Abstract
Objective:
To investigate the correlation between plasma lysophosphatidic acid (LPA), matrix metalloproteinase (MMP)-9 and carotid atheromatous plaque stability in patients with cerebral infarction.
Method:
Patients with cerebral infarction underwent carotid artery duplex ultrasonography and transcranial Doppler monitoring for detection of microemboli. Patients were stratified by plaque type (no plaque, intima thickening, unstable plaque, stable plaque) and presence or absence of micoremboli. Plasma LPA and MMP-9 were quantified.
Result:
LPA and MMP-9 concentrations were significantly higher and microemboli positivity was significantly more common in patients with unstable plaque (n = 21) than the other three groups (intima thickening, n = 16; stable plaque n = 41; no plaque; n = 12). There was a significant positive correlation between LPA and MMP-9 concentrations.
Conclusion:
Plasma LPA and MMP-9 concentrations may be useful biomarkers in the clinical identification and prediction of unstable plaque, and in guiding treatment.
Keywords
Introduction
Lysophosphatidic acid (LPA) is a low molecular weight, mildly oxidized, low-density lipoprotein that is released from activated platelets. Via activation of its G protein-coupled receptors, LPA exerts a range of effects on the cardiovascular system including modulation of platelet activation, recruitment and activation of inflammatory cells, and migration, phenotypic modulation and proliferation of vascular smooth muscle cells.1–5 It is therefore critically involved in thrombosis and vascular diseases such as atherosclerosis and vascular remodelling.
Lysophosphatidic acid accumulates in the lipid-rich core of carotid atherosclerotic plaque 6 and is released during plaque rupture, when it activates platelets and can lead to intra-arterial thrombus formation. 7 Typically, LPA is present in very small amounts in human plasma and tissues, and is released under pathological conditions such as injury and thrombosis. 8 Tissue remodelling processes within plaque, mainly regulated by matrix metalloproteinases (MMPs), also play a major role in the pathophysiology of atherosclerosis. MMP-9, a member of the MMP superfamily, is implicated in the pathophysiology of plaque rupture, which is the most common mechanism responsible for acute ischaemic cardiac disease and cerebral stroke. 9 MMP-9 is also associated with atherosclerotic arterial remodelling 10 and is actively synthesized in vulnerable plaques. 11 Elevated plasma levels of MMP-9 in the peripheral blood have been demonstrated in patients with acute coronary syndrome (ACS) 12 and are associated with severe coronary stenosis 13 and cardiovascular mortality. 14 Elevated plasma MMP-9 concentrations in the coronary circulation of patients with ACS indicate that MMP-9 production may be enhanced in these patients.15,16 Investigation of the relationship between LPA, MMP-9, and carotid atheromatous plaque stability in patients with cerebral infarction is far from adequate, however.
The aim of the present study was to determine the relationship between plasma LPA and MMP-9, and carotid atheromatous plaque stability in patients with cerebral infarction, and to explore its significance in the evaluation of vulnerable carotid plaque.
Patients and methods
Study population
The study recruited patients diagnosed with cerebral infarction in the arteriae carotis interna system who were admitted to the Department of Neurology, Tianyou Hospital Affiliated to Wuhan University of Science and Technology, Wuhan, China, between November 2012 and April 2013. Diagnoses were made according to criteria established during the Fourth Chinese National Cerebrovascular Disease Academic Meeting (1995). 17 Exclusion criteria were: cancer; surgery or major trauma in the previous month; obvious signs or clinical evidence of hospital-acquired infection. Full clinical history and neurological examination were performed for each patient.
The Ethics Committee of Tianyou Hospital approved the study, and all patients provided written informed consent.
Carotid artery duplex ultrasonography
Carotid artery duplex ultrasonography examination was performed with an ATL HDI® 5000 imaging system (Philips, Andover, MA, USA). With the patient lying in a supine position, the extracranial carotid arteries were imaged in the longitudinal (anterior, lateral and posterior views) and transverse planes. Both common carotid arteries, carotid bifurcations and internal carotid arteries were examined for the presence of atherosclerotic plaque. When plaque was visualized, the image of the thickest plaque was frozen and the intima–media wall thickness (IMT, including the plaque) was measured with an electronic cursor and recorded as the maximal carotid plaque thickness. IMT > 1.2 mm was defined as carotid plaque, and IMT ≥0.8 mm–≤1.2 mm was defined as thickened intima. Carotid plaque was divided into four types: flat, hard, soft and mixed, according to the ultrasound audiovisual pathology classification of plaque. 18 Flat and hard types are stable, while soft and mixed types are vulnerable.
Transcranial Doppler monitoring
All patients were monitored with transcranial Doppler (TCD) for 1 h preoperatively, 3 days after onset of stroke. Continuous TCD monitoring of the bilateral middle cerebral artery was performed with an EMETC-2020 TCD unit (Nicolet Biomedical, Madison, WI, USA). Signals were recorded on digital audiotape for offline analysis and interpretation of embolic signals as described. 19 Patients were classified as microembolic signal-positive or microembolic signal-negative.
LPA and MMP-9 quantification
Peripheral blood (10 ml) was collected from each patient within 24 h after TCD monitoring for quantification of plasma LPA and MMP-9. For MMP-9, blood was immediately mixed with sodium ethylenediamene tetra-acetic acid (EDTA,) centrifuged at 3000 g for 5 min at room temperature, separated, and stored at −80℃ until use. MMP-9 was quantified using one-step sandwich enzyme immunoassay (Santa Cruz Biotechnologies, Santa Cruz, CA, USA), according to the manufacturer’s instructions.
For LPA, blood was collected in tubes containing EDTA, and plasma was collected and either processed immediately or stored at −70℃ until lipid extraction. LPA was quantified by colorimetric assay of platelet-poor plasma as described, 20 following lipid extraction at 0℃ to 4℃ to minimize damage to ester bonds. 21
Vascular risk factors
Study definitions were: smoking, current or previous tobacco use; hypertension, high blood pressure requiring treatment; diabetes, insulin- or noninsulin-dependent diabetes requiring treatment (including dietary control); hyperlipidaemia, current treatment and/or fasting lipid concentrations (total cholesterol ≥ 6.2 mmol/l or triglycerides ≥ 2.3 mmol/l).
Statistical analyses
Data were presented as mean ± SEM. Between group differences in LPA and MMP-9 were analysed using Student’s t-test for pairwise comparisons and one-way analysis of variance for multiple comparisons. Between-group comparisons of the incidence of microembolic-positivity were made using χ2-test. Pearson correlation coefficient was used to analyse the relationship between LPA and MMP-9 concentrations. P-values <0.05 were considered statistically significant. All statistical analyses were performed with SPSS® version 19.0 (SPSS Inc., Chicago, IL, USA).
Results
Demographic and clinical characteristics of patients with cerebral infarction, stratified according to carotid artery duplex ultrasonography findings.
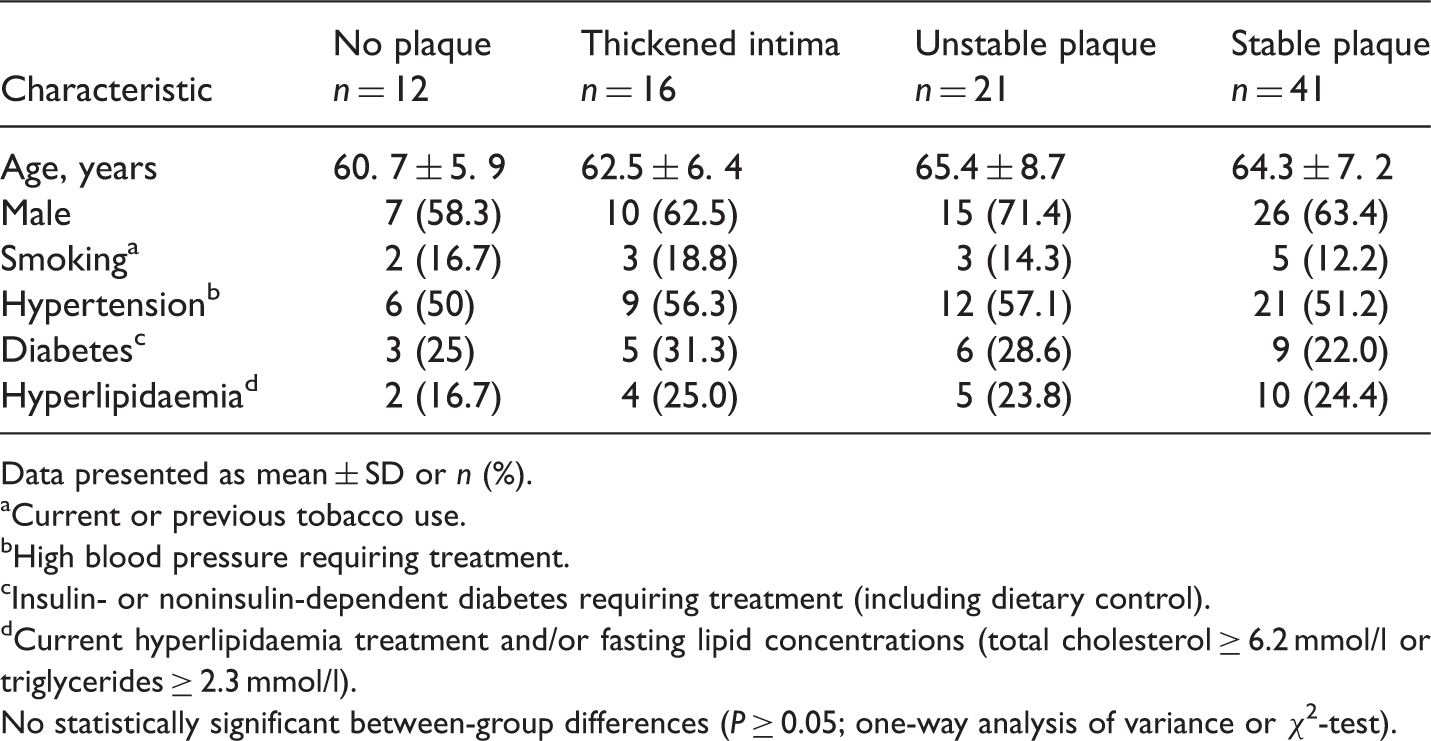
Data presented as mean ± SD or n (%).
aCurrent or previous tobacco use.
bHigh blood pressure requiring treatment.
cInsulin- or noninsulin-dependent diabetes requiring treatment (including dietary control).
dCurrent hyperlipidaemia treatment and/or fasting lipid concentrations (total cholesterol ≥ 6.2 mmol/l or triglycerides ≥ 2.3 mmol/l).
No statistically significant between-group differences (P ≥ 0.05; one-way analysis of variance or χ2-test).
Data regarding MMP-9 and LPA concentrations and the presence of microemboli in patients stratified according to plaque findings are shown in Table 2. LPA and MMP-9 concentrations were significantly higher, and microemboli-positivity was significantly more common, in patients with unstable plaque than the three other groups (P < 0.01 for LPA and MMP-9; P < 0.001 for emboli; Table 2). LPA and MMP-9 concentrations were significantly lower in the no-plaque group than in all other groups (P < 0.05 for each comparison; Table 2). In addition, LPA concentrations were significantly higher in the thickened intima group than the stable plaque group (P < 0.01; Table 2). There was a significant positive correlation between LPA and MMP-9 concentrations in the study population as a whole (r = 0.453, P < 0.05; Figure 1).
Correlation between serum lysophosphatidic acid (LPA) and matrix metalloproteinase (MMP)-9 concentrations in patients with cerebral infarction (n = 90; r = 0.453, P < 0.05). Plasma concentrations of lysophosphatidic acid (LPA) and matrix metalloproteinase (MMP)-9, and the presence of cerebral microemboli in patients with cerebral infarction, stratified according to carotid artery duplex ultrasonography findings. Data presented as mean ± SD or n (%). aP < 0.001 vs all other groups; bP < 0.01 vs all other groups; cP < 0.01 vs no plaque group; dP < 0.05 vs no plaque group; eP < 0.01 vs thickened intima group; one-way analysis of variance (a, b, c, d) or χ2-test (e).

Plasma concentrations of lysophosphatidic acid (LPA) and matrix metalloproteinase (MMP)-9 in patients with cerebral infarction, stratified according to the presence or absence of cerebral microemboli (determined by transcranial Doppler monitoring).
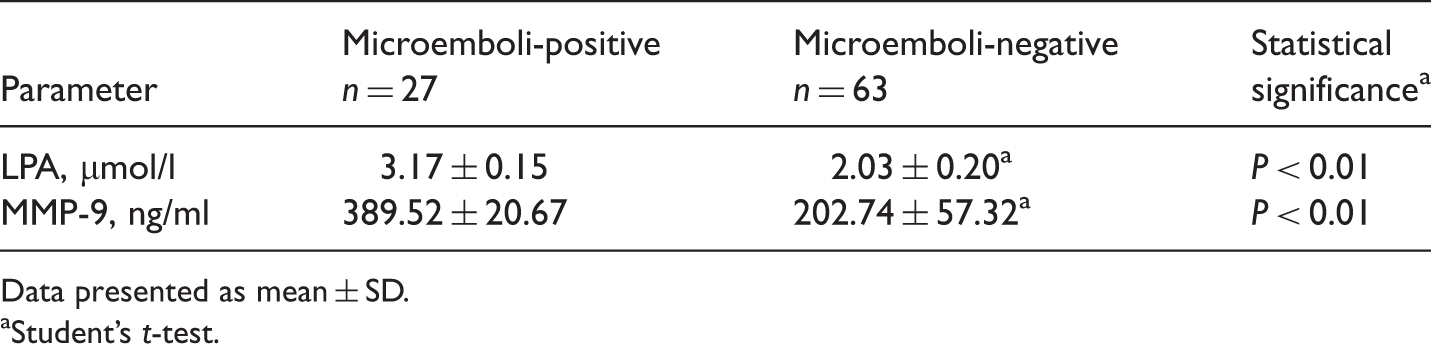
Data presented as mean ± SD.
aStudent’s t-test.
Discussion
The structural integrity of atherosclerotic plaque depends on its extracellular matrix, which is continually remodelled. 22 Acute changes within the plaque, such as haemorrhage, cap rupture and cap ulceration, precede the onset of clinical ischaemic events. 22 Each phase of the atherosclerotic process is mediated by MMPs, 23 which are the main physiological regulators of the extracellular matrix. The zinc-dependent endopeptidase MMP-9 is synthesized and secreted in monomeric form as a zymogen, 24 is associated with atherosclerotic arterial remodelling, 10 and is actively synthesized in vulnerable plaque. 11 MMP-9 affects plaque stability in association with various inflammatory cytokines.25,26
Lysophosphatidic acid regulates inflammation in many cell types inducing wound healing, cell proliferation, and the expression of intercellular adhesion molecule-1, interleukin-8, monocyte chemoattractant protein-1 and MMP-2. 27 LPA can activate mononuclear cells and T lymphocytes, and promote their expression and secretion of MMPs, 28 resulting in plaque instability and disruption. The LPA-induced migration of T cells and MMP secretion that occurs in atheromatous plaque can accelerate cap degradation and eventually lead to plaque rupture. 29 In addition, LPA can increase plaque instability by upregulating the expression of proinflammatory cytokines and adhesion molecules, promoting inflammatory cell migration and infiltration, and inhibiting apoptosis of macrophages and T lymphocytes. 29 Plaque infiltrated by a large number of inflammatory cells has been shown to have a high positive rate of microemboli, and could lead to cerebral infarction. 30 Others have demonstrated a significant positive correlation between plaque MMP-9 concentrations and TCD findings. 31
Levels of LPA in human atheromatous plaques have been shown to be 13-fold higher than in normal arterial tissue, with the highest LPA concentrations present in the lipid-rich core. 3 LPA was found to be the primary platelet-activating lipid in atherosclerotic plaques, with other lipid fractions having comparatively minor biological activity. 3 Intravascular ultrasound assessment of the culprit lesion in patients presenting with acute myocardial infarction or unstable angina found MMP-9 to be an independent predictor of plaque rupture, even after adjustment for the traditional risk factors C-reactive protein and troponin. 32 In patients with acute coronary syndrome, MMP-9 concentrations were 1.4–2.0-fold higher in those with plaque rupture compared with those without, supporting the association between plasma MMP-9 and plaque rupture. 32 Concentrations of LPA and MMP-9 in the plasma of patients with cerebral infarction in the present study were significantly higher in those with unstable plaque than in those with no plaque, thickened intima or stable plaque. In addition, the present finding that plasma LPA and MMP-9 concentrations were significantly higher in patients with evidence of microemboli than in those without microemboli suggests that LPA and MMP-9 are closely related to the formation and disruption of unstable plaque. It is possible that the high concentration of LPA present in the lipid core of atherosclerotic plaque was exposed following cap rupture, resulting in local embolus formation. This may manifest as transient ischaemic symptoms or clinical acute cerebral infarction. Local embolus after plaque rupture may then produce additional LPA, forming a positive feedback loop.
The present finding of a positive correlation between LPA and MMP-9 concentrations is in accordance with a study in a rat atherosclerotic plaque model. 33 This correlation may be due to LPA-induced production of tumour necrosis factor-α, which then enhances MMP-9 expression and activity and influences the stability of atherosclerotic plaque.25,29 Limitations of the present study were that cerebral infarction may influence LPA and MMP-9 release. In addition, plasma LPA levels may be influenced by certain medicines, such as aspirin. Also, plaque stability was only determined by the ultrasound audiovisual pathology classification; further pathological examination of plaque sections would have provided importatnt information. In conclusion, plasma LPA and MMP-9 concentrations may be useful biomarkers in the clinical identification and prediction of unstable plaque and in guiding treatment.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This work was supported by a grant from the Scientific Research Project of Hubei Provincial Department of Education, China (No. Q20121117).
