Abstract
Objective
To determine the effect-site concentration (Ce) of remifentanil target-controlled infusion required for a smooth inhalational induction without airway irritation using desflurane in a stepwise incremental manner for 50% of patients (EC50) and 95% of patients (EC95).
Methods
Patients with an American Society of Anesthesiologists physical status I and II, aged 19–60 years undergoing elective surgery were enrolled in this study. When target Ce of remifentanil was reached, desflurane was inhaled at 4 vol% initially and then it was increased to 8 and 12 vol% at intervals of 30 s. Smooth induction was regarded as an absence of airway irritation signs and excitatory movements. The EC50 and EC95 values for remifentanil were determined using a modified Dixon’s up-and-down method as well as an isotonic regression method with a bootstrapping approach.
Results
The EC50 and EC95 of remifentanil for smooth induction during inhalation of desflurane were 3.40 ng/ml (95% confidence interval [CI] 2.42, 4.38 ng/ml) and 4.31 ng/ml (95% CI 2.15, 5.98 ng/ml), respectively.
Conclusion
Prior administration of remifentanil could provide smooth inhalational induction with desflurane in a stepwise increment.
Introduction
Desflurane has the lowest blood–gas partition coefficient among all inhalation anaesthetics. 1 Therefore, it has been predicted that desflurane is suitable for rapid induction and recovery from general anaesthesia.2,3 However, the pungent nature of desflurane limits its use in inhalation induction because it can cause a high incidence of airway irritation, including breath holding, coughing, excessive secretion, laryngospasm, and excitatory movements.4,5
Opioids are effective in reducing the airway irritation caused by desflurane.4–7 Preadministration of an opioid before desflurane has been reported to be able to make the transition from being awake to loss of consciousness (LOC) smooth and feasible without causing airway irritation.4,5,8,9 Remifentanil is an ultrashort-acting opioid; the onset time is very rapid and the context sensitive half-life is 3–5 min.10,11 Therefore, it is easy to titrate and adjust remifentanil to obtain its optimal dose without the concern of delayed recovery. Two studies have reported airway irritation-free results following the administration of remifentanil prior to desflurane being used for inhalation induction.8,9 The desflurane used in these previous studies was limited to 1 minimum alveolar concentration (MAC) of the dial of the vapourizer,8,9 implying that the expiratory concentration was lower than 1 MAC, which means the actual concentration of desflurane was lower than 1.0–1.5 MAC, the threshold for airway irritation.12,13 Therefore, the previous airway irritation-free results could be due to the low MAC of desflurane used in these studies,8,9 regardless of the prior administration of remifentanil. In addition, the use of a low MAC of desflurane cannot guarantee the loss of consciousness under the potent stimuli of intubation. Therefore, we hypothesized that the effect of remifentanil on airway irritation could work on desflurane induction in a stepwise incremental manner using a higher concentration of desflurane, which is more common for inhaled induction to ensure that LOC is achieved in patients. The objective of this study was to determine the effect-site concentration of remifentanil target-controlled infusion required for a smooth inhalational induction without airway irritation using desflurane in a stepwise incremental manner for 50% of patients (EC50) and 95% of patients (EC95).
Patients and methods
Patient population and study design
This prospective observational study enrolled consecutive patients who met the following criteria at the Department of Anaesthesiology and Pain Medicine, Ajou University Hospital, Suwon, Republic of Korea between January 2015 and August 2015. The inclusion criteria were as follows: (i) American Society of Anesthesiologists physical status I and II; 14 (ii) aged 19–60 years; (iii) patients who were undergoing elective surgery under general anaesthesia. The exclusion criteria were as follows: (i) suspected difficult airway; (ii) known history of reactive airway disease. The study protocol was approved by the Institutional Review Board (no. AJIRB-MED-CT4-14-327) of Ajou University Hospital, Suwon, Republic of Korea. It was registered at ClinicalTrials.gov (NCT 02379715) and written informed consent was obtained from all patients.
Anaesthesia induction methods
Venous access was obtained from all patients using a 20 G intravenous catheter inserted into the forearm whilst the patient was on the ward. Patients received no premedication. Pulse oximetry, electrocardiography, and noninvasive blood pressure were performed for patients after they arrived at the operating room. Remifentanil (40 µg/ml solution) was loaded into a target-controlled infusion device (Orchestra® Base Primea; Fresenius Vial, Brézins, France) using the pharmacokinetic model of Minto et al. 15 Inspiratory and expiratory concentrations of desflurane were measured using a gas analyser within an anaesthesia ventilator (Dräger Primus®; Drägerwerk, Lübeck, Germany).
After patients were preoxygenated with 100% oxygen at 4 l/min using a face mask for 3 min, infusion of remifentanil was commenced. If the respiratory rate was <8 breaths/min, the patient was encouraged to breath. Their chest wall rigidity (chest tightness and difficulty in breathing) accompanied by desaturation (< 95%) was assessed for active management. When the target effect-site concentration of remifentanil was reached, the vapourizer of desflurane was dialled at 4 vol% initially. The concentration of desflurane was then increased to 8 and 12 vol% at intervals of 30 s. The concentration of desflurane was also calculated as a MAC value. The MAC value was the age-corrected MAC value of each patient calculated by Mapleson’s method. 16 Each patient was asked to open his or her eyes every 10 s during inhalation of desflurane. The time when the patient did not respond to this command was regarded as being when the patient had achieved LOC. After LOC was achieved, 0.6 mg/kg rocuronium was administered via intravenous (i.v.) injection. Tracheal intubation was then performed 90 s after the administration of 0.6 mg/kg rocuronium i.v. If airway irritation signs, such as breath holding, coughing, laryngospasm and excitatory movements (head movement, limb movement, verbal/forceful removal of the mask by the patient), developed during the induction period, the dial of the vapourizer of desflurane was then set to zero. In addition, 4 mg/kg thiopental i.v. and 0.6 mg/kg rocuronium i.v. were administered immediately. After that, intubation and routine anaesthesia were continued. Breath holding was considered no breathing movements for > 30 s. Laryngospasm was defined as complete airway obstruction associated with decrease in oxygen saturation for > 20 s. Regardless of the severity of the airway irritation sign, the presence of irritability was considered as a failed state and the onset time of airway irritation signs, inspiratory and end-tidal concentrations of desflurane were recorded.
The LOC time was defined as the time from the start of desflurane inhalation via a face mask to the point that the patient did not respond to the verbal command. The LOC time, inspiratory and end-tidal concentrations of desflurane at the LOC point were recorded. Haemodynamic data were recorded at baseline (T0), when the target effect-site concentration of remifentanil was reached (T1), 1 min after desflurane inhalation (T2), and at LOC (T3). In the case of bradycardia (heart rate < 45 beats/minute [bpm]) or tachycardia (heart rate > 130 bpm), 0.5 mg atropine i.v. or 0.2 mg/kg esmolol i.v. were administered, respectively. Hypertension and hypotension were treated at the discretion of the attending anaesthesiologists (J.Y.Y., Y.J.C., and S.Y.L.). The investigators (H.W.J. and H.B.P.) who conducted the inhaled induction of desflurane and assessed the complications and airway irritation were blinded to the effect-site concentration of remifentanil.
The effect-site concentration of remifentanil for preventing airway reactivity was determined using a modified Dixon’s up-and-down method.17,18 The initial effect-site concentration of remifentanil was 4 ng/ml. The next target effect-site concentration of remifentanil was determined based on the response of the previous patient. A successful response was regarded as the absence of breath holding, coughing, laryngospasm, or excitatory movements. If any of these symptoms occurred, it was regarded as a failure and the next target effect-site concentration of remifentanil was increased by 0.5 ng/ml. Conversely, if the desflurane induction was successful, the next target effect-site concentration of remifentanil was decreased by 0.5 ng/ml.
Statistical analyses
The patients were enrolled in this study until eight crossover pairs were obtained according to Dixon’s sequential allocation method. 18 The sample size was decided based on prior literature that demonstrated that the modified Dixon’s up-and-down method required more than the minimum six crossover points to minimize the inaccuracy of the individual study. 19 Thus, data from eight independent crossovers of patients were collected for this study. The EC50 of remifentanil was defined as the mean value of independent crossover pairs (i.e. failure to success of smooth induction). The R statistical software package version 3.3.3 (R Foundation for Statistical Computing, Vienna, Austria) was used for calculations using the isotonic regression method to estimate EC50 and EC95 along with 95% confidence interval (CI). The CI was estimated using the bootstrapping approach. 20 Other statistical analyses were performed using the SPSS® statistical package, version 11.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Comparisons between the success and failure groups were analysed using independent t-test, χ2-test, Fisher’s exact test, or Mann–Whitney U-test where appropriate. Haemodynamic data were analysed with repeated measures of analysis of variance, followed by paired t-test for comparisons across successive time-points. Data were reported as mean ± SD or the n of patients. A P-value < 0.05 was considered statistically significant.
Results
Demographic characteristics of patients undergoing elective surgery under general anaesthesia who participated in this study to examine the effect-site concentration of remifentanil target-controlled infusion required for a smooth inhalational induction without airway irritation using desflurane.
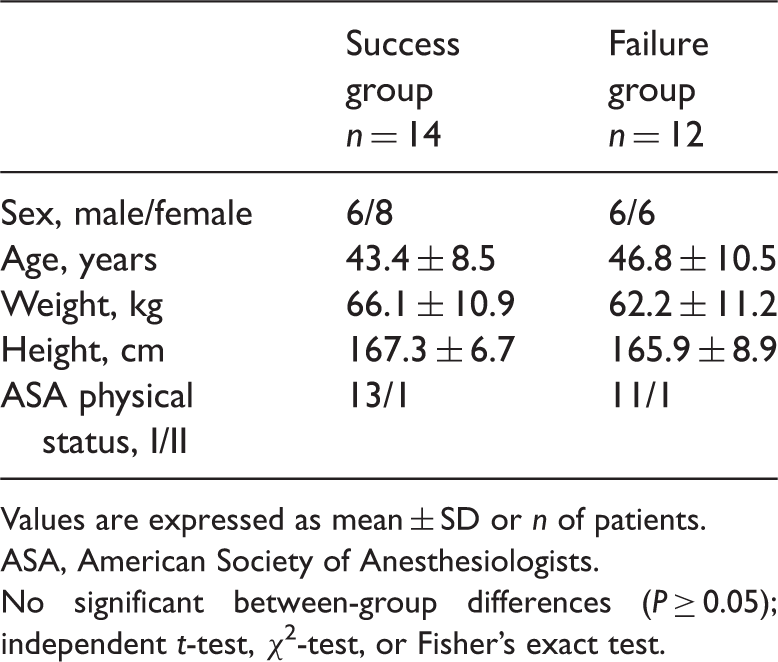
Values are expressed as mean ± SD or n of patients.
ASA, American Society of Anesthesiologists.
No significant between-group differences (P ≥ 0.05); independent t-test, χ2-test, or Fisher’s exact test.
Anaesthesia induction characteristics of patients undergoing elective surgery under general anaesthesia who participated in this study to examine the effect-site concentration of remifentanil target-controlled infusion required for a smooth inhalational induction without airway irritation using desflurane.
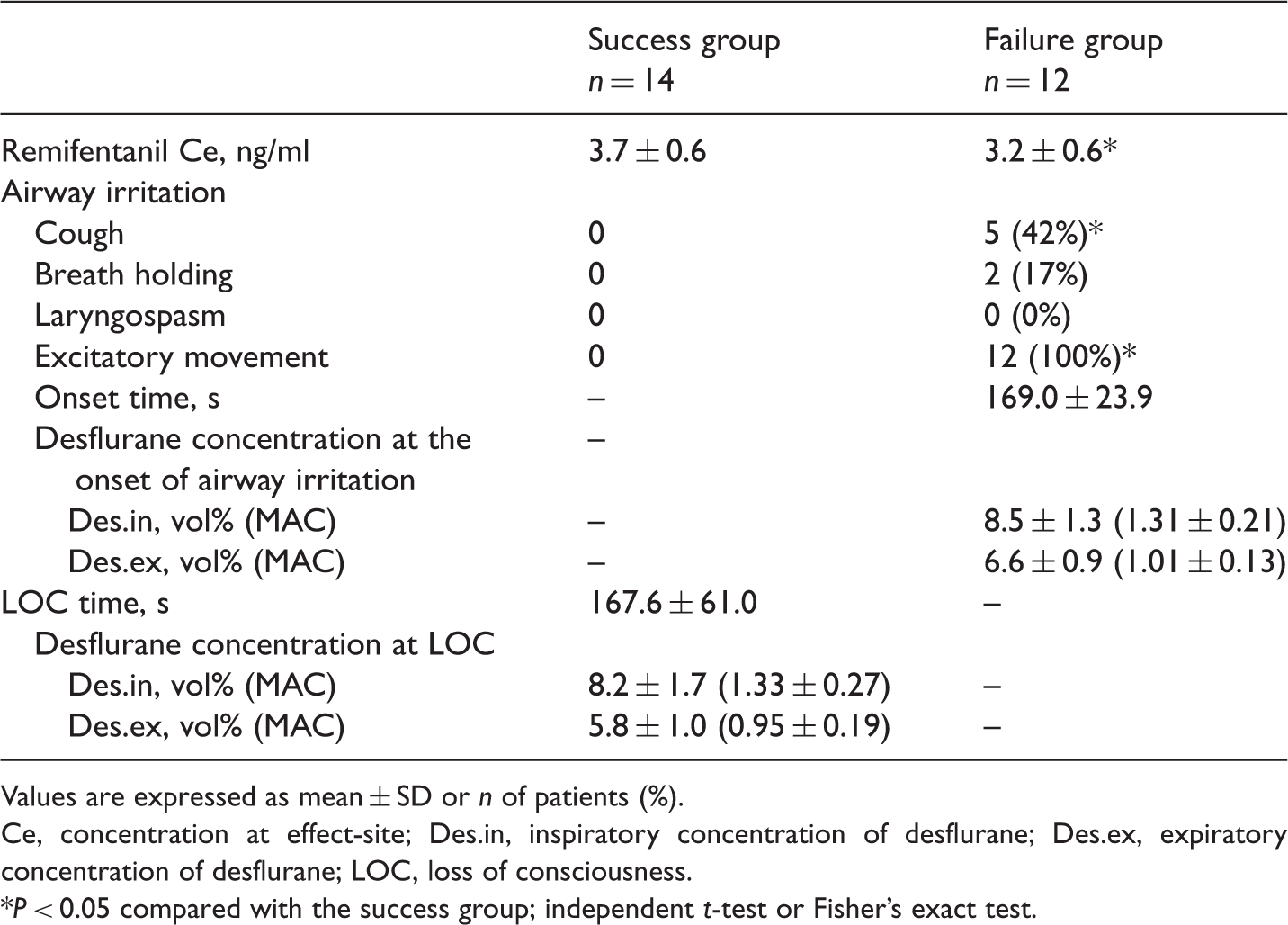
Values are expressed as mean ± SD or n of patients (%).
Ce, concentration at effect-site; Des.in, inspiratory concentration of desflurane; Des.ex, expiratory concentration of desflurane; LOC, loss of consciousness.
P < 0.05 compared with the success group; independent t-test or Fisher’s exact test.
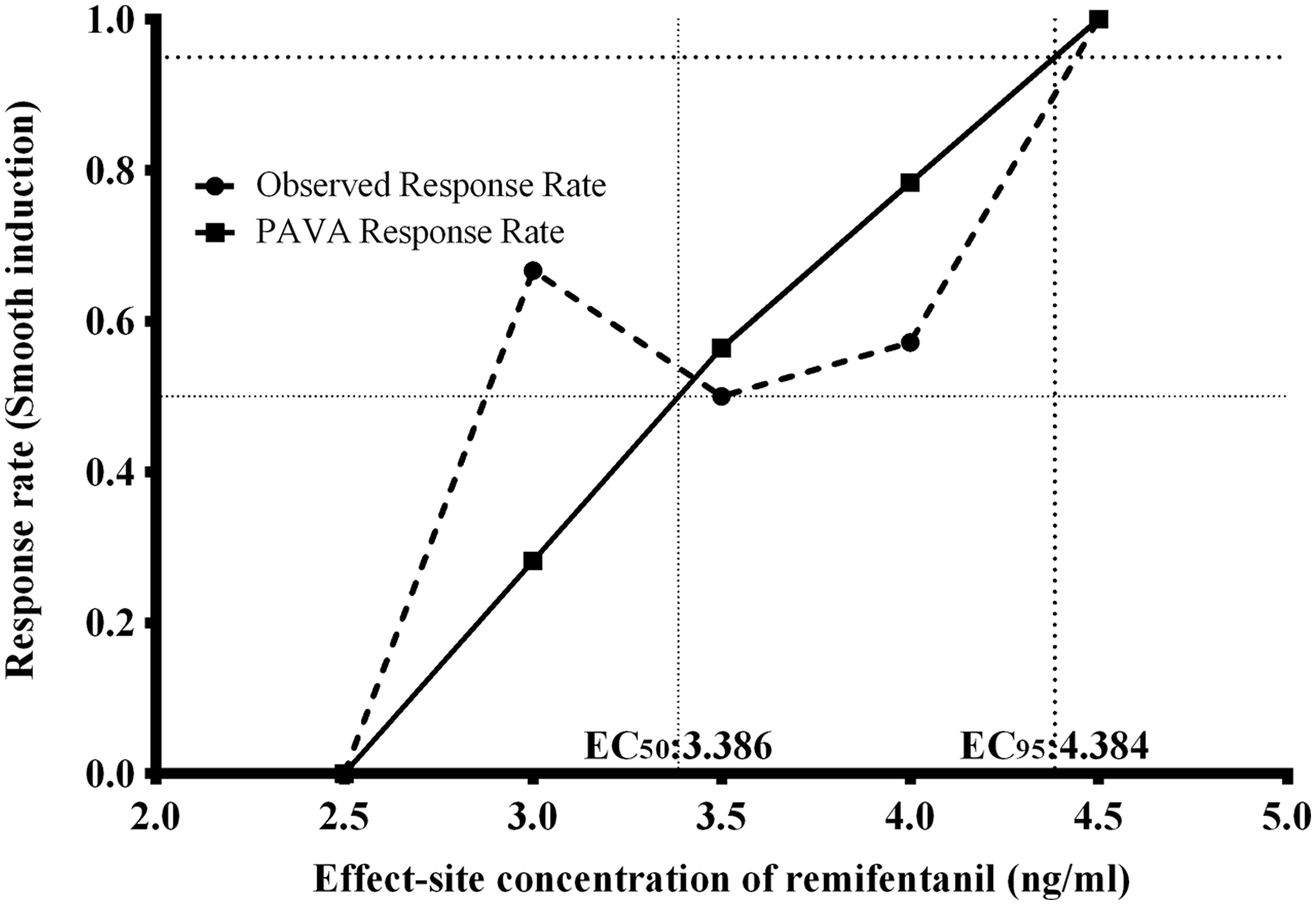
Individual data of concentration-response within the up-and-down sequences are shown in Figure 1. The mean ± SD EC50 of remifentanil for smooth induction without airway irritations during inhalation of desflurane via a modified Dixon’s up-and-down method was 3.56 ± 0.70 ng/ml. The EC50 and EC95 of remifentanil estimated by the isotonic regression method and the bootstrapping approach were 3.40 ng/ml (95% CI 2.42, 4.38 ng/ml) and 4.31 ng/ml (95% CI 2.15, 5.98 ng/ml), respectively (Figure 2).
Consecutive successful or failed smooth induction over predetermined concentrations of remifentanil with desflurane inhalation (initial predetermined concentration of remifentanil was 4.0 ng/ml for the first patient). Eight pairs of failure–success sequences were received for analysis with the modified Dixon’s up-and-down method. The mean ± SD effect-site concentration of remifentanil for smooth induction with desflurane in 50% of patients was 3.56 ± 0.70 ng/ml. Observed and pooled adjacent violators algorithm (PAVA) response rate. The EC50 of remifentanil was 3.40 ng/ml (95% confidence interval [CI] 2.42, 4.38 ng/ml). The EC95 of remifentanil was 4.31 ng/ml (95% CI 2.15, 5.98 ng/ml). EC50, effect-site concentration of remifentanil for smooth inhalational induction with desflurane in 50% of patients; EC95, effect-site concentration of remifentanil for smooth inhalational induction with desflurane in 95% of patients.
Mean arterial pressure and heart rate during inhalational induction of patients undergoing elective surgery under general anaesthesia who participated in this study to examine the effect-site concentration of remifentanil target-controlled infusion required for a smooth inhalational induction without airway irritation using desflurane.

Values are expressed in mean ± SD.
T0, baseline; T1, when the target effect-site concentration of remifentanil was reached; T2, 1 min after desflurane inhalation; T3, at the loss of consciousness.
P < 0.05 compared with baseline value T0; paired t-test.
No significant between-group differences (P ≥ 0.05); repeated measures analysis of variance.
Discussion
The mean ± SD effect-site concentration of remifentanil (EC50) for smooth inhalational induction with desflurane via a modified Dixon’s up-and-down method was at 3.56 ±0.70 ng/ml. The EC50 and EC95 of remifentanil estimated by the isotonic regression method were 3.40 ng/ml (95% CI 2.42, 4.38 ng/ml) and 4.31 ng/ml (95% CI, 2.15–5.98 ng/ml), respectively.
The high pungency of desflurane causes sympathetic stimulation and airway irritation during induction.21,22 Opioids are expected to be an effective adjuvant during inhaled induction of desflurane because they can prevent the drawbacks of both cardiovascular stimulation23,24 and airway irritation.4–7 The current study mainly focused on airway irritation-free smooth transition from being awake to LOC during the inhalation of desflurane without using other intravenous anaesthetics. To date, two studies have reported smooth induction by using preadministration of remifentanil and inhalation of desflurane.8,9 As described earlier, the concentration of desflurane used in these previous two studies was limited to around 1 MAC of the dial of the vapourizer.8,9 The exhaled concentration measured in one report was around 0.7 MAC, 9 which is not enough to cause airway irritation because the threshold of desflurane for irritating the airway is known to be 1.0–1.5 MAC.12,13 Therefore, these previous airway irritation-free results could be due to a low concentration of desflurane regardless of the use of remifentanil. The use of a low concentration of desflurane may not ensure LOC in all patients with the potent stimuli of intubation, although the authors reported successful induction of anaesthesia. 9 In addition, the optimal dose of remifentanil was chosen to blunt the haemodynamic response to intubation, unlike the current study. 9 This present study chose a stepwise incremental manner using a high concentration of desflurane because it would ensure LOC was achieved. Opioids can increase the threshold of airway irritation. 6 These present results revealed that remifentanil made smooth inhalation induction possible when using a high concentration of desflurane. This could make induction of anaesthesia using inhaled desflurane a feasible option in the clinical situation. With regard to cardiovascular stimulation, no severe hypertension or tachycardia was observed in the present study. One previous study reported that 4 ng/ml remifentanil was able to minimize the haemodynamic change caused by inhalation induction using 1.7 MAC desflurane. 25 Thus, the effect-site concentrations of remifentanil observed in the current study of 3.7 ng/ml and 3.2 ng/ml in the success and failure groups, respectively, seemed to be able to prevent cardiovascular stimulation. However, when 4.31 ng/ml (EC95) of remifentanil was used for smooth induction, caution was needed for haemodynamic stability.
The inspiratory and expiratory concentrations of desflurane at LOC in the success group of the current study were 8.2 vol% (1.33 MAC) and 5.8 vol% (0.95 MAC), respectively. It has been reported that the inspiratory and expiratory concentrations at LOC using desflurane alone or with nitrous oxide were 14.1–14.9 vol% and 10.1–10.9 vol%, respectively.26,27 In a study using pretreatment with fentanyl and midazolam, the inspiratory and expiratory concentrations at LOC were 8.9 vol% and 5.3 vol%, respectively. 27 These results suggest that an opioid can decrease the anaesthetic requirement for LOC. The mean ± SD time to LOC was 167.6 ± 61.0 s in this present study. However, the lack of control group means that it is not possible to determine if this time was reduced by the use of an opioid. Whether or not pretreatment with an opioid shortens the LOC time remains controversial because one controlled study found a reduction, 5 whereas another study found no difference. 27 Considering that remifentanil produces dose-dependent respiratory depression, there is still a possibility that these respiratory depression effects offset to some extent the expected shortening effect of induction time due to a decrease of MAC of desflurane.
The main airway irritation sign was excitatory movements in the current study, which was in line with previous studies.27,28 Excitatory movements occurred in all patients in the failure group. Cough was concurrently found in five of 12 (42%) patients and breath holding was found in two of 12 (17%) patients. There was no evidence of laryngospasm. Excitatory movements included verbal or forceful removal of the mask by the patient, plus head and limb movements. These excitatory movements could have been the result of the patient’s response to the pungent stimuli or the expression of an excitatory stage (stage 2) of anaesthesia. Whatever the cause of the excitatory movements, their presence limited the anaesthesiologist’s ability to increase the concentration of desflurane and they were the most common obstacle to the patient tolerating inhaled induction with desflurane.
The current study had several limitations. First, the same concentration of desflurane was used for patients of all ages, but the iso-MACs are somewhat different in patients aged 19 to 60 years. 28 Secondly, there was a basic bias between real plasma concentration and the calculated one for remifentanil based on the pharmacokinetic model of Minto et al.15,29 Thirdly, the feasibility of inhaled induction of desflurane was based on the objective signs during inhalation. There might be dissatisfaction of patients despite the fact that they seemed to be calm during inhalation. This was not checked in this present study. Fourth, Dixon’s up-and-down method has potential limitations for estimating EC95. To overcome this limitation, this present study adopted the isotonic regression method and the bootstrapping approach as supplementary analyses for reducing bias and getting greater precision rather than using conventional methods.20,30
In conclusion, the EC50 and EC95 of effect-site concentrations of remifentanil required for smooth inhalational induction without airway irritation when using desflurane in a stepwise increment were 3.40 ng/ml and 4.31 ng/ml, respectively. Therefore, prior administration of remifentanil could provide smooth inhalational induction with desflurane.
Footnotes
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
