Abstract
Objective
*These authors contributed equally to this work.
Methods
Archival samples of primary human renal cell carcinoma tissue and surrounding normal renal tissue (control samples) obtained from patients diagnosed with renal cell carcinoma were analysed for COX-2 and VEGF-A expression by immunohistochemistry using specific monoclonal antibodies. Tumour microvasculature was examined using factor VIII-related antigen antibody staining.
Results
A total of 33 renal cell carcinoma and 12 control renal tissue specimens were included. COX-2 and VEGF-A genes were overexpressed in tumour specimens compared with normal epithelia. A significant correlation was found between COX-2 and VEGF-A expression. Microvessel density was found to be increased in tumour tissues that expressed COX-2 and VEGF-A.
Conclusion
Microvessel density was increased in tumour tissues that expressed COX-2 and VEGF-A, suggesting that COX-2 and VEGF-A are related to tumour angiogenesis in human renal cell carcinoma.
Keywords
Introduction
Renal cell carcinoma is one of the most common urological malignancies worldwide, with an incidence among cases of urinary tumour ranking second only to bladder cancer in China. 1 The pathogenesis, underlying mechanisms, and prognosis of renal cell carcinoma remain poorly understood, and current measures for its prevention and treatment require further investigation to develop a greater understanding of this disease.
The cyclo-oxygenase (COX)-2 gene, officially known as prostaglandin-endoperoxide synthase 2 (prostaglandin G/H synthase and cyclo-oxygenase; PTGS2), encodes an inducible enzyme, typically with low or no expression in normal tissues, but high expression in pathological tissues. Previous studies have shown that COX-2 participates in tumour development and progression by promoting cell proliferation, inhibiting cell apoptosis, and promoting tumour angiogenesis.2,3 COX-2 expression is also shown to be high in gastrointestinal and urinary cancer tissues, and COX-2 has been shown to induce tumour angiogenesis, and promote tumour invasion and metastasis.4–8 Information regarding the role of COX-2 in the development of renal cell carcinoma, however, is limited.
In the present study, tumour angiogenesis and expression of COX-2 and vascular endothelial growth factor (VEGF)-A in renal cell carcinoma were investigated using immunohistochemical methods. The role of COX-2 and VEGF-A in tumour angiogenesis, and the related mechanisms, were discussed.
Patients and methods
Study population and archived tissue
Paraffin-wax embedded specimens of renal cell carcinoma tissue were randomly selected from those collected and archived at The Union Hospital of Fujian Medical University, Fuzhou, China between January 2006 and December 2010. Following initial dissection, tissue samples had been fixed overnight in paraformaldehyde then processed using standard paraffin-wax embedding methods. All specimens were reconfirmed by a pathologist. Tissue samples were classified using the World Health Organization (WHO) pathological grading system9 and Robson pathological staging system. 10 Normal renal paraffin-wax block tissue specimens were also selected from archives, obtained from the healthy tissue surrounding renal cell carcinoma tissue in patients diagnosed with renal cell carcinoma. These were pathologically confirmed and used as controls. There were no inclusion or exclusion criteria. The study was approved by The Union Hospital of Fujian Medical University ethical review committee and patients provided written or verbal informed consent for their samples to be included in the study.
Immunohistochemical detection of COX-2 and VEGF-A expression
Rabbit antihuman COX-2 monoclonal primary antibody, mouse antihuman VEGF-A monoclonal primary antibody, and rabbit antihuman factor VIII-related antigen primary antibody were purchased from Cell Signaling Technology (Danvers, MA, USA). Streptavidin–biotin–peroxidase complex kit and benzidine chromogenic kit were purchased from Fuzhou Maixin Bio-tech (Fujian, China).
Archived paraffin block tissue specimens were cut into 5 µm-thick sections. Heat induced epitope retrieval was performed prior to immunohistochemical staining by deparaffinizing the sections with two changes of xylene for 5 min each, then rehydrating with two changes each of 100% ethanol (3 min each), 95% ethanol and 80% ethanol (1 min each), with a final rinse in distilled water. The slides were then immersed for 20 min in a staining dish containing 1 mM ethylenediaminetetra-acetic acid (EDTA) buffer (with 0.05% Tween 20), pH 8.0, preheated in a water bath to 95–100℃. The staining dish was then removed from the water bath to room temperature and the slides were allowed to cool for 20 min. Sections were then rinsed twice for 2 min each using 0.02 M phosphate buffered saline (PBS; pH 7.2) Tween.
Immunohistochemical detection was performed following the manufacturers’ instructions. Briefly, sections were incubated with normal goat serum for 30 min at room temperature. The serum was discarded and avidin blocking was then performed by adding 0.05% avidin for 30 min at 37℃. The blocking solution was then discarded and either rabbit antihuman COX-2 primary antibody (1: 200 dilution in 0.02 M PBS, pH 7.2), mouse antihuman VEGF-A primary antibody (1 : 200 dilution), or rabbit antihuman factor VIII-related antigen primary antibody (1 : 200 dilution) were added, and sections were incubated for 1 h at room temperature. Sections were then washed three times (5 min each) in PBS-Tween followed by incubation with goat antimouse or mouse antirabbit Immunoglobulin G (1 : 400 dilution) for 30 min at 37℃. Sections were washed again three times (5 min each) in PBS-Tween, and incubated with streptavidin horseradish peroxidase for 30 min at 37℃, followed by washing three times (5 min each) time in PBS-Tween. Slides were coloured using 3,3′-diaminobenzidine (DAB), incubated in the dark at room temperature for 10 min then washed with distilled water. Slides were finally stained with haematoxylin. PBS was used in place of the primary antibody as a negative control. Positive control slides were obtained from Fuzhou Maixin Bio-tech Ltd.
For determination of COX-2 levels, ∼ 1000 cells were counted per field of view in 10 different high-power fields (400 ×magnification). The percentage of COX-2-positive cells (with brown or dark brown cytoplasm) was calculated for each field. The sample was scored positive if the mean percentage of 10 fields was ≥10% and negative if the mean percentage was <10%, in accordance with a previously described method. 11
The VEGF-A-positive cells with brown stained particles in the cytoplasm were counted in 10 different randomly selected high-power fields (400 ×magnification) per slide. 12 A total of 100 tumour cells/field were counted and the percentage of cells with positive cytoplasmic staining was calculated. VEGF-A expression was considered to be positive if the mean percentage of positive cells was ≥10% and the mean percentage of negative cells was <10%.
Microvessel density was calculated according to a previously published method. 13 Briefly, brown-staining of endothelial cell or endothelial cell clusters, clearly separate from adjacent microvessels, tumour cells, and other connective-tissue elements, were considered as a single, countable microvessel. Vessels with diameter greater than eight red blood cells or with thick smooth muscle or hardened areas were excluded. Five low-power fields (200 ×magnification) with high microvessel density were selected for microvessel counting using a high-power lens (400 ×magnification). For each sample, microvessel density was calculated as the mean of five fields of view.
Statistical analyses
Data were presented as n incidence or mean ± SD. χ2-test was used to assess between-group differences and κ-analysis was used to assess the correlation between COX-2 and VEGF-A expression levels. All tests were performed using SPSS® software, version 10.0 (SPSS, Inc., Chicago, IL, USA), and a probability less than P < 0.05 was considered to be statistically significant.
Results
Renal cell carcinoma tissue specimens from 33 patient cases were included (20 male and 13 female patients, aged 17−86 years, mean age 54.3 years). On the basis of the WHO pathological grading system, 11, 15 and seven specimens were classified as grade G1, G2, and G3, respectively. On the basis of the Robson pathological staging system, four, 16, eight and five specimens were classified as stage I, II, III, and IV, respectively. Among the specimens, 20 cases were clear cell carcinoma, eight were granular cell carcinoma, and five cases were clear–granular mixed cell carcinoma.
A total of 12 cases of normal renal tissue were pathologically confirmed and used as controls. The selected specimens were from eight male and four female patients, aged 26–77 years, with a mean age of 42.1 years. On the basis of the WHO pathological grading system, four, six and two specimens were classified as grade G1, G2, and G3, , respectively. On the basis of the Robson pathological staging system, two, six, three and one specimens were classified as stage I, II, III, and IV, respectively. Among the specimens, six cases were from patients with clear cell carcinoma, three had granular cell carcinoma, and three had clear–granular mixed cell carcinoma.
COX-2 and VEGF-A expression in renal cell carcinoma
Renal cell carcinoma COX-2 expression appeared as brown or dark brown staining that was mainly diffusely distributed in the cytoplasm and in parts of the nucleus of tumour cells, and sporadically distributed in interstitial tissue. COX-2 expression was either weakly positive or not expressed in the stroma (Figure 1). VEGF-A expression appeared as brown or dark-brown staining, mainly in the cytoplasm and membrane of the tumour cells, but also in the cytoplasm of stromal vascular epithelial cells (Figure 2). Normal renal tissues were lightly stained in the collecting system or pink in the cytoplasm. The rate of COX-2 expression was significantly higher (χ2 = 7.77, P = 0.005) in the renal cell carcinoma tissue (63.6% [21/33]) compared with the normal renal tissue (16.7% [two of 12]) samples. The rate of VEGF-A expression was also significantly higher (χ2 = 5.16, P = 0.023) in the renal cell carcinoma tissue (51.5% [17/33]) compared with the normal renal tissue (8.3% [one of 12]) samples.
Representative photomicrograph showing a human renal cell carcinoma tissue sample with positive immunohistochemical staining for cyclo-oxygenase-2 (streptavidin–biotin–peroxidase staining; original magnification× 400). Representative photomicrograph showing human renal cell carcinoma tissue with positive immunohistochemical staining for vascular endothelial growth factor-A (streptavidin–biotin–peroxidase staining; original magnification× 400).

Correlation between COX-2 and VEGF-A expression in renal cell carcinoma
Correlation between cyclo-oxygenase-2 (COX-2) and vascular endothelial growth factor-A (VEGF-A) expression in human renal cell carcinoma tissue samples (n = 33 cases), assessed by immunohistochemical methods using rabbit antihuman COX-2 monoclonal primary antibody and mouse antihuman VEGF-A monoclonal primary antibody. κ = 0.389, P = 0.021.
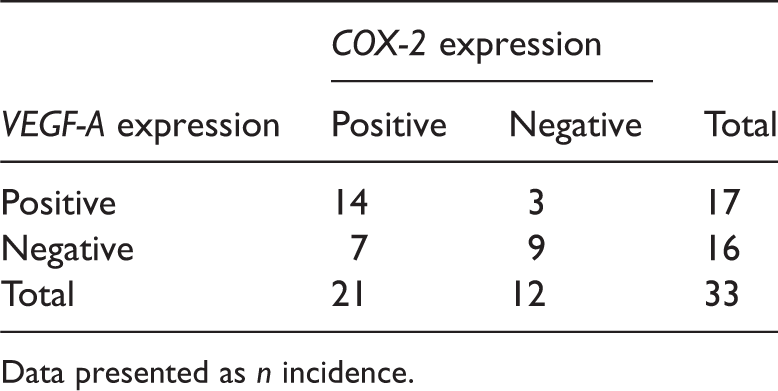
Data presented as n incidence.
Relationship between microvessel density and expression of COX-2 and VEGF-A in renal cell carcinoma
Microvessel density was significantly higher (t = 5.30, P < 0.001) in renal cell carcinoma tissue samples that scored positive for COX-2 expression compared with those scoring negative for COX-2 expression (57.80 ± 8.26 versus 41.75 ± 8.57, respectively). Microvessel density was also significantly higher (t = 2.41, P < 0.022) in renal cell carcinoma tissue samples that scored positive for VEGF-A expression compared with those scoring negative for VEGF-A expression (55.76 ± 9.24 versus 48.31 ± 8.50, respectively).
Discussion
Cyclo-oxygenase is an important rate-limiting enzyme involved in the synthesis of prostaglandins, which can metabolize arachidonic acid into prostaglandin-related products, thereby maintaining the body’s pathophysiological processes. 14 Mammalian COX includes at least two types of isoenzymes (COX-1 and COX-2); of these, the inducible COX-2 is widely studied in cancer research. Studies have shown that COX-2 is only minimally detected in normal tissues, but can be rapidly produced when stimulated by mitogens (such as cytokines, endotoxins, carcinogens and oncogenes), and is thus highly expressed in tumours.15–18 In addition, COX-2 can promote tumour development through its many functions, as it is known to induce carcinogen activation, stimulate cell proliferation, inhibit cell apoptosis, affect cell cycle, suppress immunity, promote tumour angiogenesis, and improve tumour adhesion and metastasis.16,19–24
One study found that COX-2 expression was significantly higher in renal tubular epithelial neoplasia and renal cell carcinoma than in normal kidney tissue and pyelonephritis, 25 and moderate to high COX-2 expression was reported in the renal tissues of two out of three canines with renal cell carcinoma, 26 suggesting that increased COX-2 expression may be related to the occurrence of renal tumours. COX-2 expression has been shown to correlate positively with tumour size, grade, and stage of renal cell carcinoma. 27 In addition, progression-free survival is shown to be low in patients with renal cancer and high COX-2 expression, and the survival rate of patients is negatively correlated with tumour grade, stage and COX-2 expression. 28 Consistent with the results of these published studies, the present study showed that COX-2 was overexpressed in 63.6% (21/33) of renal cell carcinoma cases, which was significantly higher than the rates of expression observed in normal tissue. The present study results suggest that COX-2 expression is related to the pathogenesis of renal cancer.
It is thought that VEGF-A is the most effective angiogenesis regulatory factor with the highest specificity. VEGF-A is also one of the determinants of tumour angiogenesis that can efficiently and specifically have mitogenic and chemotactic effects on endothelial cells, induce angiogenesis and ingrowth of host capillaries, and affect tumour growth, invasion and metastasis. 29
The pathological pattern of renal cell carcinoma is reported to be associated with VEGF-A expression. 30 One study of 99 pathologically confirmed renal cell carcinoma cases found that COX-2 and VEGF-A expression were positively correlated. 31 COX-2 has been shown to upregulate VEGF-A expression in urinary prostate cancer cell lines, and inhibition of COX-2 expression can result in inhibition of VEGF expression. 24 Another study 32 reported that COX-2 can promote the synthesis of prostaglandins (such as prostaglandin E2 and 15-Deoxy-Δ-12,14-prostaglandin J2) by interaction with the relevant receptor. 23 COX-2 can also induce upregulation of VEGF expression by entering the nucleus through nuclear receptors, disrupting the regulation of VEGF expression, leading to increased VEGF, and can induce tumour angiogenesis.23,24,32 In the present study, the VEGF-A expression rate was 51.5% (17/33) of renal cell carcinoma cases, which was significantly higher than the rate of expression in normal renal tissue surrounding the carcinoma; further analysis showed that COX-2 and VEGF-A expression were correlated. Microvessel density in COX-2-positive or VEGF-A-positive renal cell carcinoma cases was significantly higher than in renal cell carcinoma cases that were negative for COX-2 or VEGF-A (P < 0.03). These findings suggest that COX-2 and VEGF-A are both involved in tumour angiogenesis.
The results of the present study may be limited by the relatively small number of samples and the type of tissues used as controls. Further research is therefore required to understand the proliferation, invasion and metastatic mechanisms of renal cell carcinoma.
Renal cell carcinoma is an archetypal solid tumour with highly active angiogenesis, and a high level of vascularization.33–35 Because of its particular genetic properties and heterogeneity, renal cell carcinoma is extremely insensitive to conventional chemotherapy and radiotherapy. 36 Thus, renal cell carcinoma is a popular target tumour for clinical research relating related to angiogenic suppressors. 37 An investigation that used an in vitro and in vivo model suggested that a combination of several antiangiogenesis factors may be more effective than single factors alone in treating renal cancer. 37 Additional studies are necessary to clarify the angiogenic mechanisms in renal cell carcinoma and targeted antiangiogenesis therapy.38,39 The present study further supports other evidence that COX-2 and VEGF-A expression is upregulated in renal cell carcinoma, and suggests that microvessel density is increased in cancer tissues that express COX-2 and VEGF-A. These results provide additional information that will be helpful in the development of preventative and therapeutic options for renal cancer.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
