hospital.
RNA extraction and real-time polymerase chain reaction analysis
Splenic total RNA was extracted using TRIzol reagent (Shanghai Novl and BioPhama) and quantified using visible spectrophotometry (Nano Drop 2000, Thermo Fisher). The reverse transcription reaction mixture contained: 10 μ l of 2× reverse transcription buffer, 1 μ l of 20 μ mol/ μ l Oligo (dT), 0.2 μ l of 200 U/ μ l M-MLV reverse transcriptase and diethyl pyrocarbonate (DEPC) water in a total volume of 20 μ l. The reaction conditions were 42°C for 30 min and 85°C for 10 min.
Real-time polymerase chain reaction (PCR) was performed to assess expression of rat genes using specific primer sequences as follows: CXCR3 forward: 5′-
PCR contained 10 μ L of 2 × PCR Master Mix, 0.08 μ l of 20 μ mol/ μ l forward primer, 0.08 μ l of 20 μ mol/ μ l reverse primer, 2 μ L of cDNA, and dd H2O in a total volume of 20 μ l. The PCR reaction parameters were set as follows: 95°C for 3 min, 95°C for 12 sec and 60°C for 40 sec for 40 cycles. The relative quantification of gene expression was calculated using the ΔΔct method.
Immunohistochemistry (EnVision method)
Paraffin-embedded rat spleen tissue sections were deparaffinized in xylene and rehydrated in alcohol. Antigen retrieval was performed in pH 6.0 sterile sodium citrate buffer. The tissue slides were cooled at room temperature, incubated in 3% H2O2 for 10 min to block endogenous peroxidase activity and rinsed three times in phosphate-buffered saline (PBS) with gentle agitation for 3 min. Following this, the slides were incubated at 4°C overnight in 1:400 diluted rabbit polyclonal CXCR3 antibody (Boster, PB0038) or 1:50 diluted rabbit polyclonal CCR5(Abzoom, AM2320) or CCR3 (Boster, BA2232) antibodies. After subsequent incubation in enzyme-labelled goat anti-rat IgG (ZSGB-BIO, PV-6000) for 20 min at 37°C, each slide was rinsed twice in PBS. The slides were then immersed in diaminobenzidine (DAB) detection solution (Polymer, Beijing, China, ZSGB, BIO, PV-6000-D) for 5 min. Sections were counterstained with haematoxylin for 1 min, dehydrated and mounted. Negative controls (no primary antibody) were included in all runs.
Image pro plus software v6.0 was used to evaluate the splenic expression of chemokine receptors, CXCR3, CCR5 and CCR3. The integrated optical density (IOD) and the rate of labelled cells in the entire spleen were calculated with five visual fields in one tissue section (×400) and the IOD value of positive cells of the fields was taken as the protein level of the chemokine receptors.
Western blotting analysis
For whole-protein extracts, tissues were washed with ice cold PBS and lysed in lysis buffer (Shanghai Novl and BioPhama). The lysates were mixed with SDS-PAGE loading buffer, incubated at 100°C for 5 min and then centrifuged for 5 min at 12,000 g to remove the insoluble precipitate. The samples were subsequently detected by 10% SDS-PAGE and then transferred onto a PVDF membrane (Millipore Millex, USA) in 25 m MTris base, 0.2 M glycine, and 20% methanol using a semidry blotter for 60 min at 30 mA. The membranes were blocked at 4°C overnight in Tris-buffered saline containing 0.1% Tween-20 (TBST) with 5% fat-free milk and incubated at 37°C for 2 h with the 1:200 diluted primary antibodies (Santa Cruz, anti-CXCR3, sc-6226; anti-CCR5, sc-17833; anti-CCR3, sc-7897).
Subsequently, the membranes were washed at least three times with TBST at 15 min intervals and incubated in horseradish peroxidase-conjugated goat anti-mouse or anti-rabbit IgG (1:2000 dilution, Jakeson). for 2 h at room temperature. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as a loading control. The proteins were visualized using Super-GL ECL Western Blotting Kit (Shanghai Novl and BioPhama) and exposed to clear-blue x-ray film. The density of the bands was assessed using the Gel-Pro Analyzer software.
Statistical analyses
Data were analysed using the Statistical Package for Social Sciences (SPSS®) for Windows® release 19.0 (SPSS Inc., Chicago, IL, USA) and a
Results
Characteristics of cirrhotic, hypersplenic rats
Of the 46 animals in this study, 10 were used as controls and 36 were included in the hypersplenic test group. After eight weeks of CCL4 administration, 15 rats died (42% mortality rate) and 21 had developed liver cirrhosis with ascites and hypersplenism. Compared with the control group, the hypersplenic group showed distortion of liver architecture and reduction of vascular architecture, regenerative nodules surrounded by fibrous tissue, significant fibrous hyperplasia in the portal area, pseudo lobule with proliferation, fibrous hyperplasia separated into different size pseudo lobules, cellular morphology changes, increased nuclear volume, and abundant adipose cells and multinucleate cells (Figure 1).

Haematoxylin and eosin (H&E) staining (a, b) and Masson trichrome (c, d) staining of the liver tissue of rats with hypersplenism (original magnification, ×200). Control samples are shown in (b) and (d) and test samples in (a) and (c). Pseudo lobules with proliferation and fibrous hyperplasia are shown in (a) and (c). The arrows on (c) and (d) indicate that the Masson trichrome stain has highlighted liver fibrosis in blue.
Serum ALT and AST levels of the cirrhotic hypersplenic rats were higher than those of the controls (264 ± 111 vs. 26.5 ± 7.9 [

(a & b) show levels of serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were significantly (**

Haematoxylin and eosin (H&E) staining (a, b) and Masson trichrome (c, d) staining of the spleen tissue from control (b, d) and hypersplenic (a, c) rats (original magnification, ×200). In hypersplenic tissue (a, c), the germinal central zone was enlarged and the marginal zone was thickened. The central arteries were thickened and fibrotic and the density of the capillary vessels was increased. The arrows on (c) and (d) indicate that the Masson trichrome stain has highlighted splenic fibrosis in blue.
Routine blood examination showed that there was a statistically significant increase in the white blood cell (WBC) count between cirrhotic hypersplenic rats and controls (23.9 ± 5.0 × 109/l vs. 6.2 ± 2.4 × 109/l [

The white blood cell (WBC) count in cirrhotic hypersplenic rats was significantly higher than in normal controls (a), while the red blood cell (RBC) and platelet (PLT) counts in hypersplenic rats were significantly lower than in normal controls (b, c). *
Variation in mRNA expression of the chemokine receptors
The mRNA levels of CXCR3 and CCR5 determined by RT-PCR, were significantly increased in hypersplenic rats compared with controls (3.1 ± 0.7 vs. 1.0 ± 0.04 [

The relative quantification of CXCR3, CCR5, and CCR3 gene expression in rat spleens. Quantification was based on the ratio between chemokine receptors and glyceraldehyde 3-phosphate dehydrogenase (GAPDH). By comparison with normal controls, spleens from hypersplenic rats showed significantly enhanced mRNA expression of Th1-associated chemokine receptors CXCR3 and CCR5 and reduced mRNA expression of Th2-associated chemokine receptors CCR3. **
Immunohistological changes in CXCR3, CCR5, and CCR3 in cirrhotic, hyper-splenic rats
CXCR3-, CCR5-, and CCR3-positive staining on the cell membrane and/or cytoplasm was yellow, brown, and dark brown, respectively (Figure 6). Cirrhotic hypersplenic rats showed higher rates of CXCR3- and CCR5-positive cells compared with normal controls (51.8% vs. 21.3% [
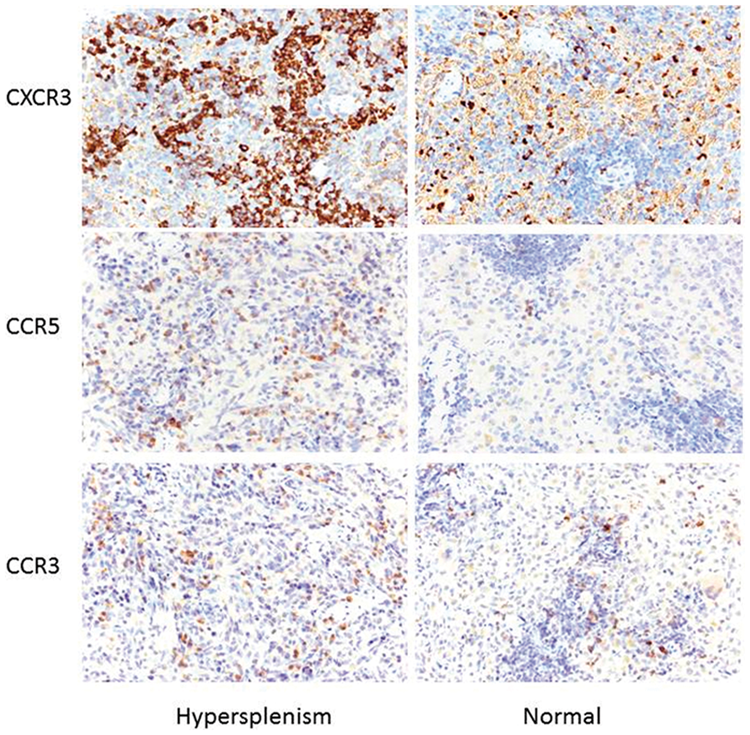
Immunohistological differences in CXCR3, CCR5, and CCR3 expression in the spleens of hypersplenic rats and normal controls (EnVision×400).

The rates of CXCR3- and CCR5-positive cells in hypersplenic rats were comparatively higher than in normal controls but there was no difference in the rate of CCR3-positive cells. ▼
Immunohistochemical staining showed that by comparison with controls, the mean densities of Th1 CXCR3 and CCR5 positive cells were significantly increased in cirrhotic, hypersplenic rats compared with controls (128 ± 33.9 ×103 vs. 26.7 ± 7.8 ×103 [

Compared with normal controls, the integrated optical density (IOD) values of CXCR3 and CCR5 were significantly elevated in cirrhotic hypersplenic rats, while CCR3 values were not significantly different. ▼
Variation in protein expression of the chemokine receptors in cirrhotic hyper-splenic rats
Western blot analysis showed that protein levels of CXCR3 and CCR5 in hypersplenic rats were significantly increased compared with controls (2.5 ± 0.9 vs. 1.3 ± 1.0 [

Western blot detection of CXCR3, CCR5, and CCR3 protein levels in the spleens of hypersplenic rats and normal controls. Quantification was based on the ratio between chemokine receptors and glyceraldehyde 3-phosphate dehydrogenase (GAPDH). (a) Representative western blots of CXCR3, CCR5, and CCR3 in the spleens of normal controls and hypersplenic rats. (b) Compared with normal controls, Th1-associated chemokine receptors CXCR3 and CCR5 were significantly upregulated, whereas Th2-associated chemokine receptor CCR3 was downregulated in the spleens of cirrhotic, hypersplenic rats. *
Discussion
CCL4-induced toxicity in rats has been widely used as an experimental model of cirrhosis.11–13 Acute CCL4 administration causes hepatotoxicity due to the highly reactive free radical metabolites generated by cytochrome P-450 enzymes, primarily CYP2E1, in hepatocytes.14,15 Despite the high mortality rate in this present study, data from the remaining animals showed that approximately eight weeks of CCL4 administration induced liver cirrhosis and hypersplenism. This was evidenced by the significant increase observed in serum levels of AST and ALT, significant reduction in serum TP levels and significant falls in RBC and PLT counts. In addition, histopathological alterations of the liver and spleen were observed that were similar to those seen in human hepato-cirrhosis with accompanying portal hypertension, hypersplenism and peripheral cytopenia.16,17 We believe that the high WBC count we found may have been due to an inflammatory response.
The balance of Th1/Th2 cells plays an important role in maintaining in normal physiological function. 18 Th1-driven responses are mediated by pro-inflammatory cytokines produced by Th1 cells (e.g., interleukin 1 [IL-1], IL-2, IL-12, interferon [IFN]- γ, and tumour necrosis factor [TNF]- α]) whereas Th2-driven responses are mediated by anti-inflammatory cytokines such as IL-4, 1L-10. 18 The interaction of chemokines with their receptors is an important step in the control of leucocyte proliferation, migration, and polarization in their particular environment.19,20The chemokine receptors CXCR3 and CCR5 have been found to be predominantly expressed on Th1 cells, while CCR3 receptor is expressed on Th2 cells.19,20 Studies have suggested that immune disorders mediated by a dysfunctional imbalance of T-cell subpopulations may be correlated to alterations in immune thrombocytopenia.19,20 Therefore, an abnormal expression of Th1/Th2 chemokine receptors may have a role in splenic immune disorders in cirrhosis and hypersplenism which in turn may explain peripheral cytopenia.
In our experimental model of cirrhosis and hypersplenism, immunohistochemical staining showed a higher rate of CXCR3- and CCR5-positive cells compared with controls but no difference between groups in the rate of CCR3-positive cells. In addition, results from RT-PCR demonstrated that by comparison with controls, mRNA expression of Th1-associated chemokine receptors CXCR3 and CCR5 was remarkably higher in hypersplenic animals but mRNA levels for Th2-associated chemokine receptor CCR3 was lower. Western blot analysis confirmed these results and showed that by comparison with controls, protein expression of the Th1-associated chemokine receptors CXCR3 and CCR5 was significantly increased in hypersplenic animals but the protein level for Th2-associated chemokine receptor CCR3 was decreased.
In conclusion, the abnormal expression of Th1-associated chemokine receptors in spleens of rats with cirrhosis and hypersplenism induced by CCL4 suggests that a functional imbalance between Th1/Th2 cells may play a role in the pathogenesis of hypersplenism. However, a limitation of the study is that these data are from an animal model of cirrhosis and hypersplenism and so may not totally reflect the actual situation in humans. Further research is required.
Footnotes
Acknowledgements
The authors thank Chunji Chen and Aihua He for their technical assistance.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
The work was supported by the Hainan provincial key scientific and technological research and development projects (SQ2016SHFZ0044) and Hainan Provincial Academician Workstation Funding (Qiong Cai Jiao [2018]1997).
