Abstract
Objective
To investigate the anti-tumor effect and mechanism of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) in non-small cell lung carcinoma (NSCLC) in mice.
Methods
We first established NSCLC animal models using 20 BALB/c nude mice that were randomly divided into two equal groups (n = 10): TRAIL-treated and control untreated groups. We measured expression levels of B cell leukemia/lymphoma-2 (Bcl-2), Bcl-2-associated X protein (Bax), vascular endothelial growth factor (VEGF), and VEGF receptor (VEGFR). We also performed microvessel density, terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL), and immunohistochemical assays to determine the effect of TRAIL on apoptosis and angiogenesis in NSCLC tumors in vitro.
Results
TRAIL inhibited tumor growth in the NSCLC mouse model, and the TUNEL assay showed that it induced tumor cell apoptosis. Immunohistochemical staining revealed that TRAIL induced Bcl-2 protein downregulation, suggesting that the mitochondrial apoptotic pathway is involved in regulating NSCLC apoptosis. However, TRAIL did not affect Bax protein expression. Immunohistochemical staining also revealed significantly reduced VEGF and VEGFR protein expression in the TRAIL group, indicating that TRAIL limits angiogenesis in NSCLC tumor tissues.
Conclusions
In conclusion, TRAIL inhibits NSCLC growth both by inducing tumor cell apoptosis and restricting angiogenesis in tumors.
Keywords
Introduction
Lung cancer is the leading cause of cancer-related deaths in both men and women worldwide, 1 with non-small cell lung cancer (NSCLC) accounting for approximately 85% of all cases. 2 Systemic chemotherapy is currently the primary treatment method for early-stage NSCLC. However, therapy is often unsatisfactory because most chemotherapy drugs are toxic to healthy somatic cells, and drug resistance has become increasingly problematic.3–5 Hence, there is an urgent need to develop non-cytotoxic specific agents and measures to overcome chemotherapy resistance in lung cancer therapy. Activation of death receptors was found to be effective in NSCLC therapy through directly inducing apoptosis and preventing the development of cellular drug resistance. 6
Tumor necrosis factor (TNF)-related apoptosis-inducing ligand (TRAIL), a new member of the TNF family, has shown promise as an anti-cancer drug because of its ability to specifically target tumor cells for apoptosis. 7 TRAIL mediates apoptosis by binding directly to the TRAIL DR4/5 death receptors on the plasma membrane, activating the death signaling pathway, inducing caspase-8 and its downstream effecter caspases, and resulting in apoptosis. 8 TRAIL has also been implicated in disease development, and targeting TRAIL death receptors on tumor cells, either alone or in combination with other chemotherapeutic drugs, was reported to be an effective therapeutic strategy.9–11 Hence, we hypothesized that targeting TRAIL may be similarly effective in lung cancer therapy.
Because the underlying mechanism of TRAIL in lung carcinoma has not yet been characterized, we designed this study to clarify the role of TRAIL in mediating apoptosis and angiogenesis of lung tumors using in vivo mouse models and the in vitro NSCLC A549 lung carcinoma cell line. Specifically, we investigated the expression levels of the apoptotic markers B cell leukemia/lymphoma-2 (Bcl-2) and Bcl-2-associated X protein (Bax), as well as angiogenesis markers vascular endothelial growth factor (VEGF) and vascular endothelial growth factor receptor (VEGFR). We also performed microvessel density (MVD), terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL), and immunohistochemical assays.
Materials and methods
Animals
Twenty 4–7-week-old male BALB/c nude mice (Shanghai SLAC Laboratory Animal Co., Ltd., Shanghai, China) weighing between 20–22 g were bred in pathogen-free conditions under a 12-hour light/dark cycle. Mice were allowed food and water ad libitum. All animal experiments were performed according to International Ethical Guidelines and National Institutes of Health Guidelines on the Care and Use of Laboratory Animals, and with the approval of the Institutional Animal Care and Use Committee of Fujian Cancer Hospital, Fujian Medical University Cancer Hospital, China (reference number K201427).
Cell culture
Human NSCLC A549 cells (American Type Culture Collection, Manassas, VA, USA) were cultured in Dulbecco’s modified Eagle’s medium (DMEM) containing 10% (v/v) fetal bovine serum (FBS) and penicillin–streptomycin (100 U/mL; 100 µg/mL) at
Animal model and grouping
Subcutaneous injection of human NSCLC A549 cells into the right flank of BALB/c nude mice was performed to establish our NSCLC animal model. Tumor-bearing mice were randomly divided into two equal groups (n = 10) after 7 days: the control group (treated with normal saline) and the TRAIL-treated group (treated with 0.08 mg/kg TRAIL). Recombinant human TRAIL Apo II ligand was purchased from PeproTech, Inc. (Rocky Hill, NJ, USA). According to our previous study, 12 TRAIL was injected once daily into the mouse abdominal cavity for 15 days. Mouse body weights were measured once every 5 days from experimental Day 1, and the tumor size was measured once every 5 days from experimental Day 7. Tumors were excised from mice and weighed on Day 22. Tumor volume (mm3) was calculated as d2 × D/2, where d and D represent the shortest and longest diameter, respectively.
In situ TUNEL assay
Tumor sections were treated with formalin for 48 hours at 4°C, then with 4% formaldehyde in phosphate-buffered saline (PBS) for 25 minutes at 4°C, and finally immersed in 0.2% TritonX-100 in PBS for 5 minutes. Equilibration Buffer (100 µl) was added to the slides at room temperature for 5–10 minutes, and staining was performed using the TUNEL assay kit (Promega Systems, Madison, WI, USA). Apoptotic cells that stained pale brown were visualized at × 100 magnification under light microscopy (Olympus Optical Co., Tokyo, Japan) equipped with the Moticam 5000 C camera (Richmond, BC, Canada). Five randomly selected fields were used to count apoptotic cells, and image analysis was performed using Motic Med 6.0 software (Xiamen Motic Software Engineering Co., Ltd., Xiamen, China). The apoptosis index was calculated as the number of apoptotic cells/total number of cells × 100%. All other chemicals including xylene and ethanol were purchased from Sigma (St. Louis, MO, USA) unless otherwise stated.
Immunohistochemistry
Immunohistochemistry was performed to assess protein levels of Bcl-2, Bax, VEGF, and VEGFR2, as well as MVD in tumor tissues. Tissue sections were incubated with Bax, Bcl-2, VEGF, and VEGFR2 primary antibodies (Maixin Bio, Fuzhou, China) for 1 hour at 37°C. Yellow or tan particles at the cell membrane or cytoplasm were indicative of Bcl-2, Bax, VEGF, and VEGFR2 expression. Staining intensities and areas were quantified as the average integral absorbance. To determine MVD, two areas with a relatively high number of new vessels were selected by preliminary scanning at × 10–100 magnification. Five random fields of each selected area at × 400 magnification were then analyzed.
Statistical analysis
Statistical analysis was performed using SPSS 20.0 software (SPSS Inc., Armonk, NY, USA). Data are presented as the mean ± standard deviation (SD). Comparisons between two groups were performed using the Student’s t-test. Values of P<0.05 were deemed statistically significant.
Results
TRAIL inhibits tumor growth in mice
There was no significant change in the body weight of tumor-bearing mice in the TRAIL group compared with the control group during the 15-day experiment (Figure 1a). However, there was a significant increase in both tumor volume (P < 0.05; Figure 1b) and tumor weight (P < 0.05; Figure 1c) in tumor-bearing mice in the TRAIL group compared with the control group, indicating that TRAIL inhibits tumor growth.

The effect of TRAIL on tumor growth in vivo. Tumor-bearing BALB/c nude mice were treated with TRAIL 0.08 mg/kg once daily for 15 days. The same volume of NS was administered to the control group. Body weight (a), tumor volume (b), and final tumor weight (c) were then measured. Values are shown as mean ± SD, n = 10, *P < 0.05 versus the control group.
TRAIL promotes tumor cell apoptosis via the Bcl-2 pathway
Following the characterization of the effect of TRAIL on tumor growth in NSCLC mouse models, we next determined the underlying mechanism by which TRAIL regulates tumor cell apoptosis. We observed a significant increase in apoptotic A549 cells in the TRAIL group compared with the control group (TRAIL vs. control: 15.2% vs. 10.5%; P < 0.05; Figure 2), indicating that TRAIL induces tumor cell apoptosis. This observation was consistent with the significant downregulation of Bcl-2 protein expression (TRAIL vs. control: 16.5% vs. 24.1%; P < 0.05). No significant change was detected in Bax protein expression (TRAIL vs. control: 24.6% vs. 26%; Figure 3).
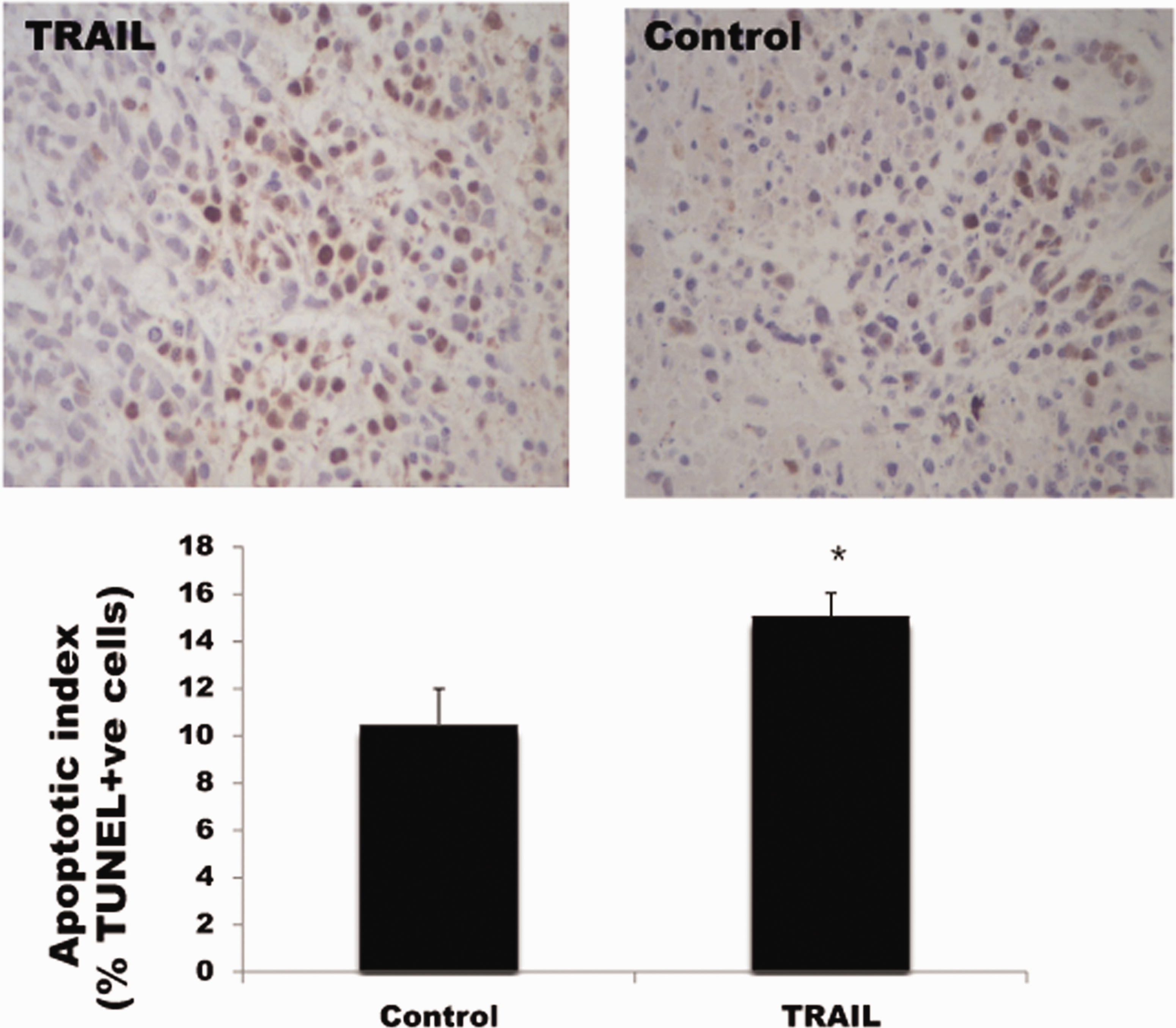
The effects of TRAIL on apoptotic cells. The tumor apoptotic index was detected by the TUNEL assay. Quantification of the results was performed as a percentage of positively-stained cells. Data are presented as mean ± SD from six individual mice per group. *P < 0.05 versus the control group. Positive apoptotic cells are those stained pale brown.

The effects of TRAIL on related apoptosis factors. Bcl-2 and Bax protein expression were determined using immunohistochemical staining. Yellow or tan particles on cell membranes or in the cytoplasm represent positive Bcl-2 or Bax protein expression. Data are presented as mean ± SD from six individual mice per group. *P < 0.05 versus the control group.
TRAIL inhibits angiogenesis in an NSCLC mouse model
Next, we investigated the effect of TRAIL on modulating angiogenesis in tumors by assessing MVD and angiogenesis markers including VEGF and VEGFR2. Immunohistochemical staining revealed significantly reduced MVD (TRAIL vs. control: 14.5% vs. 26.3%; P < 0.05; Figure 4) and VEGF (TRAIL vs. control: 14.0% vs. 36.8%; P < 0.05; Figure 5), and reduced VEGFR2 (TRAIL vs. control: 33.1% vs. 37.6%; Figure 5) expression in tumor-bearing mice compared with controls. Taken together, TRAIL treatment effectively inhibited angiogenesis in our NSCLC mouse model.

The effects of TRAIL on the expression of microvessel density (MVD). MVD was detected by immunohistochemical staining for CD31 as a marker. MVD is represented as a percentage of CD31-positive tumor cells. Values are shown as mean ± SD, n = 6; *P < 0.05 versus the control group.

The effects of TRAIL on VEGF and VEGFR2 protein expression evaluated using immunohistochemical staining. Data are presented as mean ± SD from six individual mice per group. *P < 0.05 versus the control group.
Discussion
TRAIL is a well-studied cytokine with a promising anti-cancer function 13 because of its ability to directly induce tumor cell apoptosis through the activation of its death-inducing receptors (TRAIL-R1 and TRAIL-R2).14,15 However, the role of TRAIL in lung carcinoma and the underlying mechanism are poorly characterized. Hence, we performed this study to clarify how TRAIL regulates apoptosis and angiogenesis in NSCLC. We found that tumor growth was inhibited in our TRAIL-treated NSCLC mouse model, but that there was no significant change in the body weight of tumor-bearing mice during the 15-day experiment, indicating that TRAIL treatment did not induce side effects in vivo. We also observed increased apoptosis using the TUNEL assay and an increased Bax/Bcl-2 ratio, as determined by immunohistochemical staining, in the TRAIL group. Additionally, significantly reduced MVD and angiogenesis marker expression were detected in TRAIL-treated mice. Taken together, our findings suggest that increased apoptosis and decreased angiogenesis are the underlying mechanisms of TRAIL-mediated inhibition of NSCLC tumor growth in vivo.
Apoptosis is regulated by several signaling cascades including the mitochondrial death pathway. Bcl-2 and Bax are factors in this pathway and form heterodimers; Bcl-2 preserves the mitochondrial membrane integrity while Bax promotes cytochrome C release from the mitochondria into the cytosol to induce apoptosis. The Bax/Bcl-2 ratio determines the response of cells to various apoptotic stimuli.16,17 Hence, the increased Bax/Bcl-2 ratio observed in this study parallels the detected increase in apoptosis, suggesting that NSCLC A549 cell apoptosis may be mediated via the mitochondrial death pathway. Because the modulation of MVD and changes in VEGF and VEGFR2 levels affect tumor angiogenesis, invasion, and metastasis,18–20 these are ideal markers of tumor angiogenesis. The observed changes in MVD, VEGF, and VEGFR2 levels upon TRAIL treatment of NSCLC mice in the present study demonstrate the effect of TRAIL on angiogenesis.
In conclusion, we demonstrated that TRAIL inhibits NSCLC tumor growth by inducing tumor cell apoptosis potentially via the Bcl-2/Bax mitochondrial death pathway and restricting tumor angiogenesis. Our results suggest that TRAIL could be targeted in clinical NSCLC therapy.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the Natural Science Foundation of Fujian Province, China (Nos. 2011J01035 and 2019J05140) and the Fujian Provincial Health and Family Planning Research Talent Training Program, China (No. 2016-ZQN-20) for funding this study.
