Abstract
Background
A meta-analysis and systematic review of randomized controlled trials to compare the oropharyngeal leak pressure (OLP) and clinical performance of LMA ProSeal™ (Teleflex® Inc., Wayne, PA, USA) and i-gel® (Intersurgical Ltd, Wokingham, UK) in adults undergoing general anesthesia.
Methods
Searches of MEDLINE®, EMBASE®, CENTRAL, KoreaMed and Google Scholar® were performed. The primary objective was to compare OLP; secondary objectives included comparison of clinical performance and complications.
Results
Fourteen RCTs were included. OLP was significantly higher with LMA ProSeal™ than with i-gel® (mean difference [MD] −2.95 cmH2O; 95% confidence interval [CI] −4.30, −1.60). The i-gel® had shorter device insertion time (MD −3.01 s; 95% CI −5.80, −0.21), and lower incidences of blood on device after removal (risk ratio [RR] 0.32; 95% CI 0.18, 0.56) and sore throat (RR 0.56; 95% CI 0.35, 0.89) than LMA ProSeal™.
Conclusion
LMA ProSeal™ provides superior airway sealing compared to i-gel®.
Introduction
Use of supraglottic airway (SGA) devices is increasingly common in clinical anesthesia. 1 Second-generation SGAs including LMA ProSeal™ (Teleflex® Inc., Wayne, PA, USA) and i-gel® (Intersurgical Ltd, Wokingham, UK) were introduced in 2000 and 2007, respectively. These devices provide better airway sealing characteristics than classic LMA™, have an additional drainage tube for stomach decompression to reduce the risk of pulmonary aspiration, and are designed for use with spontaneous or positive pressure ventilation (PPV). 2
Oropharyngeal leak pressure (OLP), measured by closing the expiratory valve of the anesthetic circle system at a fixed gas flow rate and noting the equilibrium airway pressure, is used to quantify the efficacy of airway sealing in SGA devices. 3 Importantly, OLP indicates airway protection, successful SGA placement, and PPV.3,4 Several methods are used to quantify OLP, including audible noise detection, oral capnography, stethoscopic noise and manometric stability.3,4
The clinical performance and safety of both LMA ProSeal™ and i-gel® have been studied extensively,5–19 but reports vary as to which device offers superior OLP. Studies have shown LMA ProSeal™ to have comparable OLP to i-gel®,5,9,11,12,14 or significantly higher7,8,10,13,15,17 or lower 19 OLP than i-gel®.
The present meta-analysis of published randomized controlled trials (RCTs) was performed to compare the clinical performance and airway-sealing characteristics, including OLP, of LMA ProSeal™ and i-gel® in adult patients undergoing general anesthesia.
Materials and methods
This meta-analysis was performed based upon the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statements. 20
Literature search
The electronic databases MEDLINE®, EMBASE®, CENTRAL (Cochrane Central Register of Controlled Trials) and KoreaMed, as well as the web search engine Google Scholar®, were searched for eligible studies. All searches were conducted in April 2014 and updated in December 2014. The Medical Subject Heading search terms and text words included ‘LMA ProSeal’, ‘ProSeal LMA’, ‘PLMA’, and ‘i-gel’. The search was performed across all languages. The title and abstract of each paper were screened by two reviewers (H.W.S. and H.J.K.) and potentially relevant references retrieved.
Study selection
Prospective RCTs that compared LMA ProSeal™ and i-gel® for general anesthesia in patients aged >18 years were included in the analysis. Studies were selected according to predetermined inclusion criteria by two independent reviewers (H.N.Y. and G.E.B.). Any discrepancies were resolved through discussion or consultation with a third independent investigator (H.S.A).
Data extraction
Data were extracted independently and in duplicate by two reviewers (G.E.B. and H.S.Y.) and were recorded using a predefined form that included: name of the first author; year of publication; total number of patients studied; OLP; time required for device insertion; rate of insertion on the first attempt without assistance; fiber-optic view of the glottis (glottis visualization); ease of gastric tube insertion; incidence of blood on the device after removal; and incidence of patient sore throat. The primary objective was to compare OLP between the two devices; secondary objectives were to compare their clinical performance and rate of complications. Attempts were made to contact the authors of studies that had insufficient or missing data; if attempts were unsuccessful, data were extrapolated from the study text or tables to obtain the target information.
Risk of bias assessment
The quality of the RCTs was independently assessed by two authors (H.N.Y. and M.K.P.) using the risk-of-bias tool in RevMan version 5.2 (The Cochrane Collaboration, London, UK). Quality was evaluated using the following potential sources of bias: random sequence generation; allocation concealment; blinding; incomplete outcome data; selective outcome reporting; other sources of bias. The methodology for each RCT was graded as ‘high,’ ‘low’ or ‘unclear’, to reflect either a high, low or uncertain risk of bias, respectively.
Statistical analyses
RevMan 5.2 software was used for statistical analyses. The mean difference (MD) with 95% confidence interval (CI) was computed for continuous variables; risk ratio (RR) with corresponding 95% CI was calculated for dichotomous outcome data. Statistical heterogeneity was estimated using the I2 statistic, which was deemed significant when I2 >50%. Due to the relatively small number of RCTs and the resulting clinical heterogeneity in our meta-analysis, the Mantel–Haenszel or inverse variance random effects model was used instead of the fixed effect model. In the absence of heterogeneity, a Mantel–Haenszel or inverse variance fixed effects model was used. 21 Subgroup analysis for OLP was performed to determine the influence of the use of neuromuscular blocker (NMB; without or with NMB) and type of surgery (non-laparoscopic or laparoscopic). Sensitivity analysis was performed for OLP to evaluate the sequential effect of excluding studies. Subgroup analysis for device insertion time was performed according to the use of NMB (without or with NMB) and study publication year (2009–2012 or 2013–2014). Differences were considered statically significant if P < 0.05. Publication bias was assessed by visual inspection of funnel plots. If the funnel plot was visually asymmetrical, the Egger’s linear regression test was used.
Results
The initial electronic publication search identified 699 potential studies (267 from MEDLINE®, 282 from EMBASE®, 136 from CENTRAL, 12 from KoreaMed and 2 from Google Scholar®). After exclusions, the analysis included 14 RCTs5–18 published between 2009 and 2014, comprising 1104 patients (545 with LMA ProSeal™ and 559 with i-gel®). No records were obtained from ClinicalTrials.gov. The study selection strategy is shown in Figure 1.
Process for inclusion of randomized controlled trials in the meta-analysis to compare the oropharyngeal leak pressure, clinical performance and rate of complications of LMA ProSeal™ and i-gel®.
Characteristics of randomized controlled trials comparing LMA ProSeal™ and i-gel® for oropharyngeal leak pressure, clinical performance and rate of complications.

BMI, body mass index; OLP, oropharyngeal leak pressure.
All studies mentioned randomization, but only seven6–8,11–13,15 included details of concealed allocation. However, the operator inserting the device and the OLP assessors were not blinded in any of the studies (due to the impossibility of blinding their use). Risk of bias in individual studies is summarized in Figure 2. There were no funnel asymmetries in OLP, time required for device insertion, insertion on the first attempt without assistance, fiber-optic view of the glottis, ease of gastric tube insertion, blood on device after removal or sore throat (data not shown).
Risk of bias for randomized controlled trials comparing oropharyngeal leak pressure, clinical performance and rate of complications of LMA ProSeal™ and i-gel®. The color version of this figure is available at: http://imr.sagepub.com.
Data from RCTs that quantified OLP5,7–15,17 indicated significantly lower OLP with i-gel® compared to LMA ProSeal™ (MD −2.95 cmH2O; I2 = 71%; P < 0.0001) with high heterogeneity. Subgroup analyses revealed significantly lower OLP with i-gel® compared with LMA ProSeal™ with the use of NMB and laparoscopic surgery (P < 0.0001 and I2 = 0% for both analyses; Figure 3A and Table 2). There were no between-subgroup differences in OLP with respect to use of NMB and type of surgery (Table 2). Sensitivity analyses revealed no interactions for OLP. There was no funnel plot asymmetry.
Forest plot comparing LMA ProSeal™ and i-gel® for (a) oropharyngeal leak pressure (cmH2O) stratified according to the use of neuromuscular blockade; (b) device insertion time (s) stratified according to publication year of studies. CI, confidence interval; I2, I-square heterogeneity statistic; IV, inverse variance. Subgroup meta-analysis for oropharyngeal leak pressure with LMA ProSeal™ and i-gel®. MD, mean difference; CI, confidence interval; I2, l-square heterogeneity statistic; NMB, neuromuscular blocker; NS, not statistically significant (P ≥ 0.05).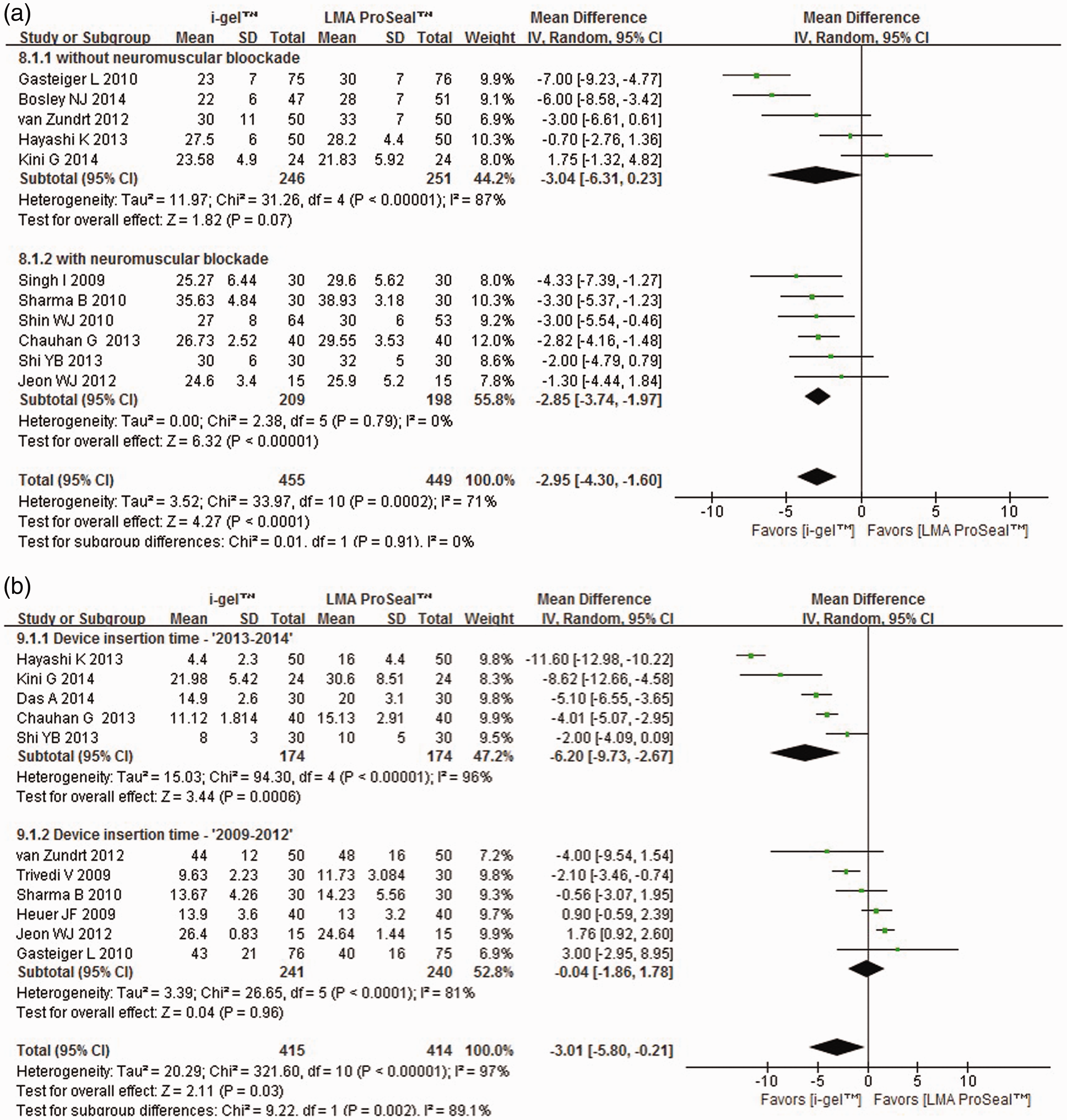

Device insertion time5,6,8–13,15,16,18 was significantly shorter for i-gel® than for LMA ProSeal™, with high heterogeneity (MD −3.01 s; I2 = 97%; P = 0.03). Subgroup analysis indicated significantly shorter insertion time for i-gel® than for LMA ProSeal™ in studies published in 2013–2014, with sustained high heterogeneity (MD −6.20 s; I2 = 96%; P < 0.00001; Figure 3B). Subgroup analyses revealed significant differences based on study publication year (P = 0.002) but not on use of NMB. There was no funnel plot asymmetry.
Blood on the device after removal5–10,12,14,15,17 and sore throat5–10,12,14,15,18 were significantly more common with LMA ProSeal™ than with i-gel® (for blood RR 0.32, I2 = 0%, P < 0.0001; for sore throat RR 0.56, I2 = 18%, P = 0.01; Figures 4A and 4B). There were no between-device differences with respect to insertion on the first attempt without assistance5–7,9,11-18, fiber-optic view of the glottis5,8,12,15 or ease of gastric tube insertion.8–10,15,17
Forest plot comparing LMA ProSeal™ and i-gel® for (a) blood on device after removal; (b) sore throat. CI, confidence interval; I2, I-square heterogeneity statistic; M–H, Mantel–Haenszel.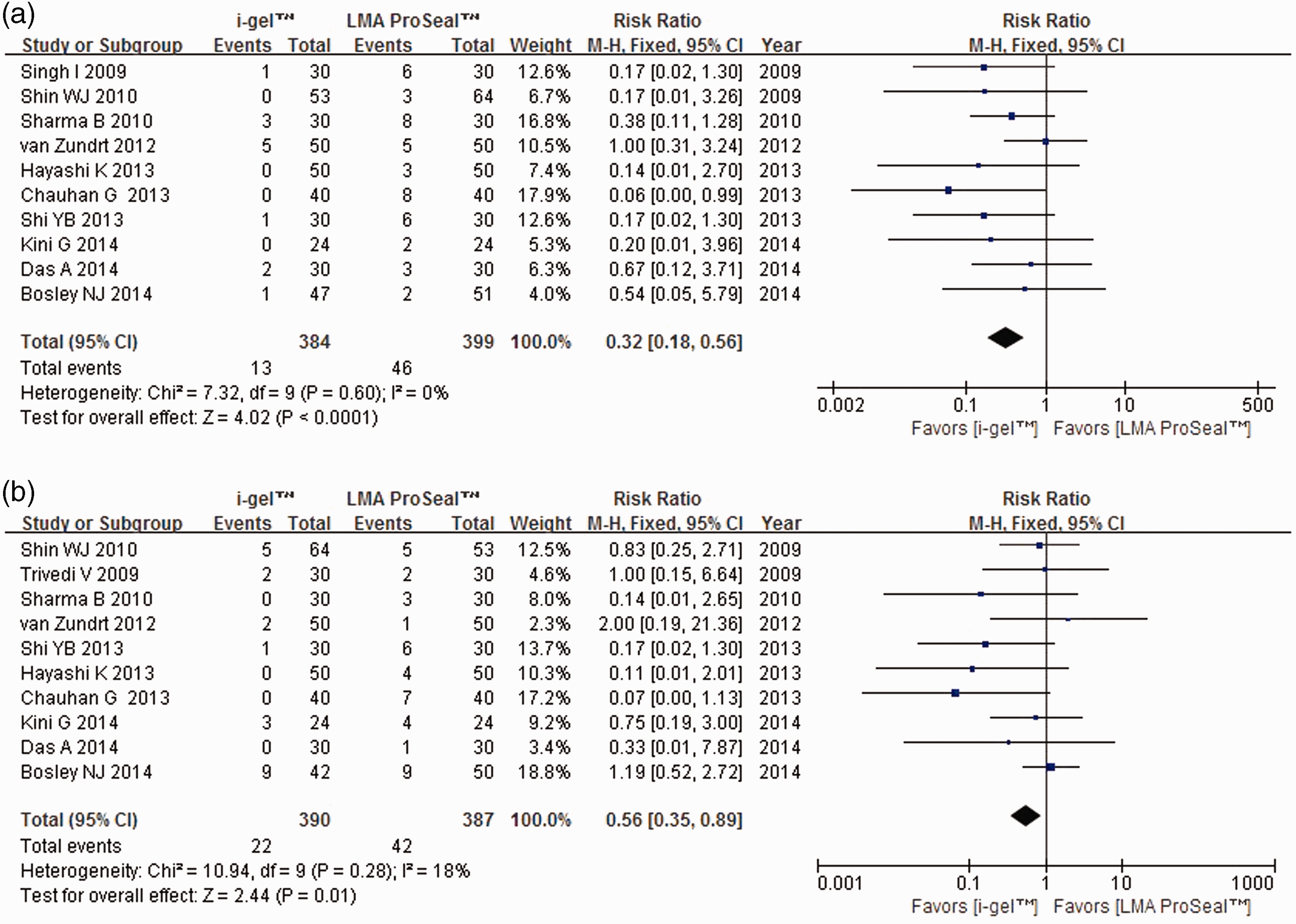
Discussion
The present meta-analysis indicated that i-gel® results in lower OLP, shorter insertion times, lower incidences of blood on device after removal, and sore throat, than LMA ProSeal™.
A potential risk of SGA use is incomplete airway sealing, which may cause gastric insufflation; inflation of airways at pressures above 20cmH2O can induce opening of the esophageal sphincter. 22 Case reports have noted regurgitation and aspiration in patients with both LMA ProSeal™ and i-gel® during anesthesia.23–25 However, a cadaver study reported fast drainage of esophageal fluid using SGAs with gastric channels. 26
Airway sealing in SGA is characterized by OLP as assessed via an audible noise from the mouth or in the neck using a stethoscope, sampling of end-tidal carbon dioxide in the mouth or manometer equilibrium pressure at fixed fresh gas flow rates. OLP is also referred to as airway sealing pressure or airway leak pressure. 3 All four OLP evaluation methods provide similar OLP values, with good correlation in children, 4 and the manometric stability test has been shown to accurately measure OLP in adults. 3 An airway sealing study using a cadaver aspiration model reported that the lack of an inflatable cuff may reduce the airway sealing ability of i-gel® compared with that of LMA ProSeal™. 26
Other factors that may affect OLP include the use of NMB, intra-abdominal pressure during surgery and intracuff pressure of the SGA device.15,26,27 In our meta-analysis, the substantial overall heterogeneity (I2 = 71%) was reduced by subgroup analysis based on NMB use (I2 = 0%) and laparoscopic surgery (I2 = 0%). Our findings suggest that OLP may be variable during surgery without NMB and non-laparoscopic surgery.
Device insertion time was shorter for i-gel® than for LMA ProSeal™ in the studies published in 2013–2014. There appears to be a preference for i-gel® over LMA ProSeal™,2,28 possibly due to the convenience of a disposable device, ease of insertion by stiff bite block and the natural oropharyngeal curvature of i-gel® compared with LMA ProSeal™. 28 Device insertion time showed high heterogeneity after subgroup analysis with use of NMB and publication year; this was possibly due to differences in measurement standards among the studies included in our analysis.
It is possible that the fiber-optic view is better with i-gel® than with LMA ProSeal™ due to interference from folding of the LMA ProSeal™ cuff after insertion, but the absence of a between-group difference in this parameter suggests that both devices might function similarly as a conduit during airway management. The ease of gastric tube insertion was similar with each device in our review. The esophageal drain tube of i-gel® is smaller than that of LMA ProSeal™ (12 F versus 16 F for size 4, respectively). 25 Correct SGA positioning is important to prevent gastric aspiration; the i-gel®, with its good positional stability, may be superior to LMA ProSeal™. 28 The gastric channels of both devices allow early identification of regurgitation and prompt response to prevent aspiration.2,28 The inflated cuff of LMA ProSeal™ may contribute to the higher incidence of sore throat seen with this device compared with i-gel® (which has no cuff).
Meta-analyses comparing LMA Proseal™ and i-gel® have reported similar OLP for both devices.29,30 This is in contrast to our findings, which showed that LMA ProSeal™ provided higher OLP than i-gel®. This disparity may be due to differences in data collection. OLP is also referred to as airway sealing pressure and airway leak pressure.3,4 We included ‘OLP’, ‘airway sealing pressure’ and ‘airway leak pressure’ as search terms, but other studies searched only for ‘OLP’.29,30 Subgroup analysis for OLP including ‘OLP’, ‘oropharyngeal seal pressure’ and ‘airway sealing pressure’ as search terms found that second-generation LMAs (ProSeal™, Supreme™) had lower OLP than i-gel®. 30 This partially incomplete search strategy would have omitted several studies that were included in the present meta-analysis.10,16–18
There are many situations in which SGA devices are required to maintain high OLP against increased intra-abdominal pressure in laparoscopic surgery, obese patients and patients with restrictive lung disease. A meta-analysis of pediatric studies found higher OLP with i-gel® than with LMA ProSeal™. 19 This contradictory finding may be explained by the lack of dorsal cuffs in sizes 1.5–2.5 for LMA ProSeal™.4,19 Anesthetists must weigh up the clinical performance and airway sealing safety of SGAs in clinical practice. The LMA ProSeal™ is regarded as a choice for airway sealing in adults that has a good safety profile, but i-gel® is preferred for pediatric procedures because it has a good safety profile in children.5–19
A limitation of this review is the clinical heterogeneity without power analysis or sample-size determination of the included studies. Other limitations are the performance and detection bias arising from the impossibility of blinding to device insertion, measurement of OLP and clinical performances.
In conclusion, our findings are that LMA ProSeal™ provides superior airway sealing (higher OLP) compared to i-gel®, while i-gel® offers rapid insertion time, and lower incidences of blood on the device after removal and sore throat compared to LMA ProSeal™ in anesthetized adult patients.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
