Abstract
Objective
This study was performed to assess the efficacy of memantine in patients with amnestic mild cognitive impairment (aMCI).
Methods
Thirty healthy controls and 45 patients diagnosed with aMCI based on the Petersen criteria were classified into 3 groups. Group 1 comprised patients who received a single memantine dose following examination (n = 25), Group 2 comprised patients who did not receive memantine treatment following examination (n = 20), and Group 3 comprised healthy age-matched volunteers (n = 30). Neuropsychological testing was performed, and the response to memantine was examined at baseline and at 12, 24, and 48 weeks. Single-photon emission computed tomography was performed at baseline and at 48 weeks in patients who received memantine treatment.
Results
Memantine treatment significantly improved the symptoms of aMCI according to the Wechsler Adult Intelligence Scale-Revised vocabulary subtest, backward digit span, and Blessed Dementia Rating Scale, all of which were recorded for the duration of the study.
Conclusion
These data indicate that patients with aMCI receiving memantine develop an improved semantic memory compared with no treatment. Further studies including larger patient cohorts are necessary to validate these findings.
Keywords
Introduction
Mild cognitive impairment (MCI) is defined as the transitional phase between normal cognitive aging and dementia. This impairment is common, and nearly 19.0% of individuals aged >65 years are affected. Compared with older individuals with normal cognition, patients with MCI have a three- to five-times higher risk of developing Alzheimer’s disease (AD). Petersen’s criteria are frequently used to divide MCI into two groups: the amnestic (aMCI) and non-amnestic (naMCI) forms. Interestingly, aMCI is associated with frequent memory loss and progression to AD.1–3 However, patients with naMCI develop memory loss as well as other cognitive issues, including Lewy body dementia. Both types can be subcategorized; in the present study, however, we did not perform subcategorization because of the limited sample size.4,5 While the US Food and Drug Administration has approved several medications for the treatment of AD, no medications have been approved for patients with MCI. 6
The importance of the glutamatergic N-methyl-D-aspartate (NMDA) receptor in memory and learning processes is well recognized. Memantine, a low-affinity noncompetitive NMDA receptor antagonist, is the only glutamatergic drug approved for the treatment of moderate to severe cognitive symptoms of AD. Recent studies have shown that memantine can also reduce the levels of amyloid β peptides, which inhibit the amyloid β oligomer and improve cognitive performance.7,8 Memantine can usually be used in addition to acetylcholinesterase inhibitors in patients with AD 9 Interestingly, memantine treatment results in slight beneficial effects on memory, activities of daily living, and behavior.
We conducted a prospective open-label study to test the hypothesis that the antiglutamatergic activity of memantine can improve cognitive functioning. Patients with aMCI were treated with and without memantine and compared using neuropsychiatric tests and single-photon emission computed tomography (SPECT).
Material and methods
Participants
The present study was a 48-week, open-label extension study involving 45 patients diagnosed with aMCI and 30 healthy controls who were consecutively examined at the Memory and Dementia Outpatient Clinic of our neurology department.
Volunteers who responded to the advertisements for this study underwent a multistage screening procedure. The inclusion criteria were an age of >55 years (with the exception of seven individuals aged 55–68 years); availability of an informant who could provide information about the participant’s daily function; absence of significant underlying medical, neurological, or psychiatric illness; and willingness to participate in the study procedures. All patients were required to be either cognitively normal or mildly impaired, but without dementia; that is, they were required to have a Clinical Dementia Rating (CDR) of either 0.0 or 0.5.
All patients with MCI experienced memory that deviated from their previous normal function. We based the diagnosis of aMCI on the following criteria established by the International Working Group on Mild Cognitive Impairment 10 :
All participants were evaluated for depression using the 15-item short version of the Geriatric Depression Scale (GDS), in which a total score of >5 indicates depression. Neuropsychological testing was performed at baseline (week 0) and at 12, 24, and 48 weeks.
In total, 75 participants were included in the study (Group 1, n = 25; Group 2, n = 20; Group 3, n = 30). Group 1 comprised patients diagnosed with aMCI who received memantine. They initially received memantine at 5 mg once daily, and this was increased weekly by 5 mg/day in divided doses to a total dosage of 20 mg/day. Group 2 comprised patients diagnosed with aMCI who received no treatment. Group 3 comprised healthy controls without aMCI.
The exclusion criteria were as follows: probable or possible AD; the presence of other neurodegenerative conditions, such as parkinsonian, frontal, vascular, or metabolic dementia; a history or diagnosis of other neurologic diseases, such as stroke or hydrocephalus; a primary psychiatric diagnosis, such as depression or schizophrenia; the presence of sedating medications at the time of testing; and a metabolic or systemic disorder that might influence cognitive performance.
All participants underwent magnetic resonance imaging or brain computed tomography examinations, medical and neurological examinations, and neuropsychological testing by the same researchers. The following laboratory tests for dementia were requested: complete blood counts, blood chemistry, serum vitamin B12 and folic acid levels, thyroid function tests, and syphilis serology.
SPECT was performed at baseline and at 48 weeks in patients receiving memantine treatment.
Neuropsychological tests
The Mini-Mental State Examination (MMSE) 11 was applied to all patients and controls by an experienced neurologist to evaluate the status of cognitive decline. All patients underwent neuropsychiatric evaluation including the digit span (forward and back), Wechsler Memory Scale subtests (immediate word recall list, delayed word recall, delayed word recognition, visual copy, and visual memory), 12 Wechsler Adult Intelligence Scale-Revised (WAIS-R) vocabulary subtest, Boston Naming Test (BNT), 13 clock drawing test, 14 verbal fluency, Blessed Dementia Rating Scale (BDRS), 15 Instrumental Activities of Daily Living scale,16,17 CDR, 18 and GDS. 19
The same person implemented the neuropsychological tests for all patients. Sessions were conducted in the morning in a quiet room and lasted for 45 to 60 minutes.
Perfusion SPECT imaging
Regional cerebral blood flow was studied at baseline using SPECT. Patients received an injection of technetium-99 m ethyl cysteinate dimer and rested for 1 h in a quiet environment with their eyes closed. SPECT image acquisitions were performed using a double-head rotating gamma camera (Siemens) equipped with a fan beam collimator. SPECT was carried out at baseline and at 48 weeks in patients receiving memantine treatment.
Semantic memory
WAIS-R, word list memory, and recall
Semantic memory was assessed with the WAIS-R and word list memory recall. Free recall and recognition were assessed with a word list comprising 10 unrelated concrete nouns. Unit-weighted composite scores were computed based on vocabulary and general knowledge tasks.
Ethics statement
All patient examinations were conducted in full compliance with the Helsinki Declaration. All patients were informed before providing written consent, and the local ethics committee approved the research.
Statistical analysis
Statistical analyses were performed using SPSS for Windows 13.0 and Sigma Stat 3.1. The Shapiro–Wilk test was used to verify data normality assumptions. For normally distributed data, the paired sample t-test and one-way analysis of variance were used for further analyses. For non-normally distributed data, the Wilcoxon t-test and Kruskal–Wallis test were used. For multiple comparisons, Dunn’s method and Tukey’s test were performed. A p-value of <0.05 was considered significant.
Multiple linear regression analysis was performed to identify independent associations of the cerebral SPECT values by including the parameters correlated with cerebral SPECT perfusion in the bivariate analysis. Standardized b regression coefficients and their significance according to the multiple linear regression analysis were reported. A p-value of <0.05 was considered statistically significant.
Results
Demographic characteristics of the study groups.

Data are presented as mean ± standard deviation or n.
Baseline neuropsychologic test scores in patients with MCI.

Data are presented as mean ± standard deviation (median).
Weekly differences in neuropsychologic tests among the study groups.
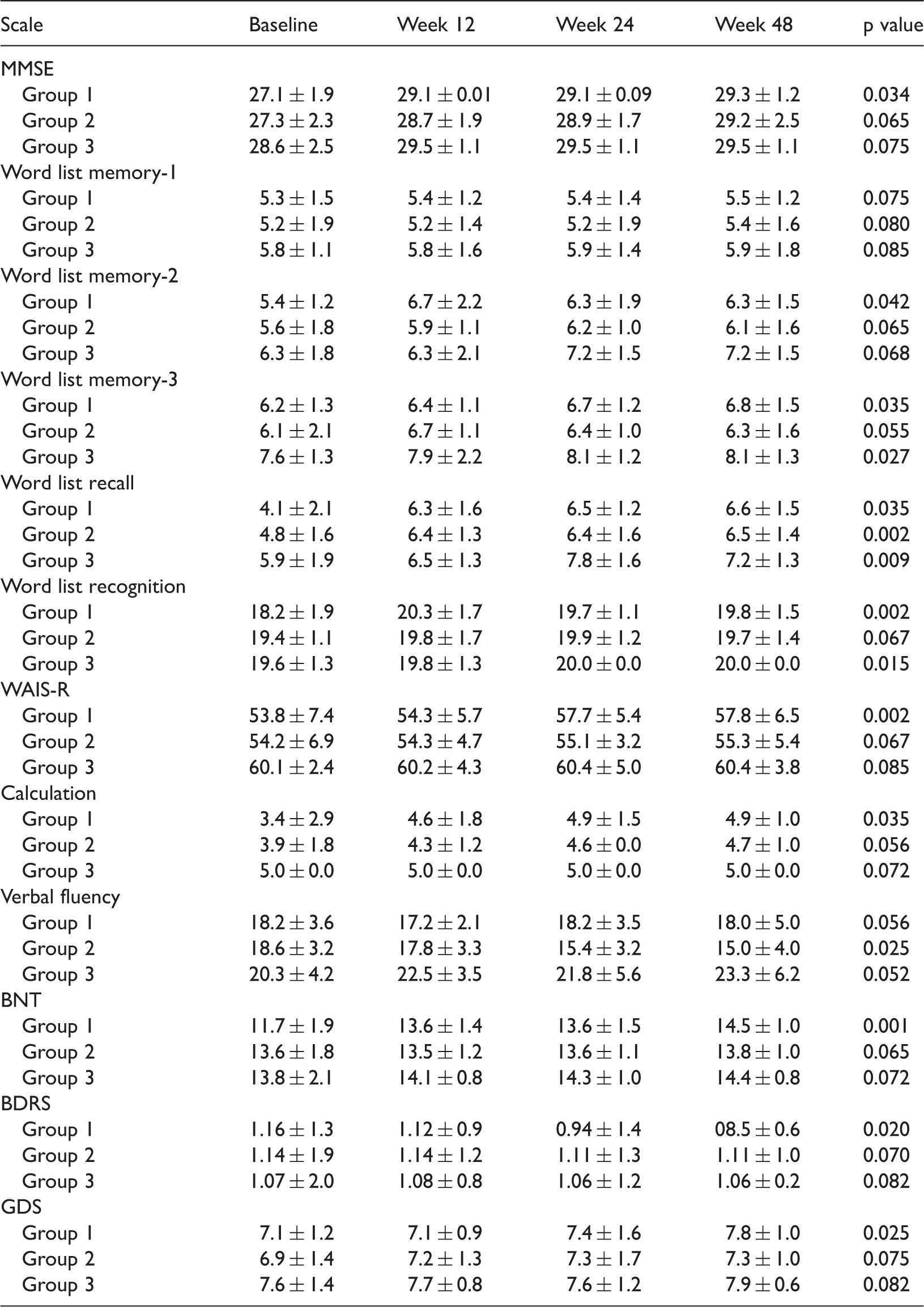
Data are presented as mean ± standard deviation
The MMSE scores were higher at week 24 than at week 12 in Groups 1 and 2. At week 24, Group 1 showed significantly higher scores for word list memory-3, word list recall, WAIS-R, backward digit span, BDRS, and GDS compared with baseline. Group 2 showed a statistically significant decrease in verbal fluency scores at week 24 (Table 3).
At week 48, the significant increase in the word list memory-3, word list recall, WAIS-R, backward digit span, BDRS, and GDS scores continued. No differences were observed in the other test scores among the three groups (Table 3).
Semantic memory composite Z-score
Z-scores were measured for the WAIS-R and word list recall test, which were used for semantic memory. The scores were compared before and after treatment. Z-scores were higher in the memantine-treated group.
Correlation with SPECT perfusion
According to the results of the bivariate analysis, semantic memory was significantly associated with the right superior frontal cortex (r = 0.167, p = 0.020), left superior frontal cortex (r = 0.165, p < 0.001), right medial frontal cortex (r = 0.224, p = 0.002), left medial frontal cortex (r = 0.221, p = 0.024), right inferior frontal cortex (r = 0.211, p = 0.024), left inferior frontal cortex (r = 0.218, p = 0.023), right superior parietal cortex (r = 0.215, p = 0.020), left superior parietal cortex (r = 0.224, p =0.020), right inferior parietal cortex (r =0.218, p = 0.022), left inferior parietal cortex (r = 0.214, p = 0.023), right superior temporal cortex (r = 0.184, p = 0.020), left superior temporal cortex (r = 0.176, p = 0.020), right inferior temporal cortex (r = 0.189, p = 0.011), and left inferior temporal cortex (r = 0.198, p = 0.023). Multiple linear regression analysis showed that the semantic memory Z-score was significantly associated with SPECT perfusion in the right inferior temporal cortex (b = 0.192, p = 0.010).
The multiple linear regression analysis showed that the semantic memory Z-score was significantly associated with SPECT perfusion in the right inferior temporal cortex (b = 0.192, p = 0.010).
Cerebral SPECT values in patients with amnestic mild cognitive impairment treated with memantine (Group 1) (Wilcoxon signed ranks test).

Data are presented as median (25th–75th percentiles).
Discussion
A previous study showed that the rate of progression to clinically diagnosable AD is 10% to 15% per year among persons who meet the criteria for the aMCI, in contrast to 1% to 2% per year among normal elderly persons. 20 Early diagnosis is essential for disease prevention and the development of new treatment strategies. Detection of AD at a very early time point would enable early intervention and a timely start of therapy, possibly preventing disease progression.
Acetylcholinesterase inhibitors have long accounted for a majority of the treatments administered for MCI; this trend follows the cholinergic hypotheses.21–23 Another approach that is widely accepted (at least as widely accepted as the histopathogenesis of AD) is the glutaminergic hypothesis, which is related to the increased effect of glutamate. 24 The discovery that the toxic effect of glutamatergic neurotransmission is present in the very early phases of the disease brought modulatory treatments up to date. In the present study, we examined the effects of memantine on neuropsychological measures for aMCI, improvements in which could be mediated by a neuron-protective effect of memantine.
The current use of memantine aims to treat cognitive and behavioral disorders in patients with mild to moderately severe AD and mild to moderate vascular dementia. No pharmacological agents have obtained regulatory approval for the treatment or prevention of these disorders. A review of published clinical trials indicated that early treatment of hypertension, a risk factor for stroke, reduces the risk of vascular dementia and slows its progression. 25 Because neuronal damage begins in the preclinical phase of the disease, it is believed that neuron-protective treatments should be initiated at very early stages.
The enhancing effects of NMDA receptor antagonists on cerebral blood flow have been demonstrated in experimental ischemic models. In a study of the efficacy of memantine on neuroimaging, glucose metabolism decreased to a lesser degree in the memantine than placebo group.26–28 This finding supports the functional and neuroprotective efficacy of memantine. Increased perfusion in the posterior parietal area, which affects attention and memory as shown by cerebral SPECT studies, has been observed in patients with AD treated with donepezil. Studies using functional imaging techniques have suggested that semantic memory impairment, which occurs relatively early in the course of AD, is caused by neural degeneration in the areas of association in the lateral and inferior temporal regions.29,30
Currently, no drugs are specifically permitted by the US Food and Drug Administration for MCI. 31 Because aMCI may be associated with AD, drug therapies targeting AD may be help to manage MCI.32–34 In one study, the combined treatment of galantamine and memantine had cognitive benefits in the short term, and cognitive decline occurred after discontinuation of galantamine.35,36 Although various studies have examined donepezil, rivastigmine, and galantamine alone or combination with memantine, reliable data on the influence of memantine alone in the treatment of aMCI are lacking.
In one related study involving 270 amnesic patients with MCI, Salloway et al. 37 investigated the effectiveness of donepezil on memory loss by comparison with a placebo group. Although donepezil treatment was not strong enough to affect pure memory test scores (a primary scale of efficacy), it had positive effects on attention, concentration, and psychomotor speed.38,39 Other similar studies have suggested that donepezil treatment improves logical memory at week 24. In a study investigating the role of galantamine in patients with MCI, global rating scales improved and ADAS-cog subscale scores decreased after 6 weeks at all dose levels. 40 In another study by Pelton et al., 41 35 patients received antidepressant and memantine treatment, which was found to be effective on cognition. The dementia conversion rate was significantly lower than in the control group.
In the present study, the recognition, word list memory and recall, WAIS vocabulary subtest, BDRS, and GDS scores differed between memantine-treated patients and controls; these scores decreased until they normalized at week 48. Similar results were observed for the MMSE and global assessment scale, for which the scores in memantine-treated patients increased at week 48 and the significant differences in the scores between these patients and the controls were no longer present. These results suggest that 48 weeks of memantine treatment improved patient functioning until their scores were comparable with those of the healthy controls, whereas this was not the case for patients with untreated aMCI. Indeed, no change was observed in the global cognitive and functional scales of the patients with untreated aMCI. However, the memory test results showed improvement. The fact that fewer significant between-group differences were apparent over time between the patients not receiving memantine and the control group suggests that memory functions improved over time in the former. We found that patients not treated with memantine showed a deterioration of the GDS score. Therefore, memantine may reduce the risk of depression.
Semantic memory deficits are frequently seen in patients with AD, even in the early phase, but not necessarily in patients in the predementia state, as in aMCI. In the present study, we observed a significant improvement in the WAIS-R, word list memory, and recall test used to evaluate semantic memory at 48 weeks in patients who received memantine.
Memantine is a noncompetitive receptor antagonist, and it delays the process of dementia by preventing the pathological activation of NMDA receptors. Its neuroprotective effects have been displayed in various neurological disorders. Importantly, memantine reportedly reduces the release of proinflammatory factors in activated microglia.42,43
In patients with aMCI, pathologic damage first occurs in the medial temporal structures, mainly the entorhinal cortex, which causes episodic memory deficits. 44 The present study showed a significant positive correlation between semantic memory and SPECT perfusion of the right inferior temporal region in patients receiving memantine. This finding may be due to the neuroprotective effects of memantine therapy in patients with aMCI.
Ramaswamy et al. 45 observed improvement in memory, core symptoms of post-traumatic stress disorder, and depression in combat veterans with post-traumatic stress disorder after open-label treatment with memantine. One limitation of the present study is the relatively small number of participants. The low prevalence of aMCI restricted our ability to include a greater number of individuals. Another limitation is the duration of the study. A longer-term study would have allowed us to better observe the efficacy of memantine and determine which patients did and did not develop progression to AD or dementia.
We found a significant increase in the WAIS vocabulary subtest score when evaluating language functions and semantic memory in our study. SPECT revealed cerebral hyperperfusion in the right inferior temporal region in the memantine-treated patients before and 48 weeks after the start of treatment.
In conclusion, neuropsychological tests and cerebral SPECT imaging showed that memantine may be effective in improving the semantic memory of patients with aMCI. Memantine may inhibit oxidative stress and inflammation during the early stage of the disease. The optimal treatment for patients with MCI is controversial. Importantly, however, our study has shown that memantine can be used for initial therapy of MCI. Future studies should further investigate this topic by including greater sample sizes and performing long-term follow-up.
Footnotes
Ethics and consent
Our Clinical Research Ethics Committee approved the study (13 February 2011-90). All patients provided written informed consent.
Consent for publication
Consent for publication was obtained from all authors.
Data and materials availability
The dataset was available for all authors of the study.
Acknowledgements
The authors thank all participants who took part in this study.
