Abstract
Objective
To determine whether a multiplex polymerase chain reaction (PCR)-based test could reduce the time required for initial pathogen identification in patients in an intensive care unit (ICU) setting.
Methods
This double-blind, parallel-group randomized controlled trial** enrolled adults with suspected pulmonary or abdominal sepsis caused by an unknown pathogen. Both the intervention and control groups underwent the standard blood culture (BC) testing, but additional pathogen identification, based on the results of a LightCycler® SeptiFast PCR test, were provided in the intervention group.
Results
The study enrolled 37 patients in the control group and 41 in the intervention group. Baseline clinical and demographic characteristics were similar in both groups. The PCR-based test identified a pathogen in 10 out of 41 (24.4%) patients in the intervention group, with a mean duration from sampling to providing the information to the ICU of 15.9 h. In the control group, BC results were available after a significantly longer period (38.1 h).
Conclusion
The LightCycler® SeptiFast PCR test demonstrated a significant reduction in the time required for initial pathogen identification, compared with standard BC.
Keywords
Introduction
Based on data from a large international prevalence study, half of all patients being treated on an intensive care unit (ICU) developed an infection and this was associated with a doubling of the mortality rate. 1 Sepsis remains one of the most common complications in critically ill patients and is associated with fatal outcomes. 2 The ongoing continuum from infection to sepsis and the resulting organ dysfunction requires prompt pathogen identification and therapeutic intervention, because time has been identified as a critical factor that influences outcomes.3–5 Based on national and international guidelines, diagnostics should be tailored according to patient status, and diagnostic procedures should never delay initiation of empirical antimicrobial therapy in patients with septic shock.5,6 On the other hand, blood culture (BC) diagnostics and other microbiological culture-based methods depend on living organisms; thus, antibiotic therapy reduces the microbiological detection rate. 7 Modern diagnostic approaches include bacterial identification based on genomic polymerase chain reaction (PCR) analytics; these methods are not dependent on the viability of the organisms being sampled. 8 One of these innovative methods is the LightCycler® SeptiFast PCR test (Roche Diagnostics GmbH, Mannheim, Germany), which is a commercially available multiplex PCR system for the detection of bacterial and fungal DNA in blood. This assay demonstrated sufficient sensitivity (62–80%)9,10 and excellent specificity (70–95%) in several observational studies.9,11
Prompt microbiological identification of the underlying pathogen in infected patients and the consequent adaption of the antimicrobial therapy might lead to improved patient outcomes in the ICU. At a time when there is a growing problem of drug resistant micro-organisms, there is a considerable need for rapid diagnostic tests. 12 The time advantage for PCR diagnostics might be of importance especially in patients being treated with empirical antimicrobial therapy that does not target the specific causative species. However, the impact of PCR-based pathogen identification on infection management in the ICU setting has not been evaluated in randomized controlled trials (RCTs) to date.
Against this background, the aim of the present study was to determine whether rapid pathogen identification using a multiplex PCR test in ICU patients could reduce the time required for initial pathogen identification and subsequent antimicrobial therapy modification to target the specific pathogen. We hypothesized that the rapid availability of the results of the PCR-based pathogen identification would be accompanied by a reduced delay of antimicrobial therapy modification in critically ill patients with severe infections.
Patients and methods
Study design
This double-blind, parallel-group RCT was undertaken at two university hospitals in Berlin, Germany: Charité University Hospital Campus Charité Mitte; Charité University Hospital Campus Charité Virchow Klinikum. A total of six ICUs enrolled patients between August 2010 and March 2012; five anaesthesiological ICUs and one surgical ICU. This study was approved by the local Ethics Review Board (Ethics Commission, Charité–Universitaetsmedizin Berlin Campus Charité Mitte, Berlin, Germany) and the data safety authorities; it was also registered with the International Standard Randomized Controlled Trial number ISRCTN70694559. Written informed consent was obtained from every included patient or their legal representative.
Patient inclusion and randomization
Patients were eligible for study inclusion when they presented with signs of sepsis of suspected abdominal or pulmonary origin, caused by an unknown pathogen when BC diagnostics were indicated. Sepsis was defined as suspected or proven infection causing systemic inflammation with at least two of the following: (i) leucocyte count <4 or >12 /nl; (ii) body temperature <36℃ or fever >38℃; (iii) tachypnoea >20/min or hyperventilation (paCO2 <32 mmHg); (iv) tachycardia >90 bpm. 5 Infections were defined by the treating physicians using standardized criteria for infection management implemented on the participating wards since 2006. 13 All patients were included only once even if they had multiple episodes of sepsis during their ICU stay. Additionally, infection onset was required to be <72 h, to reduce the risk of detecting persistent circulating DNA. Exclusion criteria were age <18 years, pregnancy, police custody, missing or withdrawn informed consent or participation in another prospective clinical study.
Patients fulfilling the inclusion criteria were asked to provide written informed consent. A substantial number of critically ill patients are unable to give consent due to their clinical status, their need for mechanical ventilation, or altered mental status. In these cases, patients were included in the randomization process with a delayed consent procedure. Informed consent was obtained as soon as possible, from the patient or their legal representative.
Eligible patients ≥18 years old were randomized to either the control or intervention group in a 1 : 1 ratio, according to consecutive sample processing by the Institute for Microbiology and Hygiene Berlin, Charité–Universitaetsmedizin Berlin, Berlin, Germany. Randomization was achieved using computer-generated random numbers and performed in blocks of four. The randomization scheme was not known to the treating physicians on the ICUs. To minimize the risk of bias, allocation was blinded to all clinical study participants and staff members on the ICU. Furthermore, to achieve maximum allocation concealment, treating physicians on the ICUs were not aware of group assignment until the results of the multiplex PCR analysis were available and communicated to the study ICU wards.
Microbiological procedures, study intervention, data collection
Microbiological diagnostics were performed on participating ICU wards, whenever the patient’s clinical condition indicated possible systemic infection. Typically, in patients with suspected pneumonia, two BC series (each containing one anaerobic and one aerobic bottle) and samples from the respiratory tract were taken. In suspected abdominal infection, two BC series (each with 10 ml of blood) and at least one culture from the infection site (e.g. intraoperative sample) were obtained. In all patients, an additional blood sample was taken from a venous or arterial site; a sterile BC puncture was obtained and an additional volume of 1.5 ml of whole blood was added to a 1.8 mg/ml ethylenediaminetetra-acetic acid sample tube. Samples were stored at 7℃ until transfer to the laboratory.
In patients assigned to the intervention group, biological samples were processed immediately for the LightCycler® SeptiFast PCR test (see below for details) and results were communicated to the physician team on the study ICU ward as soon as they were available; in the control group, biological samples were stored at −80℃ for later analysis. All microbiological samples were processed using standard methods as required by the clinical care process. Positive results from the BC Gram staining were immediately communicated by the microbiologist to the treating physicians on the study ICU wards. BC bottles (BACTEC™; Becton Dickinson, Franklin Lakes, NJ, USA) were inoculated for 6 days until growth was detected. Positive bottles were streaked onto a set of agar plates and subjected to direct Gram staining. Any colonies that grew in culture were identified using matrix-assisted laser desorption ionization–time of flight equipment (MALDI-TOF, microflex LT; Bruker Daltonic, Bremen, Germany) or biochemical identification tests (Vitek® 2; bioMérieux, La Balme-les-Grottes, France). Commercial procedures were used for antimicrobial susceptibility testing (Vitek® 2 or Etest®; bioMérieux).
The multiplex PCR evaluated in this trial was the commercially available LightCycler® SeptiFast test using a sequential semiautomatic workflow (Roche Diagnostics GmbH, Mannheim, Germany). The sample processing included mechanical cell lysis using SeptiFast MagNALyser followed by magnetic adsorption DNA purification in the MagNA Pure Compact System and detection of pathogen-specific DNA in a LightCycler® PCR instrument. All results were processed using the LightCycler® SeptiFast identification software provided by the manufacturer. The test system allows identification of 25 common BC pathogens without analysis of resistance patterns. The LightCycler® SeptiFast PCR test system is described in detail elsewhere. 14
Clinical data were taken prospectively from all patients during their ICU stay based on all available written information and the electronic patient chart. This system contains all relevant data for every ICU day including laboratory data, physiological parameters (e.g. heart rate, body temperature), as well as information regarding ventilation, results from physical examinations and diagnostic or therapeutic intentions). Measurements included the Simplified Acute Physiology Score II to assess severity of disease on admission. 15 In patients receiving any immunosuppressive agents or chemotherapy within the past 6 weeks, an impaired immune system was recorded. Patients fulfilled criteria for septic shock when hypotension (systolic blood pressure <90 mmHg) was noted or when there was the need for vasopressor usage to maintain a sufficient blood pressure. Medical history of patients was also evaluated for documented alcohol abuse. Microbiological information was taken from the main hospital patient database.
Study endpoints
The study design was intended to be comparable with the clinical situation where multiplex PCR testing would be an available diagnostic option, comparable with current standard BCs. 3 Multiplex PCR assays are undergoing rapid development; bedside tests as well as automated analyses are already available. BC bottles are transported to the laboratory throughout the day and are inoculated even during the night and at weekends. The multiplex PCR test analysed in this study required ∼170 min of skilled hands-on time and thus was only available during the daytime and not at weekends. For the purpose of this study, the availability of the multiplex PCR test processing was requested to be within 12 h after sampling. Against this background, patients were included between 18.00 h and 06.00 h for comparable transport durations and duration until sample processing for both treatment arms. Therefore, this study included patients with progression of infection during this timeframe. However, when BC diagnostics had already been undertaken during daytime hours, patients did not fulfil the inclusion criteria. The choice of antimicrobial therapy and the clinical-care process were completely under the responsibility of the ICU physicians without any interference from the study team. Members of the study team were not part of the clinical care staff on ICU wards.
The primary aim of the study was to compare the duration from sampling until availability of the microbiological results. The time was calculated from the moment of sterile puncture of the vein and allocation of the necessary blood sample for both tests (LightCycler® SeptiFast PCR and BC bottles) until the information from the LightCycler® SeptiFast PCR test or a positive BC result were communicated by the microbiologist to the ICU physician by telephone. Furthermore, this study aimed to assess the influence of the additional information on therapy modification, and to describe the corresponding time frame for the bedside decision. Therefore, all related changes in antimicrobial therapy were monitored. Secondary endpoints included a comparison of the spectrum of pathogens that were detected in the two groups and the sensitivity and specificity of the LightCycler® SeptiFast PCR test.
Statistical analyses
All statistical analyses were performed using the SPSS® statistical package, version 19.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Variables were analysed using numbers and percentages, median and quartiles (25%–75% percentile), or mean ± SD based on scale level and distribution of normality, as appropriate. All tests for statistical significance were performed with a two-sided alpha < 0.05. Depending on the scale level, Fisher’s exact χ2-test, Student’s t-test, or Mann–Whitney U-test were used. It was not possible to calculate the sample size requirements a priori: the time demand for the decision-making process on the basis of pathogen detection using PCR technology was unpredictable, based on previous data. Consequently, a conservative approach was used by enrolling 100 patient samples. Based on the findings of the study in the context of its pilot nature, a power analysis was performed to evaluate the number of patients needed to achieve a power of 80% with a two-sided alpha of 5% (nonparametric unpaired Wilcoxon test), to demonstrate a difference in the time requirement for the PCR technology in a future confirmatory RCT (nQuery Advisor® software, version 5.0; Statistical Solutions, Boston, MA, USA). A P-value < 0.05 was considered statistically significant.
Results
During the study period, 100 consecutive patients were eligible for inclusion in the study. Of these, 22 were unable to provide written informed consent because the patients or their legal representatives refused to participate in the study or the patient experienced rapid clinical deterioration and subsequent death with no legal representative being nominated. According to the institutional review board vote, all data from these 22 excluded patients were deleted. As a consequence, the study followed-up and analysed 78 patients: 37 in the control group and 41 in the intervention group (Figure 1).
Consort flow diagram for patient enrolment into a randomized controlled clinical trial that evaluated whether rapid pathogen identification (using a multiplex polymerase chain reaction test) reduced time required for the initial pathogen identification and subsequent therapy modification to target the specific pathogen, in patients treated in intensive care units.
Baseline clinical and demographic characteristics of patients treated in intensive care units (ICUs), randomized to undergo either a multiplex polymerase chain reaction test for initial pathogen identification (intervention group) or standard blood culture analysis (control group).

Data presented as median (25–75 percentile) or n (%).
SAPS II, Simplified Acute Physiology Score II. 15
No significant between-group differences (P ≥ 0.05).
Results of standard blood culture (BC) microbiological analyses, LightCycler® SeptiFast polymerase chain reaction (PCR) tests and corresponding clinical pictures for patients treated in intensive care units and randomized to undergo either a multiplex polymerase chain reaction test for initial pathogen identification (intervention group) or standard BC analysis (control group).

TBS, tracheobronchial secretion; BAL, bronchoalveolar lavage.
In the control group, BCs were positive in seven of 37 patients (18.9%) growing S. epidermidis, E. faecium, E. faecalis and S. aureus (Table 2). In the post-hoc LightCycler® SeptiFast PCR test of collected samples, eight pathogens were identified (E. coli, E. faecium, E. faecalis, Streptococcus pneumoniae, S. aureus and S. epidermidis). In one case with BCs growing two pathogens (E. faecalis and S. epidermidis), the LightCycler® SeptiFast PCR test only identified E. faecalis.
The concordance rate between the BC results and those of the LightCycler® SeptiFast PCR test were analysed for the intervention and control groups. Seven samples were positive for the same pathogen in both the LightCycler® SeptiFast PCR test and the corresponding BC; 49 samples were validated as negative for both assays. In six control-group samples analysed after short-term frozen storage, the LightCycler® SeptiFast PCR test result had a technical signal in the test summary and was not validated: in two samples, no pathogens were detected but the internal control was also negative; in four samples, the negative control channel was positive, indicating potential system contamination. None of these patients had a pathogen detected in BCs. These results demonstrated a concordance rate of 56 out of 78 analysed samples (71.8%).
For analysis of the test performance of the LightCycler® SeptiFast PCR test, BC results were used as the diagnostic gold standard for detecting bacteraemia. The resulting sensitivity for a positive LightCycler® SeptiFast PCR test result was 58.3% (seven of 12 samples) with a corresponding specificity of 74.2% (49 of 66 samples).
Modifications of empirical antimicrobial therapy (standard regimens), based on results of microbiological analyses, in patients treated in intensive care units and randomized to undergo either a multiplex polymerase chain reaction test for initial pathogen identification (intervention group) or standard blood culture analysis (control group).

Results of time analyses for patients being treated in intensive care units, randomized to undergo either a multiplex polymerase chain reaction (PCR) test for initial pathogen identification (intervention group) or standard blood culture analysis (control group).
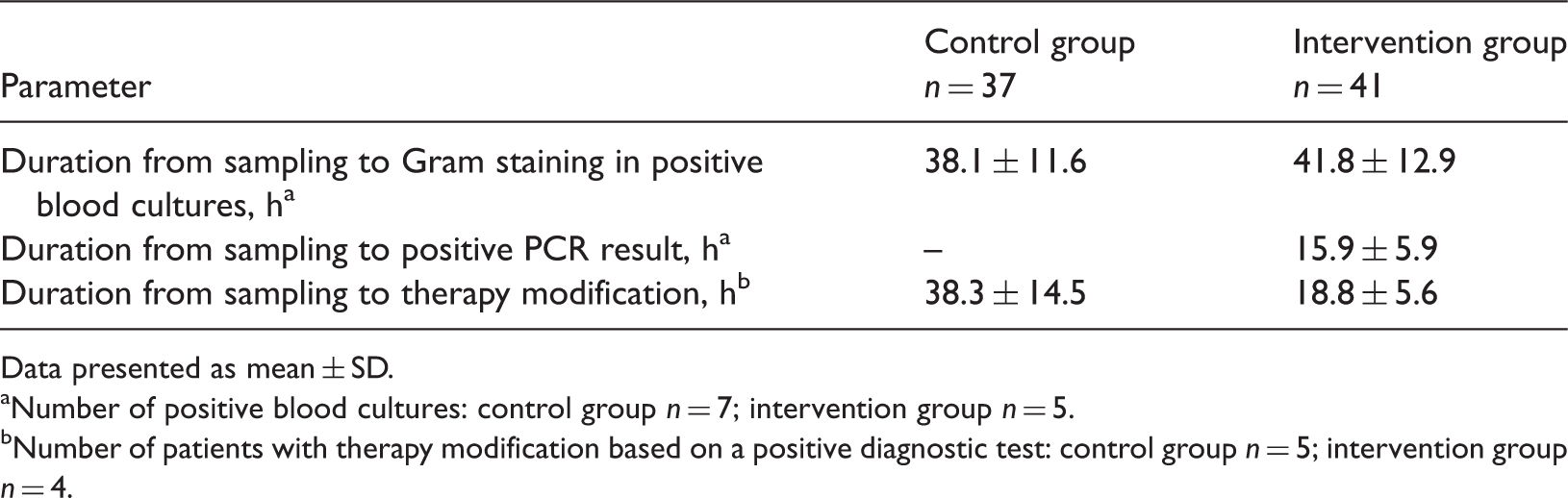
Data presented as mean ± SD.
Number of positive blood cultures: control group n = 7; intervention group n = 5.
Number of patients with therapy modification based on a positive diagnostic test: control group n = 5; intervention group n = 4.
For the reduction in the time to antimicrobial therapy modification, this study reached a power of 55% with an exact alpha of P = 0.063. Based on these findings, a second a priori power analysis was performed to assess the sample size for a future confirmatory RCT evaluating this endpoint. With a conservative assumption of 80% power and a relevant effect size of 1.691, a nonparametric unpaired Wilcoxon test analysis would be able to determine a significant difference between the two study groups if they included nine patients with therapy modifications in each study arm (i.e. a total of 18 patients).
Discussion
This double-blind, parallel-group RCT demonstrated a significant reduction in the time required for initial pathogen identification using a multiplex PCR detection platform, compared with standard BC diagnostics, in ICU patients with clinical sepsis of pulmonary and abdominal origin. Most importantly, this very early identification of the causative pathogen within 24 h of admission was integrated into the clinical decision-making process and resulted in earlier modifications of empirical antimicrobial therapy. Using the LightCycler® SeptiFast PCR test shortened the duration from sampling to availability of microbiological results by 22.2 h. Two cases of candidaemia were detected in the intervention group, both of which were validated with subsequent standard BC diagnostics. PCR-based methods might be most beneficial for patients with a high incidence of fungal infections or ongoing antibiotic therapy. 16
Results from the standard BCs and the LightCycler® SeptiFast PCR test were concordant in seven of 12 BC positive cases (i.e. the gold standard). Therefore, the LightCycler® SeptiFast PCR test had a sensitivity of 58.3%. The specificity of the LightCycler® SeptiFast PCR test was 74.2% (49 of 66 samples). Other clinical research showed a sensitivity of 50–100%, even with low bacterial counts of 3 colony forming units/ml, depending on the species. 14 Although authors reported a specificity of 100%, contamination during sampling and sample workflow might be an issue. 14 This difficulty is also a well-described phenomenon for standard BC diagnostics and a contamination rate of ∼5% is reported for the ICU setting. 17 A Japanese group reported a higher positive rate using a PCR-based method than with BC, but demonstrated a slightly higher sensitivity and specificity than was observed in this current study. 18 The sensitivity and specificity of the LightCycler® SeptiFast PCR test in this current study were similar to those reported in recent studies conducted in Germany and Spain.19,20 The Spanish group evaluated patients with complicated bloodstream infections and reported a sensitivity of 56.0% and a specificity of 79.5%. 20 It should be noted that in all these studies including the current study,14,18–20 BC results were used as the reference gold standard, even though the sensitivity of this standard diagnostic methods is also limited. 7 As shown in Table 2, there were cases with negative BCs but positive PCR-based pathogen identification. These pathogens were also identified in other microbiological samples. Results from other biological samples, such as urine and tracheobronchial or abdominal drainage secretions, may be included in the clinical assessment of a specific patient’s condition. In some cases, BCs may have shown false-negative results, but analysis of diagnostic performance remains limited to the reference diagnostics.
Timely identification of the causative pathogen provides important information to ICU physicians that can be used to guide antimicrobial therapy usage while also reducing nonessential use of antimicrobial agents. In this current study, two cases of unsuspected candidaemia were identified using the LightCycler® SeptiFast PCR test. In this context, a quasi-experimental, before and after, observational cohort study demonstrated that a conservative strategy with prompt diagnostics and reluctant antibiotic prescribing was associated with reduced overall mortality in ICU patients. 12 These authors also reported that their microbiological results within 24 h. 12 The application of fast pathogen detection methods might enable physicians to tailor antibiotic prescribing to individual patients in the future.
Although the LightCycler® SeptiFast PCR test was expected to reduce the time to initial pathogen diagnosis based on the methodological characteristics of the test, the effects on the subsequent clinical decision making process was not predictable a priori. A reduction in the time to pathogen identification through the use of PCR-based technology has been previously reported, but the study used an open design with the physicians knowing the results for all patients and them not reporting the time until they received the results of the BCs. 19 PCR-based methods might be particularly useful in patients pretreated with antibiotics, as reported by a Japanese group. 18 The best results in terms of rapid pathogen identification would probably require the combination of both standard BC methods and PCR-based technololgy.21,22 There remains an element of uncertainty about PCR-based pathogen identification because circulating DNA does not necessarily mean that the patient is infected with the pathogen. In this current study, the risk of detecting persistent circulating DNA was reduced by only including patients in the first 72 h after the onset of sepsis.
This study had a number of strengths and limitations. The strengths included external patient randomization, which included allocation concealment and separation of clinical care teams from the study team, to maximize the reduction in bias. Furthermore, the study groups were similar with regard to their baseline demographic and clinical characteristics. The limitations included the fact that the a priori scenario assumed that the availability of the multiplex PCR-based test was equal to that of standard BC diagnostics, which it was not. As a consequence of the limited availability of trained staff to undertake multiplex PCR-based test during evenings and weekends, and to ensure an adequate comparison of the two diagnostic techniques, patients were only enrolled into the study between 18.00 h and 06.00 h because the multiplex PCR-based test was only available for 12 h during the daytime on weekdays. This assumption was made because of the recent technical developments in the field. Multiplex PCR technology is used in platforms with improved automation and has also been developed for point-of-care settings. In contrast, the workflow for standard BCs allowed for immediate growth of micro-organisms and automated incubation was also available during the night and at weekends. This limited enrolment period led to a substantial number of patients not fulfilling the inclusion criteria because of having already had BCs taken during the regular morning shifts on the ICU ward. The limited enrolment period also meant that patient recruitment lasted longer than initially expected. As a result of this particular limitation, generalizability of these findings is limited to the precondition that the multiplex PCR-based test was only available within a distinct time frame. Compared with previous trials assessing specific clinical settings,23,24 this current study focused on a broader target population, which was recruited from six different ICUs at two study sites and represented a broad spectrum of patients admitted to postoperative ICUs. It was not in the scope of this current study to evaluate different PCR-based diagnostic techniques that are currently available. 25 The current study only aimed to assess the use of DNA-based pathogen detection using one specific assay, the LightCycler® SeptiFast PCR test. Furthermore, this trial did not evaluate the economic impact of this innovative technology. Similarly, the impact on patient outcomes is one of the most important aspects, but this was not investigated in this current study. Morbidity and mortality outcomes should be addressed in future trials, to demonstrate if advantages of rapid multiplex PCR-based pathogen diagnosis lead to clinical benefits for patients.
This trial was designed to provide an estimate of the number of patients that would be needed to be included in a confirmatory RCT with a comparable design in future. Based on these findings, with a power of 80%, 18 patients with therapy modifications based on rapid PCR-based diagnostic information would be needed to demonstrate a statistically significant difference on a two-sided alpha level of 5%. If there was a low pathogen detection rate of 10% in the population, 180 patients would be required.
In conclusion, multiplex PCR-based identification of microbial pathogens might represent a fundamental change in the way that physicians manage bacterial and fungal infections. PCR-based technology is developing quickly in this field, and sample processing and workflow have already improved compared with the LightCycler® SeptiFast PCR test platform used in this study. 25 With the innovations being made and the growing clinical availability of these PCR-based techniques, a time requirement of <6 h from sampling to pathogen identification could mean that infection management shifts from modifying empirical therapy to providing pathogen-specific targeted regimens right from the initiation of therapy. However, more clinical data are required to prove the reliability of PCR-based technology, as well as its usefulness, in different clinical settings.
Footnotes
Declaration of conflicting interest
All authors declare that they have no conflicts of interest concerning the specific subject of this study. I.N. and S.T. received lecture fees from Roche Deutschland GmbH and Pfizer Deutschland GmbH. C.S. received lecture fees or grants for other projects from Abbott, Aspect, Baxter, Deltex, Care Fusion, Fresenius, Hutchinson, Köhler, MSD, MCN, Novartis, Sysmex, Pajunk, Köhler Chemie, Essex Pharma, Pfizer and GSK.
Funding
This study was supported by a restricted grant, from Roche Deutschland GmbH, which covered laboratory transportation and microbiological diagnostic materials.
Acknowledgements
We would like to thank the patients and their relatives for their willingness to participate in this study in a difficult clinical situation. Also, we would like to gratefully mention Sara Casu MD, Susanne Heim MD, Robert Powollik MD, and Andreas Rothbart MD for their support in this study.
