Abstract
Objective
To determine the effect of the upregulation or knockdown of the ephrinB2 (
Methods
Rat BMSCs were infected with lentivirus vectors carrying EphrinB2 and shRNA-EphrinB2. EphrinB2 mRNA and protein levels were quantified. At 28 days of culture with neuronal cell-conditioned differentiation medium, levels of microtubule-associated protein 2 (MAP2), CD133 and nestin were detected in EphrinB2/BMSCs and shEphrinB2/BMSCs using quantitative polymerase chain reaction and immunofluorescence. The ability of these cells to migrate was evaluated using a transwell assay.
Results
BMSCs were successfully isolated as indicated by their CD90+ CD29+ CD34– CD45– phenotype. Three days after ephrinB2 transduction, BMSC cell bodies began to shrink and differentiate into neuron-like cells. At 28 days, levels of MAP2, CD133 and nestin, as well as the number of migratory cells, were higher in lenti-EphrinB2-BMSCs than in the two control groups. The shEphrinB2/BMSCs had reduced levels of MAP2, CD133 and nestin; and a lower rate of cell migration. Similarly, increased levels of Grb4 andp21-activated kinase in the EphB4/EphrinB2 reverse signalling pathway were observed by Western blot.
Conclusions
LV-EphrinB2 can be efficiently transduced into BMSCs, which then differentiate into neuron-like cells.
Introduction
Bone marrow mesenchymal stem cells (BMSCs) are precursor cells distinct from haematopoietic stem cells. Accumulating evidence from
Erythropoietin-producing hepatocellular receptors can be divided into two groups: EphA (EphA1–EphA8, EphA10) and EphB (EphB1–EphB4, Eph B6). 5 Ephrin ligands are divided into two groups: EphrinA (EphrinA1–A5) and EphrinB (EphrinB1–3). 5 Currently, there are limited reports on the roles of EphB4 and EphrinB2 in stem cells. 6 Similar to other tyrosine kinase receptors, signal transduction between EphB4 and EphrinB2 is bidirectional, as each can be activated by the other, producing ‘forward signals’ and ‘reverse signals’. 3 Autophosphorylation of the receptor is initiated by EphrinB2, with phosphoinositide 3-kinase, Src, mitogen-activated protein kinases, Akt and other proteins mediating the cascade reaction, thus giving rise to what is known as a forward signal. 5 In contrast, the reverse signal is triggered when EphrinB2 binds to EphB4 as a ligand (Src-family kinases), a tyrosine residue of EphrinB2 is phosphorylated, and the downstream signalling pathway is activated by interaction with the SH2/SH3 domain of the adapter protein growth-factor-receptor-bound protein 4 (Grb4). 3 A previous study demonstrated that p21-activated kinase (PAK) acts as a key downstream component of ephrin-B3-Grb4 reverse signalling to mediate axon retraction and pruning. 7 The PDZ domain-binding C-terminus of EphrinB2 can interact with the PDZ domain of various proteins. 5 One of these, regulator of G-protein signalling 3 (PDZ-RGS3), acts downstream of EphrinB2 and is involved in regulating cell migration. 5 The EphrinB2 reverse signalling pathway for PDZ-RGS selectively promotes G protein-coupled stromal cell-derived factor 1 chemotaxis. 8 Forward signalling was shown to inhibit angiogenesis, preventing endothelial cell adhesion, migration, and vascular sprouting, whereas reverse signalling had the opposite effect. 9
In this current study, the effects of lentiviral-mediated upregulation and knockdown of the ephrin B2 (
Materials and methods
Animals
Ten male specific pathogen-free Sprague–Dawley rats aged 4–6 weeks and weighing 160–200g obtained from the Experimental Animal Centre of Nanjing Medical University, Nanjing, Jiangsu Province, China were used in this study. Rats were exposed to a 12-h light–dark cycle with free access to food and water. All animal experiments were approved by the Animal Care Committee in China and were performed according to institutional guidelines.
Isolation, purification, and identification of rat BMSCs
Rats were sacrificed by cervical dislocation and placed in 75% ethanol for 5 min. Briefly, the bone marrow was obtained from the bilateral femurs and tibias, flushed out with 5 ml Dulbecco's modified Eagle's Medium (DMEM)/F12 (Gibco Life Technologies, Carlsbad, CA, USA) and mixed with equivalent percoll separating medium (1.073 g/l; Gibco Life Technologies). Following centrifugation at 900
Preparation of lentiviral vectors
The
Short hairpin (sh)RNA sequences of EphrinB2.

Lentivirus transduction
Rat BMSCs were cultured for three generations and then seeded into 24-well plates at a density of 1 × 105 cells/well. The cells were cultured for 2 or 3 days, whereby the cell fusion rate was approximately 50%. Viruses at multiplicity of infection (MOI) rates of 10 and 20 were added together with 5 µg/ml polybrene. To establish EphrinB2-BMSCs, the lenti-EphrinB2-GFP or lenti-GFP empty vector was used to transduce naive BMSCs at passage 5 (MOI = 10 and 20). In the negative-transduction group, the lenti-GFP empty vector was used. The no-transduction control group was not treated at all. After transduction for 24–48 h, enhanced green fluorescent protein (EGFP) levels were observed every day using a BX41 upright fluorescence microscope (Olympus, Tokyo, Japan) and transduction efficiency was determined.
After transduction, four groups were set up: control group (no transduction, other culture conditions were the same as those used for the transduction groups), NC group (negative control group, transduced with lenti-GFP empty vector), lenti-10moi-EphrinB2-GFP group (transduced with lenti-EphrinB2-GFP at a MOI of 10), and lenti-20moi-EphrinB2-GFPgroup (transduced with lenti-EphrinB2-GFP at a MOI of 20). Each group was tested in triplicate.
Third-passage BMSCs were transduced with no lentiviral vector, negative control lentiviral vector, and LV-shRNA1-EphrinB2, LV-shRNA2-EphrinB2, LV-shRNA3-EphrinB2, with a MOI of 20, and 5 µl polybrene (10mg/ml) was added, respectively. The stable control, negative control group, and EphrinB2-knockdown cell lines, termed BMSCs, GFP/BMSCs, and EphrinB2-shRNA1/BMSCs, EphrinB2-shRNA2/BMSCs and EphrinB2-shRNA3/BMSCs, were established in culture medium containing puromycin (5 µl/ml).
Quantitative PCR (qPCR) analysis of EphrinB2 mRNA levels in BMSCs
To determine the abundance of EphrinB2 mRNA, total RNA was extracted from cells using an RNA Extraction kit (Invitrogen). Based on the company’s protocol, cDNAs were synthesized from 0.1 mg of total RNA using a PrimeScript™ RT Reagent Kit (Takara). RT was carried out using a Step OnePlus™ Quantitative Real-Time PCR System (Applied Biosystems) with Syber Green PCR Master Mix (Applied Biosystems) according to the manufacturer’s guidelines under the following conditions: preliminary denaturation at 95 °C for 3 min, followed by 30 cycles of denaturation at 95 °C for 30 sec, annealing at 56 °C for 45 sec and elongation at 72 °C for 45 sec, with a final elongation step at 72 °C for 10 min using a GeneAmp® PCR System 9700 thermal cycler (Applied Biosystems).The following qRT–PCR primer sequences were used (Genechem Biotech): for the internal control glyceraldehyde 3-phosphate dehydrogenase (GAPDH) forward: 5ʹ-
Western blot analysis of EphrinB2 protein levels in BMSCs
Total cellular protein was extracted 48 h after transduction and the protein concentration was measured using a BCA kit (Solarbio, Shanghai, China). Total protein (30 μg) was boiled with 2 × loading buffer at 95 °C for 10 min. Samples were then separated by 15% sodium dodecyl sulphate–polyacrylamide gel electrophoresis and electrophoretically transferred to a polyvinylidene fluoride membrane (Millipore, Billerica, MA, USA) at 100 V for 45–70 min. Then, the membrane was blocked with 5% bovine serum albumin (BSA) for 1 h at room temperature, followed by incubation with a rabbit polyclonal anti-EphrinB2 primary antibody (1:200 dilution; Abcam, Cambridge, UK) at 4 °C overnight. The blots were then rinsed three times with tris-buffered saline-Tween 20 (TBS-T; pH 7.6) for 5 min followed by incubation with horseradish peroxidase-conjugated goat anti-rabbit secondary antibody (1:5000 dilution; Solarbio) at room temperature for 1 h. After washing the membrane three times for 5 min each with TBS-T (pH 7.6), the blots were developed by chemiluminescence followed by imaging with a Bio-Rad Geldoc EZ formatter (Bio-Rad Laboratories, Hercules, CA, USA). All membranes were incubated also with a monoclonal anti-GAPDH antibody (1:1000 dilution; Santa Cruz Biotechnology, Santa Cruz, CA, USA) as an internal reference control. Band intensity was quantified by ImageJ software (NIH, Bethesda, MD, USA). GAPDH was used as loading control. The levels of EphrinB2 protein were detected using Western blotting and the most effective siRNAs were used for further investigation.
Differentiation of BMSCs into neural-like stem-like cells in vitro
The potential of rat BMSCs to differentiate into neural-like stem cells was analysed. To induce BMSC differentiation, third-passage BMSCs were collected, and 0.25% trypsin was added to digest the cells for 10 min. Supernatants were obtained and pancreatin was subsequently removed. The cells were resuspended in neuronal cell-conditioned differentiation medium containing 60 g/l glucose, 20 g/l basic fibroblast growth factor (FGF), 20 g/l epidermal growth factor (EGF) and 2% B27 DMEM/F12 (1:1) at a density of 5 × 107 cells/l. The medium was changed based on cell growth. Cell morphology was observed directly using phase-contrast microscopy (BX41; Olympus). Cells were cultured in 24-well plates and then fixed with 4% paraformaldehyde at 20 °C. To analyse the effects of the increased levels of EphrinB2 protein on BMSC differentiation, the third-passage BMSCs were divided into four groups: control group, NC group, lenti-10moi-EphrinB2-GFP group and lenti-20moi-EphrinB2-GFP group. The morphology of the BMSCs and the levels of specific neural markers, including microtubule-associated protein 2 (MAP2), CD133 and nestin in each group were observed using a BX41 upright fluorescence microscope (Olympus). After 28 days of culture, positive induction was detected by immunofluorescence staining. Cells were washed with 10 mM PBS (pH 7.4) and permeabilized with 0.2% Triton X-100 for 5 min. After washing with 10 mM PBS (pH 7.4), cells were incubated in a blocking buffer containing 5% BSA for 30 min at 20 °C, followed by incubation with anti-MAP2 (1:500 dilution; Proteintech, Chicago, IL, USA), anti-CD133 (1:500 dilution; Abcam), and anti-nestin (1:500 dilution; Abcam) antibodies overnight at 4 °C. Secondary antibodies labelled with fluorescein (1:500 dilution, Abcam) were applied for 120 min at room temperature. After incubation with 0.1% 2-(4-amidinophenyl)-6-indolecarbamidine dihydrochloride for 5 min, cells were washed with 10 mM PBS (pH 7.4) three times. Images were captured on a wide-field BX41 upright fluorescence microscope (Olympus). The mean fluorescence intensity of MAP2, CD133 and nestin was analysed using ImageJ software (NIH).
The level of MAP2 mRNA was detected using semi-quantitative PCR. Total RNA was extracted from cells using an RNA Extraction kit (Invitrogen). Based on the manufacturer’s instructions, cDNAs were synthesized from 0.1 mg of total RNA using a PrimeScript™ RT Reagent Kit (Takara). RT was carried out using a Step OnePlus™ Quantitative Real-Time PCR System (Applied Biosystems) with Syber Green PCR Master Mix (Applied Biosystems) based on the manufacturer’s instructions under the following conditions: preliminary denaturation at 95 °C for 3 min, followed by 30 cycles of denaturation at 95 °C for 30 sec, annealing at 56 °C for 45 sec and elongation at 72 °C for 45 sec, with a final elongation step at 72 °C for 10 min using a GeneAmp® PCR System 9700 thermal cycler (Applied Biosystems). The following qRT–PCR primer sequences were used (Genechem Biotech): for the internal control GAPDH forward: 5ʹ-
To analyse the effects of the silencing of EphrinB2 on BMSC differentiation, third-passage BMSCs were divided into three groups: control group, NC group (transduced with negative control lentiviral vector) and shEphrinB2 (transduced with LV-shRNA- EphrinB2), which were all induced by neuronal cell-conditioned differentiation medium containing 60 g/l glucose, 20 g/l basic FGF, 20 g/l EGF and 2% B27 DMEM/F12 (1:1) at a density of 5 × 107 cells/l. The levels of MAP2, CD133 and nestin proteins were detected using cell immunofluorescence and Western blotting. The mRNA levels of MAP2 was detected using semi-quantitative PCR. The morphology of the BMSCs was observed using a BX41 upright fluorescence microscope (Olympus). The mean fluorescence intensity of MAP2, CD133 and nestin was analysed using ImageJ software (NIH).
Cell migration assay
The cell migration assay was performed in a transwell chamber that contained a polyethylene terephthalate track-etched membrane with 8.0-μm pores. Briefly, 0.5-ml suspensions of LV-EphrinB2-BMSCs/shRNA-EphrinB2-BMSCs or control BMSCs at a concentration of 1 × 105 cells/ml were added to the top of the chamber layer. In the bottom chamber, different concentrations of murine hepatocyte growth factor (Peprotech, Rocky Hill, NJ, USA) were used as a chemoattractant. The cells in the transwell chambers were incubated in a humidified tissue culture incubator overnight at 37 °C and an atmosphere of 5% CO2. After 24 h, the cells were fixed and stained with 4% paraformaldehyde and 0.1% crystal violet, respectively. Migrated cells were observed, imaged and counted within three fields using a BX41 upright fluorescence microscope (Olympus).
Western blot analysis
To reveal the mechanisms underlying EphrinB2-associated regulation, phosphorylation of key kinases in the EphB4/EphrinB2 reverse signalling pathway in BMSCs was evaluated following inducing increased levels of EphrinB2. The Western blot assay was performed as described above for EphrinB2. Briefly, membranes were incubated at 4 °C overnight with primary antibodies against intracellular PAK1 (PAK1; 1:1000 dilution; ab131522), phosphorylated PAK1 (p-PAK1; 1:1000 dilution; ab75599), Grb4 (1:1000 dilution; ab80620; all from Abcam) or GAPDH (1:1000 dilution; Santa Cruz Biotechnology). The membranes were then incubated with the corresponding secondary antibodies. The blots were developed using enhanced chemiluminescence Western blotting reagents and visualized using the Bio-Rad Image Lab system (Bio-Rad Laboratories).
Statistical analyses
All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 19.0. (IBM Corp., Armonk, NY, USA). Data are expressed as the mean ± SE. Differences among groups were evaluated by one-way analysis of variance, followed by least significant difference (LSD) tests. A
Results
Ten samples of third-passage cells were examined by flow cytometry. The cells were negative for CD34 and CD45 and positive for CD29 and CD90, which indicated that they were not haematopoietic stem cells. The cultured rat BMSCs conformed to the characteristics of mesenchymal stem cells and were highly pure (Figure 1).
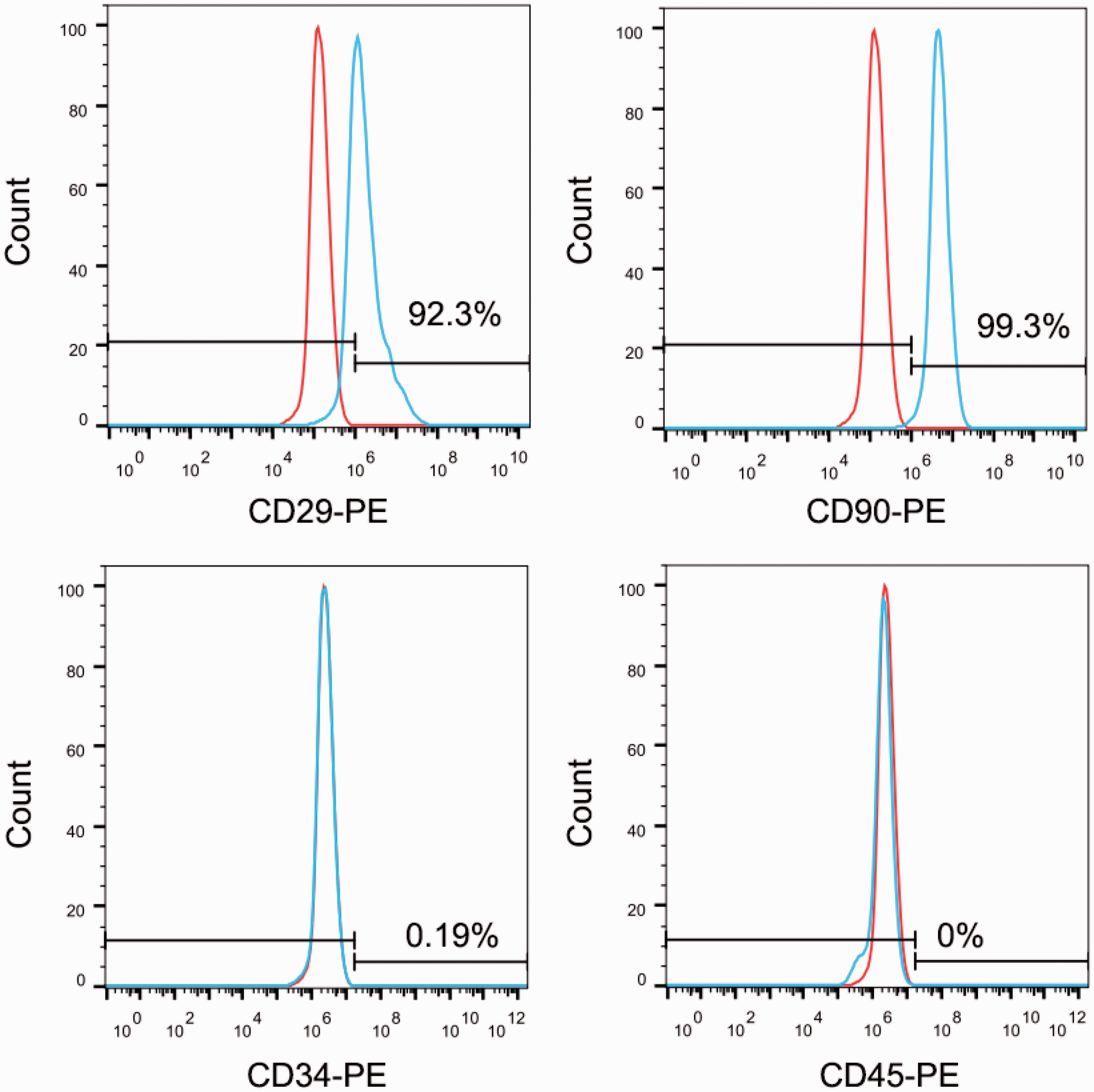
Identification of rat bone marrow mesenchymal stem cells (BMSCs) using flow cytometry. Isolated BMSCs were incubated with fluorescently-labelled antibodies against CD29, CD90, CD34, or CD45, followed by flow cytometry assessment. Cells were positive for the stem cell markers CD29 and CD90 and negative for the haematopoietic stem cell markers CD34 and CD45.
Enhanced green fluorescent protein was observed under an inverted fluorescence microscope and transduction efficiency was determined. Lenti-EphrinB2-BMSCs showed green fluorescence at 24 h, but the number of EGFP-positive cells was small and the fluorescence was weak. At intervals of 1 day, infections were repeated, and the number of EGFP-positive cells increased, leading to enhanced fluorescence at 72 h. Therefore, the levels of EphrinB2 was evaluated at this time.
Quantitative PCR showed that the levels of EphrinB2 mRNA in the NC group were not significantly different from those in the control group (Figure 2A). The transcript levels of EphrinB2 mRNA in lenti-EphrinB2-BMSCs were significantly higher than those in the control and NC group (

Upregulation and knockdown of EphrinB2 in rat bone marrow mesenchymal stem cells (BMSCs) transduced with the lenti-EphrinB2-GFP vector and LV-shRNA-EphrinB2. (A) Quantitative polymerase chain reaction (PCR) demonstrated that the levels of EphrinB2 mRNA were significantly increased in the LV-EphrinB2 group compared with the control (Ctl) and negative control (NC) groups. (B) Western blot analysis demonstrated that the levels of EphrinB2 protein were increased in the LV-EphrinB2 group compared with the Ctl and NC groups. (C) BMSCs were observed using microscopy (magnification ×400) and it was revealed that BMSCs exhibited a neuronal phenotype following transduction with LV-EphrinB2. (D) Quantitative PCR detected that LV-shRNA3-EphrinB2 was the most effective siRNA against EphrinB2. (E) Western blot analysis demonstrated that LV-shRNA3- EphrinB2 was the most effective siRNA against EphrinB2. Western blot analysis revealed that the levels of the EphrinB2 protein were significantly decreased in the LV-shRNA-EphrinB2 group compared with the Ctl and NC groups. (F) BMSCs exhibited a neuronal phenotype in the Ctl and NC groups; however, the majority of BMSCs exhibited no marked morphological changes. Scale bar 50 µm. All data shown represent the mean ± SEM of at least three independent experiments. *
Western blot analysis revealed no significant difference in the levels of EphrinB2 protein between the control and NC groups (Figure 2B). The levels of EphrinB2 protein in the lenti-EphrinB2-BMSCs were significantly higher than those in the control and NC groups (
At a MOI of 20, the number of EGFP-positive cells and fluorescence intensity were optimal, and there was no obvious effect of transduction on the morphology of BMSCs (Figure 2C). The transduction rates in the lenti-EphrinB2-BMSCs were significantly different from those in the control and the NC groups (
Quantitative PCR and Western blot analysis revealed that LV-shRNA3-EphrinB2 was the most effective siRNA against EphrinB2 (Figures 2D and 2E) so it was used for the subsequent investigations. Western blot analysis demonstrated that EphrinB2 protein was detected in the control and NC groups, but not in the LV-shRNA-EphrinB2 groups, suggesting that the shEphrinB2/BMSC line was successfully established. The BMSCs exhibited a neuronal phenotype in the control and NC groups. However, the majority of BMSCs exhibited no marked morphological changes (Figure 2F) in the LV-shRNA-EphrinB2 group.
Three days after EphrinB2 gene transduction, partial retraction of the cytoplasm to the nucleus occurred in the four groups following addition of the inducer. Furthermore, the cell bodies were reduced in size, irregular or rounded in shape, with three-dimensional, surrounding strong refraction. Frequently, several short projections and a longer projection were observed. Neuron-like cells exhibited a typical bipolar, multi-polar and tapered shape with a strong refraction. The neuron-specific markers MAP2, CD133, and nestin were used to stain the induced BMSCs. After adding 5 µg/l FGF and EGF at 28 days after transduction, cell immunofluorescence analysis revealed that the levels of MAP2, CD133 and nestin were significantly higher in lenti-EphrinB2-BMSCs than in the control and NC groups (

Upregulation of EphrinB2 promotes the differentiation of rat bone marrow mesenchymal stem cells (BMSCs) into neuron-like cells. The ability of BMSCs to differentiate into neuron-like cells was significantly enhanced when EphrinB2 was upregulated in BMSCs. (A–C) BMSCs were stained positive for specific neural markers including microtubule-associated protein 2 (MAP2), CD133 and nestin in the LV-EphrinB2 groups under induction conditions as determined using immunofluorescence analysis. (D) Semi-quantitative polymerase chain reaction revealed that the levels of MAP2 mRNA were increased in lenti-EphrinB2-BMSCs compared with the control (Ctl) and negative control (NC) groups. Scale bar 50 µm. All data shown represent the mean ± SEM of at least three independent experiments. *
Following induction by neuronal cell-conditioned differentiation medium, the BMSCs exhibited a neuronal phenotype in the control and NC groups; however, the majority of BMSCs exhibited no marked morphological changes in the LV-shRNA-EphrinB2 group, suggesting that neuron-like cells were successfully induced by neuronal cell-conditioned differentiation medium. Cell immunofluorescence analysis revealed that the levels of MAP2, CD133 and nestin were significantly reduced in the LV-shRNA-EphrinB2 group compared with the control and NC groups (

Ability of rat bone marrow mesenchymal stem cells (BMSCs) to differentiate into neuron-like cells is significantly reduced when the production of EphrinB2 in BMSCs is inhibited.(A–C) Immunofluorescence analysis demonstrated that the levels of microtubule-associated protein 2 (MAP2), CD133 and nestin were decreased in the LV-shRNA-EphrinB2 group compared with the control and NC groups under induction conditions. (D) Semi-quantitative polymerase chain reaction detection revealed that the levels of MAP2 mRNA were decreased in the LV-shRNA-EphrinB2group compared with the control (Ctl) and negative control (NC) groups. Scale bar 50 µm. All data shown represent the mean ± SEM of at least three independent experiments. *
Transwell experiments were conducted to investigate the effect of EphrinB2 on the migration of stem cells (Figure 5A). The mean ± SEM number of migrated cells per field in the lenti-10moi-EphrinB2-GFPgroup (130.18 ± 8.57cells) and lenti-20moi-EphrinB2-GFP group (142.32 ± 6.53 cells) was significantly higher than that in the control group (101.02 ± 4.32 cells) and NC group (102.25 ± 5.08 cells) (

Effect of EphrinB2 on the migration of rat bone marrow mesenchymal stem cells (BMSCs) by activating the p21-activated kinase (PAK) signalling through modulating Grb4. (A) Transwell migration assays of EphrinB2-BMSCs versus control BMSCs (control [Ctl] and negative control [NC]). (B) Transwell migration assays of LV-shRNA-EphrinB2-BMSCs versus control BMSCs (Ctl and NC). (C) Phosphorylation of key kinases in the PAK and Grb4 pathway was investigated using Western blot analysis. All data shown represent the mean ± SEM of at least three independent experiments. *
To obtain further evidence that EphrinB2 is important in BMSC migration,
The protein levels of Grb4, PAK and phosphorylated PAK were upregulated by Ephrin B2 upregulation, suggesting that EphrinB2 can activate the PAK signalling pathway by modulating Grb4 (Figure 5C).
Discussion
This current study investigated the effects of lentiviral-mediated upregulation and knockdown of the
Bone marrow mesenchymal stem cells have the ability to regenerate and differentiate.
10
Furthermore, they are easy to obtain and expand
Recent studies have defined the roles of EphB4/EphrinB2 and shown that their bidirectional signalling is important in neuronal cell differentiation, adhesion and migration and for stem cell transplantation. 5 EphrinB2 is derived from the Eph family, which is the largest subfamily of receptor tyrosine kinases. 3 EphB4 and EphrinB2 are both transmembrane proteins. EphB4 contains three parts: an extracellular region containing an N-terminal ligand domain, which is rich in cysteines and two type III fibronectins; a cell membrane region; and an intracellular region containing a tyrosine kinase domain, a highly conserved SAM domain, and a PDZ domain. 5 EphrinB2 also contains three parts: an intracellular region with a conserved C tail (the binding site for tyrosine kinases) and a PDZ domain, a cell membrane region, and an extracellular region that binds its receptor through glycophosphatidylinositol. 5 EphrinB proteins can also have PDZ-and SH2-independent functions, some of which presumably depend on the SH3 domain of associating proteins (e.g. GRB4).5,11 EphrinB2 binding to the Eph receptor via this extracellular portion activates phosphorylation of the tyrosine kinase binding site on Eph-expressing cells, leading to activation of downstream pathways. 12 Simultaneously, phosphorylation of tyrosine kinase occurs within the cell, and the downstream portion of the pathway is activated for unique bidirectional Eph–Ephrin signalling. 5 This bidirectional signalling can play an important role in cell repulsion and adhesion. 13 Significant progress has been made in understanding the role of EphrinB2 expression during the embryonic stage. 14 A previous study reported the role of EphrinB2 in inducing vascular endothelial cell migration during this stage. 14 Related literature also shows that EphrinB2 can induce the migration of spinal nerve cells at the embryonic stage and promote the development of many organs, including those in the olfactory systems.15–18Another study found that γ-secretase promoted migration by stimulating the reverse EphrinB2 signalling pathway in microglia derived from mouse embryonic stem cells. 19 At present, although there is some controversy over the role of EphrinB2 in angiogenesis and migration, most studies have suggested that the reverse signalling pathway mediated by EphrinB2 promotes invasion and migration of vascular endothelial cells and lumen formation rather than inhibition of these processes. 20 However, the effects of the reverse signalling pathway mediated by EphrinB2 on cell migration and the specific molecular mechanism by which this may occur remain unknown. A study in the adult subventricular zone suggested that EphrinB2 signalling might regulate these processes at the synaptic level. 21 All of these findings indicate that EphrinB2 expression may either directly or indirectly regulate the proliferation, differentiation and migration of neural stem cells in the subventricular zone. 20 EphB4 and its ligand, EphrinB2, play an important role in the development of olfactory bulb cells and migration of cerebellar granule cells. 22
This current study used qPCR and Western blot to analyse the levels of EphrinB2 mRNA and protein in cells from four groups. Compared with the control and NC groups, EphrinB2 mRNA and protein levels were significantly increased in lenti-EphrinB2-BMSCs, confirming that it is possible to select and establish cell lines with stable expression. Western blot analysis demonstrated that EphrinB2 was produced in the control and NC groups, but not in the LV-shRNA-EphrinB2 group, suggesting that the shEphrinB2/BMSC line was successfully established. BMSCs are characterized by the presence of the antigens CD29 and CD90, and by the absence of CD34 and CD45. Thus, flow cytometry was performed to determine whether the cells isolated were BMSCs. The results indicated that 0.19% of cells were CD34+, 0.0% were CD45+, 92.3% were CD29+ and 99.3% were CD90+, confirming that the isolated cells were BMSCs.
In the present study, lentiviral-mediated EphrinB2/sh EphrinB2 transfection into BMSCs was performed and the effect of EphrinB2 on the differentiation of BMSCs was investigated. In previous years, research has demonstrated that BMSCs can be differentiated into neural cells under the action of appropriate inducers and cytokines
To further investigate the role of EphrinB2 in regulating stem cell migration, the current study used a transwell assay to detect cell migration in each group. The number of migrated cells was significantly higher in lenti-EphrinB2-BMSCs than in the control and NC groups. In addition, an inhibitory effect of EphrinB2 suppression by siRNA knockdown on the migration was observed. These results suggest that EphrinB2 can increase the ability of cells to migrate
The EphB4/EphrinB2 reverse signalling pathway has been reported to participate in the differentiation of mesenchymal stem cells into cardiocytes.26,27The SH2/SH3 adaptor protein Grb4 has been implicated as a molecular bridge that connects the tyrosine phosphorylated EB3 cytoplasmic tail with the Dock180 guanine nucleotide exchange factor, Rac activation, and the downstream effector PAK to mediate axon retraction and pruning. 7 To elucidate the mechanism underlying this action, this current study evaluated the phosphorylation of key kinases in the PAK pathway in stably transduced BMSCs. EphrinB2 upregulation activated PAK, suggesting that EphrinB2 might activate PAK by modulating Grb4 in BMSCs. Taken together, these data indicate that EphrinB2 promotes BMSC migration by regulating the EphB4/EphrinB2 reverse signal pathways. At present, little is known about the function of EphrinB2 and the Eph family in stem cells. With the development of molecular biological technologies, research on the molecular mechanism underlying neural stem cell differentiation into neurons will advance. In the future, it may be possible to control the differentiation of neural stem cells artificially and to induce directional differentiation that may allow clinical application of neural stem cells.
In conclusion, EphrinB2 can promote the differentiation and migration of rat BMSCs. The findings of this study suggest that the EphB4/EphrinB2 reverse signalling pathway plays an important role in regulating BMSC migration. In the future, it will be important to study the mechanism of EphB4/EphrinB2 regulation of BMSCs and to establish theoretical and experimental frameworks to identify effective targets of gene therapy.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This study was supported by grants awarded to Min Zhu from the National Natural Science Foundation of China (no. 81401864) and the Science and Technology Development Fund Project of Nanjing Medical University of China (no. 2013NJMU097).
