Abstract
Objectives
The aim of this study was to determine the therapeutic effects of tetrahydropalmatine (Tet) on disseminated intravascular coagulation (DIC) by exploring the role of Tet using a lipopolysaccharide (LPS)-induced DIC model.
Result
Tet ameliorated the damage to organ tissues, improved coagulation indexes, and reduced the inflammatory cytokine production in LPS-induced mouse DIC. Tet also inhibited TNF-α expression by suppressing NF-κB signaling pathway activation in an in vitro LPS model using RAW 264.7 macrophages.
Conclusions
Tet has a mitigating and therapeutic effect on the LPS-induced DIC model via anticoagulant and anti-inflammatory effects, showing its potential as an adjunct to DIC treatment.
Keywords
Introduction
Disseminated intravascular coagulation (DIC) is an acquired disease that is characterized by the intravascular activation of coagulation with a loss of localization arising from different causes. 1 It has been described as a disease-mediating mechanism that occurs secondary to many conditions including sepsis, trauma, and malignancies.2–5 For endotoxin and severe infection, the coagulation and anticoagulant balance are disordered because of the enhanced coagulation function and insufficient anticoagulant and fibrinolysis functions. 6
Current DIC-based therapies include blood transfusion and anticoagulants.1,7 Clinical treatment is mainly anticoagulant therapy, but there is no anticoagulant recommended for DIC anywhere in the world. 1 Therefore, it is necessary to investigate a new drug to broaden the treatment options for DIC and to improve the efficacy of DIC.
Tetrahydropalmatine (Tet; Figure 1) is one of the main active components that is extracted from Rhizoma Corydalis, 8 which has been found to have anti-tumor 9 and anti-inflammatory pain effects. 10 A recent study has shown that Tet significantly inhibits inflammation responses and inducible nitric oxide synthase (iNOS) protein expression in ketamine-induced mice, 11 which is a typical symptom of DIC. This indicates that Tet may be a potential drug for the treatment of DIC. However, the anti-DIC role of Tet has not been reported.

Tet structural formula.
Based on the above background, in this study, we investigated the potential role of Tet using a DIC model and sought to elucidate the in vitro and in vivo mechanisms to better understand its clinical potential.
Materials and methods
Animals
Swiss female mice (aged 4–5 weeks, weighing 23–27 g, specific pathogen-free [SPF] grade) were obtained from the Medical Experimental Animal Center (Guangdong, China). Animal experiments were approved and performed in accordance with the institutional guidelines from the review board for animal care (Jinan University Animal Care and Use Committee, Guangzhou, China).
Resources and reagents
Tetrahydropalmatine (98%, w/w, Shanghai Macklin Biochemical Co. Ltd, Shanghai, China) was dissolved in dimethyl sulfoxide (DMSO; Sigma-Aldrich, St Louis, MO, USA) with maximum solubility, and diluted to different concentrations with saline. Dulbecco’s Modified Eagle medium (DMEM) and fetal bovine serum (FBS) were purchased from BD Bioscience (Franklin Lakes, NJ, USA). Heparin was obtained from Beijing Tobishi Pharmaceutical Co. Ltd. (Beijing, China). Primary and secondary antibodies (anti-rabbit IgG, HRP-linked antibody #7074) for phosphorylated IKKα/β (p-IKKα/β), nuclear factor-kappa B (NF-κB), and tumor necrosis factor (TNF)-α were obtained from Cell Signaling Technology, Inc. (Shanghai, China).
Mice models and treatment protocols
Mice were randomly assigned to one of the following groups: (1) saline control group (normal group); (2) lipopolysaccharide (LPS) group; (3) DMSO group; (4) DMSO + LPS group; (5) Tet group (Tet administered intraperitoneally 30 minutes before LPS induction and 2 and 8 hours after LPS induction, 30 mg/kg as a low-dose group and 60 mg/kg as a high-dose); or (6) heparin group (10 IU/kg heparin using the same injection procedure as for the Tet group). LPS (Sigma-Aldrich, Shanghai Trading Co. Ltd., Shanghai, China) was administered intraperitoneally at a dose of 60 mg/kg. In the DMSO and DMSO + LPS groups, a saline solution containing 8% DMSO was administered intraperitoneally. DMSO was used only as a solvent for Tet, and the DMSO volume fraction in the Tet solution was also approximately 8%.
Histological analysis
Histomorphometric analysis was performed on ten randomly selected mice per group in each of the three time periods (before LPS induction, and at 2 and 8 hours after LPS induction). After the mice were sacrificed using isoflurane, the livers and kidneys were removed for the next procedure. Sections (5 µm) of formalin-fixed, paraffin-embedded liver and kidney tissue were used for histomorphometric analysis. After rehydration, the sections were stained with hematoxylin and eosin (H&E) (Baso Diagnostics, Inc., Zhuhai, China) to observe the histopathological status using an inverted microscope.
Blood sample preservation
After removing the liver and kidneys, mouse blood samples were collected using an abdominal aortic catheter and dissolved in 3.8% sodium citrate (1:9 vol/vol citrate/blood). Before analysis, blood samples were centrifuged at 3000 rpm for 10 minutes, and they were then stored at −80°C.
Blood sample detection
The levels of activated partial thromboplastin time (APTT), prothrombin time (PT), and fibrinogen (FIB) were measured using an automatic analyzer (Sysmex CS-5100, Kobe, Japan). Plasma levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and blood urea nitrogen (BUN) were measured using an automatic biochemical analyzer (Abbott c16000, Abbott Laboratories, Chicago, IL, USA). Interleukin (IL)-1α/β production was detected using an ELISA, in accordance with the manufacturer’s instructions (RayBiotech, Norcross, GA, USA).
Cell line
The RAW 264.7 murine macrophage cell line (ATCC, Manassas, VA, USA) were cultured in DMEM, 10% FBS, and 1% penicillin/streptomycin at 37°C in a humidified atmosphere containing 5% CO2.
Cell viability assay
After plating cells at a density of 50,000 cells/well in 96-well plates, cells were cultured in DMEM containing DMSO (0.1%) or a concentration of Tet (60 or 120 µM) but without FBS. An MTT assay (Sigma) was used to check the cell viability after 12 hours of drug treatment.
Western blot analysis
RAW 264.7 macrophage cells were first treated with Tet (60 or 120 µM) for 30 minutes and then with LPS (10 µg/mL) for 6 hours. Protein was extracted from the cells using RIPA buffer and the protein concentration was determined using the bicinchoninic acid (BCA) assay. Equal amounts of total protein were resolved by SDS-polyacrylamide gel electrophoresis and then transferred to PVDF membranes. Primary antibodies against p-IKKα/β (1:1000), NF-κB (P65) (1:1000), TNF-α (1:1000), and GAPDH (1:1000) were used. After washing three times in TBST, they were incubated with horseradish peroxidase-conjugated secondary antibodies for 1 hour. Finally, the antigen–antibody reaction was visualized by enhanced chemiluminescence assay. Quantification of band intensities on western blots was calculated using ImageJ (National Institutes of Health, Bethesda, MD, USA).
Statistical analysis
Statistical analysis were performed using IBM SPSS Statistics for Windows, version 19.0 (IBM Corp., Armonk, NY, USA). All experiments were performed independently at least three times and the results were presented as the mean ± standard error of the mean (SEM). A one-way ANOVA analysis was used to analyze differences in protein expression among the groups and the Wilcoxon test was used to analyze blood sample data. A p-value of P < 0.05 was considered to indicate a significant difference.
Results
Tet ameliorated tissue destruction and reduced organ damage in the LPS-induced mouse DIC model
Eight hours after LPS induction, HE staining showed blood stagnation in the central hepatic veins and sinusoids in the LPS group, and the hepatocytes were generally enlarged with loose and transparent cytoplasm and unclear nucleus morphology. Hepatocyte swelling led to narrowing of the sinusoids; but liver damage appeared to be more severe in the DMSO + LPS group, and microvascular rupture was also more severe. Tissue damage in the DMSO group was relatively mild, and only mild microvascular rupture was evident. The low-dose Tet group showed only slight cell damage, whereas the heparin group showed microvascular rupture and cell damage. Histological analysis in the high-dose Tet group was consistent with that in the normal group (Figure 2a).

Treatment of Tet ameliorated tissue destruction and reduced organ damage in the LPS-induced mouse DIC model. (a and b) Histological morphology (HE staining) of the liver (a) and kidney (b) 8 hours after LPS injection (magnification, ×200). Dilated hepatocytes, severe glomerular rupture, and deformation were detected in the LPS and DMSO + LPS groups, which is identified by the black arrow.
Renal HE staining showed that the glomerulus ruptured and Bowman’s capsule disappeared in the LPS and DMSO + LPS groups. Additionally, accompanied by glomerular atrophy and necrosis, the renal tissue was severely deformed and obviously damaged in the above two groups. As expected, renal tissue in the Tet treatment group showed only minor damage, which was similar to that of the heparin group. The morphology of the DMSO group was similar to that of the normal group and it showed no damage (Figure 2b).
Blood specimen detection showed that ALT and AST levels in the Tet group were significantly lower compared with those in the LPS group (P < 0.05), which was similar to the therapeutic effect of the heparin group (Figure 2c,d). The results showed that compared with the LPS group, Tet treatment significantly decreased the BUN level in plasma (P < 0.05; Figure 2e).
Tet improved blood coagulability in the LPS-induced mouse DIC model
In the LPS-induced mouse DIC model, PT and APTT levels were significantly increased while FIB levels were significantly decreased (P < 0.05; Figure 3). The results showed that Tet treatment could significantly decrease APTT and PT levels in plasma in the LPS-induced mouse DIC model (P < 0.05), and the PT level in high-dose Tet treatment was lower compared with that in the heparin group at 2 hours after LPS induction (Figure 3a,b). Compared with the LPS group, the FIB level in the Tet and heparin groups increased significantly 2 hours after LPS induction (P < 0.05). Moreover, there was a trend toward recovery of the FIB level in the Tet group, but not in the heparin group or the LPS group (Figure 3c).

Treatment of Tet improved blood coagulation indexes in the LPS-induced mouse DIC model.
Tet reduced inflammatory cytokine levels in the LPS-induced mouse DIC model
Eight hours after LPS induction, we evaluated the therapeutic effect of Tet on LPS-induced DIC by detecting IL-1α and IL-1β levels in each group (Figure 4).

Treatment with Tet reduced the inflammatory cytokine levels in the LPS-induced mouse DIC model. (a and b) Plasma samples (n = 10) from each group were analyzed for IL-1α (a) and IL-1β (b) levels 8 hours after LPS injection. Data are presented as the mean ± SEM of three assays. ##P < 0.01 compared with the normal group; *P < 0.05 compared with the LPS group.
Compared with the LPS group, IL-1α and IL-1β levels in the Tet treatment group were significantly decreased (P < 0.05), and these inflammatory cytokine levels (i.e. IL-1α and IL-1β levels) decreased with an increasing Tet dose (Figure 4).
Effect of Tet on cell viability and cytotoxicity in RAW 264.7 macrophages
To assess the effect of Tet on cell viability and cytotoxicity, we examined the effect of Tet on cell viability in RAW 264.7 macrophages using the MTT assay. The experimental results showed that Tet was not cytotoxic to RAW 264.7 macrophages (Figure 5).
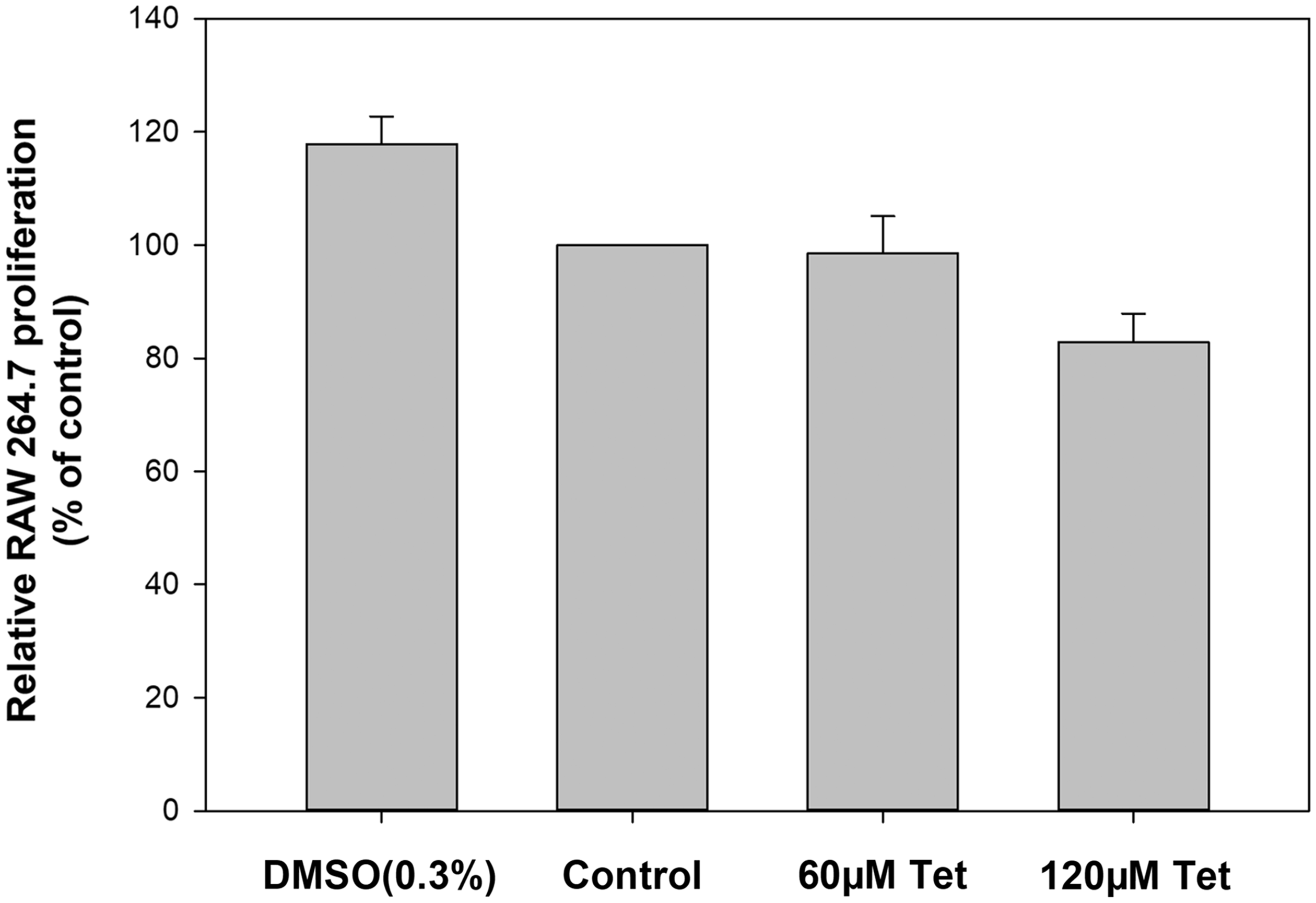
Effect of Tet on RAW 264.7 macrophage cell viability and cytotoxicity. DMSO and Tet did not produce significant cytotoxicity in RAW 264.7 macrophages. Data are presented as the mean ± SEM of three assays (n = 6 per group).
Tet inhibited TNF-α expression by regulating the NF-κB signaling pathway in RAW 264.7 macrophages
We hypothesized that Tet could attenuate the inflammatory response during DIC by inhibiting TNF-α expression, so we used LPS to establish an inflammatory cell model in vitro. Western blot was used to examine TNF-α expression in different groups. We also investigated the NF-κB signaling pathway to elucidate the underlying mechanisms of changes in the TNF-α level. The results showed that the TNF-α (P < 0.05) and p-IKKα/β (P < 0.05) levels were significantly decreased in the Tet-treated group compared with the DMSO + LPS group (Figure 6).

Effect of Tet treatment on TNF-α and p-IKKα/β expression levels in LPS-induced inflammatory model cells in vitro. (a) TNF-α, p-IKKα, and p-IKK β levels were evaluated by western blot. (b–e) TNF-α (b), p-IKKα (c), p-IKK β (d), and NF-κB (e) levels were normalized using GAPDH. Data are presented as the mean ± SEM of three assays (##P < 0.01 compared with the control group; *P < 0.05, **P < 0.01 compared with the DMSO + LPS group).
Discussion
Previous studies showed that massive microthrombosis causes microcirculatory damage, leading to organ ischemia and focal necrosis in DIC. 12 As expected, Tet significantly improved liver and kidney damage in this study (Figure 2). The histomorphology of the liver and kidney in the Tet group was similar to that in the heparin group. The damage index in the liver and kidney showed that the Tet treatment group had significantly lower levels of ALT, AST, and BUN compared with the LPS and DMSO + LPS groups (Figure 2c–e). All the above data indicated that Tet had a certain therapeutic effect on DIC. DMSO was only used as a solvent for Tet in this study. To exclude its influence in the experiment, we included a DMSO group and a DMSO + LPS group to compare its effects (Figure 2a,b). The morphology of the DMSO-treated group was consistent with that of normal tissue, demonstrating that LPS was the main pathogenic factor and Tet was the main experimental factor.
PT, APTT, and FIB are hematological tests that are commonly used as coagulation parameters to diagnose DIC. 13 APTT and PT reflect whether the endogenous and exogenous coagulation systems are normal, while FIB reflects whether the coagulation substances are sufficient and can promote the formation of a stable thrombus. 14 In this study, we observed that PT and APTT levels were significantly increased while the FIB level was significantly decreased in the LPS-induced mouse DIC model (Figure 3a-c). These data indicate that the coagulation and fibrinolytic systems in the LPS-induced mouse DIC model had been severely disrupted, which was an important typical symptom of DIC. PT and APTT levels were significantly increased (Figure 3a,b), suggesting that coagulation factors were largely consumed because of the extensive formation of microthrombi in DIC. Inadequate coagulation factors resulted in a prolonged coagulation time, which was also an important reason for hemorrhage that was induced by DIC. A decrease in FIB levels in the LPS-induced mouse DIC model was also observed (Figure 3c), which indicated that excessive consumption of fibrinogen resulted in the formation of stable microthrombi, leading to severe microcirculation disorders and aggravated the course of DIC. Thus, abnormal PT, APTT, and FIB levels in the LPS-induced mouse DIC model indicated that many microthrombi were being formed, coagulation factors and platelets were depleted, and the fibrinolytic system may be secondarily activated. All of these placed the blood into a depletive hypocoagulable state, which could cause significant bleeding symptoms and explain the bleeding in the liver and renal tissues (Figure 2). As expected, Tet treatment significantly improved the levels of coagulation parameters, indicating that it can improve coagulation to some extent, and thus, play a role in treatment of DIC.
Tet had an anticoagulant effect that was similar to that of heparin (Figure 3). Considered to be an anticoagulant, heparin plays an important role in treating patients with DIC. The function of heparin is to increase the affinity between antithrombin III and thrombin, thereby accelerating inactivation of thrombin.15,16 The FIB level tended to recover after Tet treatment (Figure 3c), but not in the heparin group, suggesting that Tet treatment may not have a hemorrhagic reaction, which is an adverse reaction to heparin. The above phenomenon suggests that Tet may be more suitable for clinical application.
Previous studies have reported that IL-1α, IL-1β, and TNF-α are important early release cytokines in the inflammatory response, and they are regulated by the NF-κB signaling pathway.17,18 The accumulation of these inflammatory factors will lead to a secondary inflammatory cascade that exacerbates the disturbance of the balance between coagulation and anticoagulation.17,19
In the current in vivo experiments, we observed a significant increase in IL-1α and IL-1β levels in the LPS group. However, Tet treatment significantly reduced IL-1α and IL-1β levels in the LPS-induced mouse DIC model (Figure 4), suggesting that Tet could play a role in the treatment of DIC by decreasing inflammatory cytokines such as IL-1α and IL-1β. In the in vitro experiment, the result of the significant reduction in TNF-α expression in the Tet-treated group compared with the DMSO + LPS group showed that Tet can ameliorate the inflammatory response in the DIC model, thus acting as an anti-DIC effect (Figure 6). This was consistent with the results of the previous in vivo experiment (Figure 4).
IKKα/β plays an extremely important role in NF-κB activation and can regulate the rapid onset and shutdown of NF-κB activation. 20 When the NF-κB canonical signaling pathway is activated, the IKK complex is activated by phosphorylation by NF-κB-inducing kinase, and phosphorylated IKK degrades IκBα, thereby activating NF-κB subunits. Activated NF-κB subunits translocate from the cytosol into the nucleus and bind to the corresponding inflammation-related genes to initiate inflammatory cytokine transcription and form an inflammatory cascade. Compared with the DMSO + LPS group, Tet treatment reduced the p-IKKα/β expression levels, suggesting that NF-κB inflammatory pathway activation was inhibited rather than stimulated, thereby inhibiting the expression of transcription factors such as TNF-α (Figure 6). This corresponds to the finding that the TNF-α expression level was significantly reduced in the Tet-treated group, indicating that Tet suppresses TNF-α expression by suppressing the NF-κB signaling in RAW 264.7 macrophages. However, TNF-α and p-IKKα/β levels in the DMSO + LPS group were higher compared with those in the LPS group (Figure 6), suggesting that DMSO combined with LPS may aggravate inflammation, but Tet can significantly inhibit this inflammation. The results of the in vitro cell experiments are not completely consistent with those of the in vivo animal experiments (Figure 4), which may be related to other pharmacological effects that are exerted by Tet and DMSO in vivo and possibly related to their concentrations. However, the anti-inflammatory effect of Tet was evident in the DIC model.
Although DIC is a serious disease, there is no single biomarker with which to make a definite diagnosis of DIC, and no anticoagulant is recommended for the treatment of DIC anywhere throughout the world.1,2 In our study, in vitro and in vivo experiments have shown that Tet has the effect of alleviating and treating LPS-induced DIC, indicating that Tet has some promising significance for broadening the treatment of DIC and improving the efficacy of DIC treatment. However, the limitations of this study include the lack of in-depth study of mechanisms and pathways, as well as the dose-response relationship of Tet and the uncertainty of the treatment safety index.
Footnotes
Acknowledgements
We thank Dr. Xiao-Mei Tang (Department of Obstetrics and Gynecology, The First Affiliated Hospital of Jinan University, Guangzhou 510630, China) for helpful discussions.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research was supported by the Guangdong Medical Science and Technology Research Fund Project (No. A2016179).
Ethical disclosure
The authors state that they have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations. The authors also followed the ethical guidelines of the National Guide for the Care and Use of Laboratory Animals, Jinan University Animal Care, and the Use Committee (Guangzhou, China) approved all the experimental procedures.
