Abstract
Objective
We previously showed that Toxoplasma gondii infection induces severe neuropathology in the form of oxidative stress, high nitric oxide production, glial activation, and apoptosis. This study examined the association between glia maturation factor-beta (GMF-β) expression, activated astrocytes/microglia, and neuropathology in toxoplasmic encephalitis (TE).
Methods
Mouse brain GMF expression was examined by immunohistochemistry on days 10 and 30 post-T. gondii infection.
Results
Neuropathology of infected mice was associated with increased GMF expression in reactive glial cells and neurons compared with healthy controls. Specific up-regulation of GMF-β expression in glial cells was associated with increased gliosis in TE.
Conclusions
GMF up-regulation in glial cells causes neuronal destruction, suggesting a TE pathological pathway involving GMF-mediated brain cell cytotoxicity. GMF-β may therefore be a good biomarker for disease risk assessment and to estimate host neuropathy after exposure to T. gondii, as well as providing a new therapeutic target. This is the first study to demonstrate the expression of GMF-β in reactive glial cells and its association with neuropathology in TE.
Introduction
T. gondii is a parasite exhibiting compulsory intracellular habitation and the ability to infect warm-blooded animals including humans.1,2 Chronic T. gondii infection proceeds subclinically in immunocompetent individuals, but it can cause severe or even fatal toxoplasmic encephalitis (TE) in immunocompromised patients based on the reactivation of tissue cysts.3–5 Although the pathogenesis of severe neuropathology in TE has been studied for many years, it has not yet been fully elucidated.
Glia maturation factor (GMF) was first identified in the bovine brain and later purified and sequenced.6–8 It is mainly located in microglia and some neurons of the central nervous system (CNS), although it is also expressed in astrocytes and oligodendrocytes.9,10
The excessive release of GMF induces proinflammatory reactions, so GMF acts as an inflammatory mediator causing neuronal death in the development of neurodegenerative and demyelinating diseases.9,11,12 13,14 It also plays a key role in the regulation, progression, and disease pathogenesis of CNS immune responses through the production of proinflammatory cytokines and chemokines.11–13 GMF-deficient mice were previously observed to stop the disease progression of experimental autoimmune encephalomyelitis and multiple sclerosis.15,16 Moreover, the up-regulation of GMF expression is thought to contribute to the neuropathology of Alzheimer’s disease.9,17 Similar studies show that GMF play a critical role on the immune regulation, progression and disease pathogenesis of CNS inflammations.
The aim of this study was to investigate the relationship between GMF-β expression and TE-related neuropathology in a mouse model of chronic toxoplasmosis on the 10th (transition from the acute period to the chronic period) and 30th day post-T. gondii infection.
Materials and methods
Ethics statement
This study was performed in strict accordance with the recommendations of the National Centre for the Replacement, Refinement, and Reduction of Animals in Research (NC3Rs). The protocol was approved by the Committee on the Ethics of Animal Experiments of Kirikkale University (Permit Number: 14/15-14/-01.09.01.2014).
Animal infection model and experimental procedures
Animals (female Swiss albino mice; age 12–16 weeks; weight 22–25 g) were obtained from Saki Yenilli Experimental Animal Production Laboratory (Ankara, Turkey) and were continually monitored for survival, clinical condition, and weight until the time of tissue collection. The low-virulence ME-49 strain of T. gondii was used for infection as previously described by Atmaca et al. 18 (2014). Tissue cysts of T. gondii were harvested from the brains of mice that had been intraperitoneally inoculated with 20 tissue cysts. We used 6 mice control groups, 6 mice 10 days after infection, and 6 mice 30 days after infections, total 18 animals in this study. Control group: 6 mice. 10 post-T. gondii infection: 6 mice 30 post-T. gondii infection: 6 mice. Typical symptoms of acute toxoplasmosis, including lethargy, ruffled fur, or hunched posture, were not observed after infection with tissue cysts. Animals are anesthetized before infection. That is, when work begins. Then 10 and 30 days later they were sacrificed. Animals were anesthetized with 20 mg/kg of ketamine and 5 mg/kg of xylazine. Mice were then perfused transcardially with saline solution followed by formalin solution using a peristaltic pump. Animals. six animals in each groups. Control group: 6 mice 10 post-T. gondii infection: 6 mice 30 post-T. gondii infection: 6 mice. Six healthy mice, sacrificed at the beginning of the study, were used as controls for immunohistochemical analysis. No animals died before euthanasia.
Necropsy and histopathology
Mice were anesthetized with pentobarbital (10 mg/kg) and sacrificed by cervical dislocation. The brains were quickly removed, and the squash smear technique was used to identify T. gondii tissue cysts. Briefly, five small randomly selected pieces of fresh brain tissue from the infected group were placed onto poly-L-lysine-coated glass slides. Sections were fixed in 10% neutral buffered formaldehyde for 1 min, and rehydrated through a graded series of alcohol for 1 min. Mayer’s haematoxylin was applied to the sections for 30 s, which were then washed with distilled water, dehydrated through a graded alcohol series for 1 min, cleared in xylene for 2 min, and mounted on glass slides. Non-stained natural image and then dyed preparations were examined under light microscope. I do the ‘squash smear’ interpretation in 2 ways. Unstained and haematoxylin and eosin (H&E)-stained tissues were analysed.
Sections were fixed in 10% neutral buffered formaldehyde for 48 h and washed under tap water overnight. Following routine tissue preparation procedures, tissue samples were dehydrated through a graded series of alcohol and xylene and embedded in paraffin blocks. Paraffin serial sections were cut at a thickness of 4–5 µm, mounted on glass slides, stained with H&E, and examined under a light microscope (Olympus BX51 microscope equipped with a DP25 camera, Tokyo, Japan). The inflammatory infiltrate of the CNS was scored according to the system proposed by Silva et al. 19 (2010) by counting the total numbers of focal or diffuse inflammatory foci in a sagittal section. Cuffing of blood vessels and inflammatory cell infiltration in the meninges were also analysed. All sections were analysed in a blind manner by two researchers. All analyses were performed at 40 × magnification.
Immunoperoxidase examination
GMF-β expression was examined by immunohistochemistry using a commercial antibody (Thermo Scientific, Waltham, MA, USA). Staining was visualized on 4 - to 5 -µm-thick paraffin sections using an indirect streptavidin/biotin kit (HRP; Thermo Scientific). Tissue sections were placed on adhesive slides, deparaffinized for 5 min in each of three xylene series, and rehydrated in a graded alcohol series and distilled water. Antigen retrieval was accomplished by boiling sections on glass slides in citrate buffer (pH 6.0; Thermo Scientific) for 20 min. Endogenous peroxidase activity was quenched in 3% hydrogen peroxide in absolute methanol for 7 min at room temperature. Sections were rinsed three times with phosphate-buffered saline (pH 7.4) for 5 min between each step of the test. Sections were incubated in blocking serum for 5 min to prevent non-specific binding. Thereafter, they were incubated with the primary antibody (Thermo Scientific) for 60 min in a humidified chamber at room temperature. Sections were treated with a biotin-labelled secondary antibody (Thermo Scientific) for 15 min and streptavidin-peroxidase enzyme for 15 min at room temperature. Finally, sections were incubated in aminoethyl carbazole chromogen (Thermo Scientific) for 5–10 min to induce the colour reaction. Mayer’s haematoxylin was applied as a counterstain for 1–2 min. Thereafter, sections were mounted with water-based mounting medium (Thermo Scientific). As a control for non-specific endogenous peroxidase and biotin activities in each test, the primary antibody step was omitted. Immunostaining was evaluated using a binocular microscope and photographed under a 20 × objective lens.
Histomorphometric analysis and statistics
The density of positive staining was measured using a computerized image system composed of a Leica CCD camera DFC420 (Leica Microsystems Imaging Solutions, Ltd., Cambridge, UK) connected to a Leica DM4000 B microscope (Leica Microsystems Imaging Solutions, Ltd.). Five representative fields were selected and consecutive pictures were captured by Leica QWin Plus v3 software under a 20 × objective lens (Leica Microsystems Imaging Solutions) at a setting identical to the image system for analysing. We used the same setting for all slides. The integrated optical density of all GMF-β-positive staining was measured, and the mean GMF-β-positive area/total area was calculated by Leica QWin Plus v3. All images were collected under the same lighting conditions. To avoid observer bias, a blinded investigator quantified all sections. Data were described in terms of mean and standard deviation (mean ± SD) for area %. After calculating the proportion (% pixels) of stained area to the whole field, the mean (in % pixels) staining area for each slide was determined. GMF-β immunohistochemical results were compared between groups using a one-way analysis of variance and Tukey’s multiple comparison test. All statistical analyses and graphs were prepared using GraphPad Prism version 6.0 (GraphPad Software, La Jolla CA, USA). P < 0.05 was considered statistically significant.
Results
Histopathological findings
H&E-stained brain sections from control group animals exhibited normal architecture, but tissue cysts were observed in the 30-day post-infection groups (Figure 1(a)). Neurohistopathological changes included nonsuppurative and/or necrotizing non-purulent meningitis, neuronal degeneration (Figures 1(a),(b)), focal gliosis (Figure 1(b)), hyperemia, perivascular mononuclear cell infiltration, and shrunken Purkinje cells/neurons with a highly dense eosinophilic and necrotic appearance. Glial proliferation was observed throughout the brain. The difference between the findings on days 10 and 30 after infection was significant. Represents values (10 DAI) significantly lower than the 30 days after inoculation (DAI) group and Represents values (30 DAI) significantly higher than 10 DAI.
Means a gradual increase in harmony between And this rise is statistically significant (Figure 2).
Histopathology of toxoplasmic encephalitis stained by haematoxylin and eosin. (a) Toxoplasma gondii tissue cysts (arrowheads). Scale bar, 50 µm (b) Necrotic/degenerative neuronal cells (arrowheads) and severe gliosis focus (arrow). Scale bar, 50 µm
Immunoperoxidase findings
GMF-β expression was shown to be significantly higher in the brain of T. gondii-infected animals on days 10 and 30 post-infection than in non-infected control animals (day 10: 3.853 %, day 30: 5.420%; P < 0.005) (Figure 2).
Comparison of GMF-β immunopositivity between infected and control groups. Significant differences represented by letters: ‘a’ represents values significantly higher than the control group; ‘b’ represents values significantly lower than the 30 days after inoculation (DAI) group; ‘c’ represents values significantly higher than 10 DAI
GMF-β expression
Fairly weak immunoreactivity for GMF-β was observed in some glial/neuronal cells in healthy control animals (Figures 3(a), (b)). However, GMF-β expression was significantly increased in vascular endothelial cells (Figures 4(a),(b)), some necrotic/healthy neurons (Figure 5(a)), and especially glial cells (Figures 5(c),(d),(e),(f)) of T. gondii-infected animals (P < 0.005).
GMF-β immunoreactivity in the healthy control group. (a, b) Low expression of GMF-β in some glial cells. Immunohistochemistry method. (Streptavidin- biotin) (anti-GMF-β), Mayer’s haematoxylin counterstain. Scale bar, 100 µm GMF-β immunoreactivity in toxoplasmic encephalitis. (a) Strong expression of GMF-β in endothelial cells (boxed) and perivascular infiltrating macrophages (arrow). Dense reactive bands accumulated around and between neuronal cells (arrowheads). ABC technique (anti-GMF-β), Mayer’s haematoxylin counterstain. Scale bar, 100 µm. (b) High magnification of boxed region in (a). Strong expression of GMF-β in endothelial cells (arrow) and perivascular infiltrating macrophage (arrowhead). ABC technique (anti-GMF-β), Mayer’s haematoxylin counterstain. Scale bar, 50 µm GMF-β immunoreactivity in toxoplasmic encephalitis. (a) Strong expression of GMF-β in neuronal cell (boxed), and endothelial and glial cells (arrowheads). ABC technique (anti-GMF-β), Mayer’s haematoxylin counterstain. Scale bar, 50 µm. (b) Strong expression of GMF-β in perivascular infiltrating mononuclear cells (boxed) and highly reactive gliosis focus (arrow). ABC technique (anti-GMF-β), Mayer’s haematoxylin counterstain. Scale bar, 50 µm. (c–f) Strong expression of GMF-β in microglia/macrophages (arrowheads) and glial cells. ABC technique (anti-GMF-β), Mayer’s haematoxylin counterstain. Scale bar, 50 µm
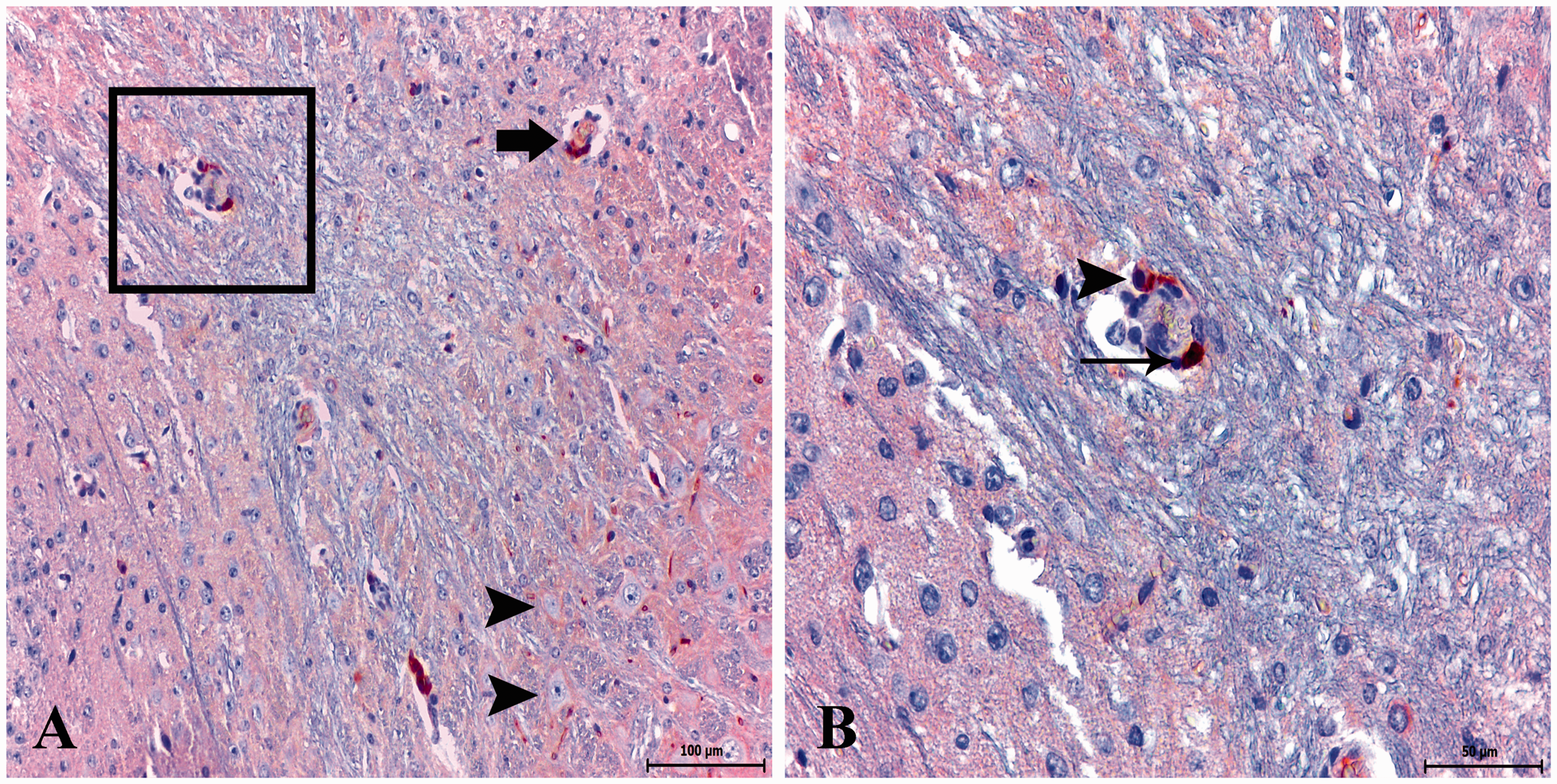

GMF-β expression was markedly increased in infiltrating mononuclear cells (gliosis focus and perivascular space) and infiltrating microglia/macrophages of the meninges on days 10 and 30 post-infection (Figure 4(a), Figure 5(b)).
GMF-β immunoreactive staining areas were also scattered in the intercellular matrix between the neurons (Figure 4(a), Figure 5(b)), and as dense reactive bands at the periphery of the neurons (Figure 4(a)).
Discussion
TE occasionally exhibits a fatal course when tissue cysts reactivate, as observed in immunosuppressive individuals infected with T gondii.3–5 However, because this TE-induced neuropathogenesis has not previously been fully investigated, its severity is only now being identified. We previously showed that tachyzoites and bradyzoites are not the only agents responsible for TE neuropathology, but that high nitric oxide (NO) production, induced oxidative stress (OS), destruction of the blood–brain barrier, and internally- and externally-induced apoptosis also play important roles.20–22 In this study, we reported increasing expression levels of GMF-β on days 10 and 30 in mice infected with T. gondii compared with healthy controls. This enhanced GMF-β expression was observed in reactive glial cells, particularly in glial foci, suggesting that the TE neuropathology is mediated by GMF-β.
GMF has a high level of oxidase activity and causes the formation of reactive oxygen species (ROS).23–25 Moreover, GMF-β plays an important role in oxidation/inflammation-induced cell death,14,23,24 as well as the apoptosis of renal proximal convoluted tubule epithelial cells through increased sensitization to OS. 23 GMF-null astrocytes also demonstrate increased antioxidant activity and reduced ROS production and lipid peroxidation, 24 indicating that GMF is key to the formation of OS-associated pathology. In previous studies, we reported that OS and apoptosis play important roles in TE neuropathology,20,22 while this state was associated with the overexpression of GMF-β in the present study. Thus, it appears that GMF-β contributes to the OS-mediated neuropathology and apoptosis observed in TE.
GMF was previously shown to induce interleukin (IL)-33 expression, which in turn leads to tumour necrosis factor (TNF) release and the production of NO, causing neuronal degeneration. 26 Other studies reported a significant increase in IL-33 expression in TE and ocular toxoplasmosis, and the IL-33/ST2 pathway was thought to be associated with the regulation of ocular immunopathology triggered by T. gondii.27,28 Pathological levels of NO were also demonstrated to cause TE neuropathology, in association with significantly enhanced TNF-α activity levels.21,29–31 Based on our present findings, we hypothesize that the increased expression of GMF exhibited in TE models is an important inducer of IL-33 expression, and acts as a bridge in the neuropathogenesis caused by increased NO and TNF-α expression.
T. gondii infection has been reported to cause schizophrenia,32,33 and the pathogenesis of this disease shares many similarities with TE. Both disorders present with elevated OS and NO production21,22,34,35, decreased activity of the antioxidant enzyme superoxide dismutase,22,36 reduced expression of the anti-apoptotic protein Bcl-2,37,20 and early apoptosis20,38,39. These similarities may be more than coincidental. Indeed, cytokine-mediated neuronal injuries are considered to contribute to the pathophysiology of schizophrenia, causing a decrease in neuronal number and density. 40 Additionally, increases in glial cell number and density, shown in this study to be associated with TE, were also previously observed in schizophrenia.41–43 In the present study, when compared with healthy control groups, mice at 10 and 30 days post-T. gondii infection showed elevated GMF expression, particularly in glial cells, triggering proinflammatory reactions. The most striking finding of this study was the establishment of a previously unidentified correlation between T. gondii and schizophrenia. Taking all these findings into account, increased GMF expression appears to be important in the pathogenesis of schizophrenia caused by T. gondii.
The present study also showed that GMF is a potential CSF biomarker for TE as an indicator of T. gondii-related neuropathy. As such a biomarker, it could be used to estimate host neuropathy after exposure to T. gondii, as well as the severity of acute/chronic TE, and to identify optimal treatment.
In conclusion, TE studies suggest that CNS neuropathology is a complicated process. Our findings implicate GMF as a key mechanism in initiating cytotoxicity through a novel proinflammatory pathway, leading to the pathogenesis of TE. This is the first report to reveal that GMF expression, as a mediator of CNS inflammation, is abnormally increased in TE. We propose that GMF could be used as a novel biomarker to inform of the severity of TE and to predict the disease course. This study showed that neuropathology seen in toxoplasmic encephalitis may be mediated by GMF-β in a very large proportion. Therefore, GMF-β-related pathology must be well-defined for this disease. Moreover, GMF inhibitor-based therapies could be used in TE clinical applications to prevent CNS inflammation.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
