Abstract
Objectives
Levosimendan is an inotropic drug with organ-protective properties due to its activation of mitochondrial KATP channels. This prospective, randomized, double-blind, placebo-controlled study investigated whether administration of levosimendan prior to cardiopulmonary bypass could reduce organ dysfunction and influence subsequent secondary endpoints.
Patients and methods
Patients with left ventricular ejection fraction <30% scheduled for elective coronary artery bypass surgery (with or without valve surgery) received either levosimendan (12.5 mg, 0.1 µg kg−1 per min; n = 17) or placebo (n = 16) central venous infusion, immediately after anaesthesia induction, as add-on medication to a goal-orientated treatment algorithm.
Results
A total of 33 patients completed the study. There were no statistically significant differences in Sequential Organ Failure Assessment scores, survival, haemodynamic parameters, time to extubation, time in intensive care unit, need for haemodialysis or health-related quality-of-life at 6 months post operation. The levosimendan group compared with the placebo group had significantly lower use of epinephrine (35% versus 81%) and nitroglycerine (6% versus 44%) 24 h postoperation, and significantly less frequent serious adverse events (13% versus 47%).
Conclusions
These preliminary results show that timely perioperative levosimendan treatment is feasible, has a favourable safety profile safe and may help to prevent low cardiac output syndrome. However, organ function was not preserved. Further studies, using larger sample sizes, are required.
Keywords
Introduction
Many patients with advanced stages of coronary artery disease need cardiac surgery, and the high rate of comorbidities in this patient group directly increases the risk of perioperative mortality. In particular, patients with severely decreased left ventricular ejection fraction (LVEF) have a significantly increased risk of in-hospital mortality with other organ system failures, as predicted by the Sequential Organ Failure Assessment (SOFA) or the European System for Cardiac Operative Risk Evaluation (EuroSCORE).1,2 Therefore, prevention of perioperative organ system dysfunction is important for improving short- and long-term endpoints after cardiac surgery.3,4
The prevention of malperfusion and ischaemia–reperfusion-related injuries to the heart and other vital organs in high-risk patients should ideally be established before operative trauma occurs. 5 While commonly used inotropes acting as beta-adrenergic agonists or phosphodiesterase inhibitors are potent in improving haemodynamics, 6 they increase intracellular calcium levels, thereby causing elevated myocardial oxygen consumption, 7 arrhythmias and diastolic impairment, and further compromising the heart. 8 Levosimendan improves myocardial contractility without increasing myocardial oxygen demand 9 and causes coronary and peripheral vasodilatation, subsequently unloading the ventricles (inodilation). 10 These effects help to achieve an optimal cardiac index and secure adequate oxygen delivery. Of great importance is the influence of levosimendan on mitochondrial potassium channels, 11 which accounts for its protective effects on organs 12 and may decrease the incidence of postoperative organ failure.5,13–15
It has been suggested that levosimendan could be used to prevent organ failure most optimally by being applied before surgical and ischaemia–reperfusion trauma,16–18 but, to the authors’ knowledge, no study has systematically investigated this. Therefore, the aim of the present study was to investigate whether treatment with levosimendan immediately after anaesthesia induction prior to cardiopulmonary bypass (CPB) could reduce postoperative organ dysfunction and influence subsequent secondary endpoints in patients with severely reduced LVEF.
Patients and methods
Patient selection and inclusion/exclusion criteria
Between March 2008 and December 2009, patients being treated at the Campus Charité Mitte, Charité – Universitätsmedizin Berlin, Berlin, Germany were sequentially enrolled in this double-blind, single-centre, prospective, randomized, placebo-controlled trial. The study was approved by the Ethics Committee of the State Office of Health and Social Affairs (EudraCT 2007-004674-49 and ISRCTN). Written informed consent was obtained from each patient.
Patients aged ≥18 years with ischaemic cardiomyopathy, scheduled for elective coronary artery bypass graft (CABG) surgery with or without valve surgery, with LVEF ≤30% (diagnosed by preoperative echocardiography or fluoroscopy during coronary catheterization) were included.
Exclusion criteria were pregnancy and/or breast feeding, liver insufficiency Child–Pugh class B or C or Model for End-stage Liver Disease >17, disease or recent operation (<2 months prior) of the oesophagus or upper airway, neurological or psychiatric disorder, diabetes mellitus treated with sulphonylurea drugs, HIV infection or hepatitis B or C infection, or alcohol abuse.
Randomization and study drug delivery
Patients received a pseudonym and were block randomized using computer-generated randomization into the levosimendan or the placebo group. The study drug and placebo were labelled with a pseudonym to ensure blinding to the group assignment. Central venous application of the study medication was commenced immediately after induction of anaesthesia. Patients in the levosimendan group received 12.5 mg levosimendan (Simdax®, Abbott Laboratories, Abbott Park, IL, USA) at a rate of 0.1 µgkg−1 per min once, without an initial bolus, to attenuate vasodilating properties. Patients in the placebo group received water-soluble vitamin concentrate (Soluvit®, Fresenius Kabi, Uppsala, Sweden; this contains vitamins B1, B2, B6, B12 and C, pantothenic acid, biotin and folate), diluted in 5% glucose to match the colour of the levosimendan preparation; this was administered at the same infusion rate.
Intraoperative management
Anaesthesia, surgery and CPB were performed in accordance with standard operating procedures 19 and the hospital’s routine clinical practice. Routine monitoring included 5-lead ECG, pulse oximetry and bispectral index. Invasive arterial blood pressure monitoring was installed prior to intravenous (i.v.) anaesthesia induction with 0.5–1.0 µgkg−1 sufentanil (Janssen, Beerse, Belgium) and 0.2–0.3 mgkg−1 etomidate (B Braun, Melsungen, Germany). After endotracheal intubation, central venous access was established using a 4-lumen catheter as well as a continuous cardiac output/mixed venous oxygen saturation pulmonary artery catheter (Edwards Lifesciences, Irvine, CA, USA), and a urine catheter (including temperature measurement) was inserted. Transoesophageal echocardiography was performed routinely.
Anaesthesia was maintained with 0.5–1.0 µgkg−1 per min sufentanil continuous i.v. infusion and inhaled sevoflurane (Abbvie, Ludwigshafen, Germany), at 1 minimum alveolar concentration endtidal volume percentage concentration, before and after CPB. This regimen was substituted on CPB by continuous i.v. infusion of 5 mgkg−1 per h propofol (B Braun). Lungs were mechanically ventilated in pressure-controlled mode with 50 : 50 air/oxygen. Heparin was administered after sternal incision, at a dose of 400 IUkg−1 i.v., to achieve an activated clotting time of ≥410 s (Rotexmedica, Frankfurt, Germany). Normothermic CPB was established after cannulation of the right atrium (or both the venae cavae and the ascending aorta) using a nonocclusive centrifugal pump at a flow rate of 2.5 l min−1 per m2. After aortic cross-clamp, antegrade blood cardioplegia was repeatedly administered at 20-min intervals. 20
Inotropic management and CPB separation were guided using an institutional standard operating procedure algorithm (Figure 1). After cross-clamp removal, ventricular fibrillation and/or tachyarrhythmia were treated with electric defibrillation/cardioversion and 20 mgkg−1 magnesium i.v. (Inresa, Freiburg, Germany), 1–1.5 mgkg−1 lidocaine i.v. (B Braun) or 300 mg bolus amiodarone i.v. followed with 900 mg amiodarone, administered i.v. over 24 h (Sanofi-Synthelabo, Paris, France). In case of difficult CPB separation despite haemodynamic optimization, an intra-aortic balloon pump and/or a ventricular assist device (VAD) was placed, according to the team’s assessment. When successful weaning off CPB had been achieved, heparin was antagonized using 1 mg protamine i.v. for each IU of heparin given intraoperatively.
Goal-orientated treatment algorithm for inotropic support and weaning from cardiopulmonary bypass. CVP, central venous pressure; IABP, intra-aortic balloon pump; LVEDD, left ventricular end-diastolic diameter; MAP, mean arterial pressure; SvO2, mixed venous oxygen saturation; TEE, transoesophageal echocardiography.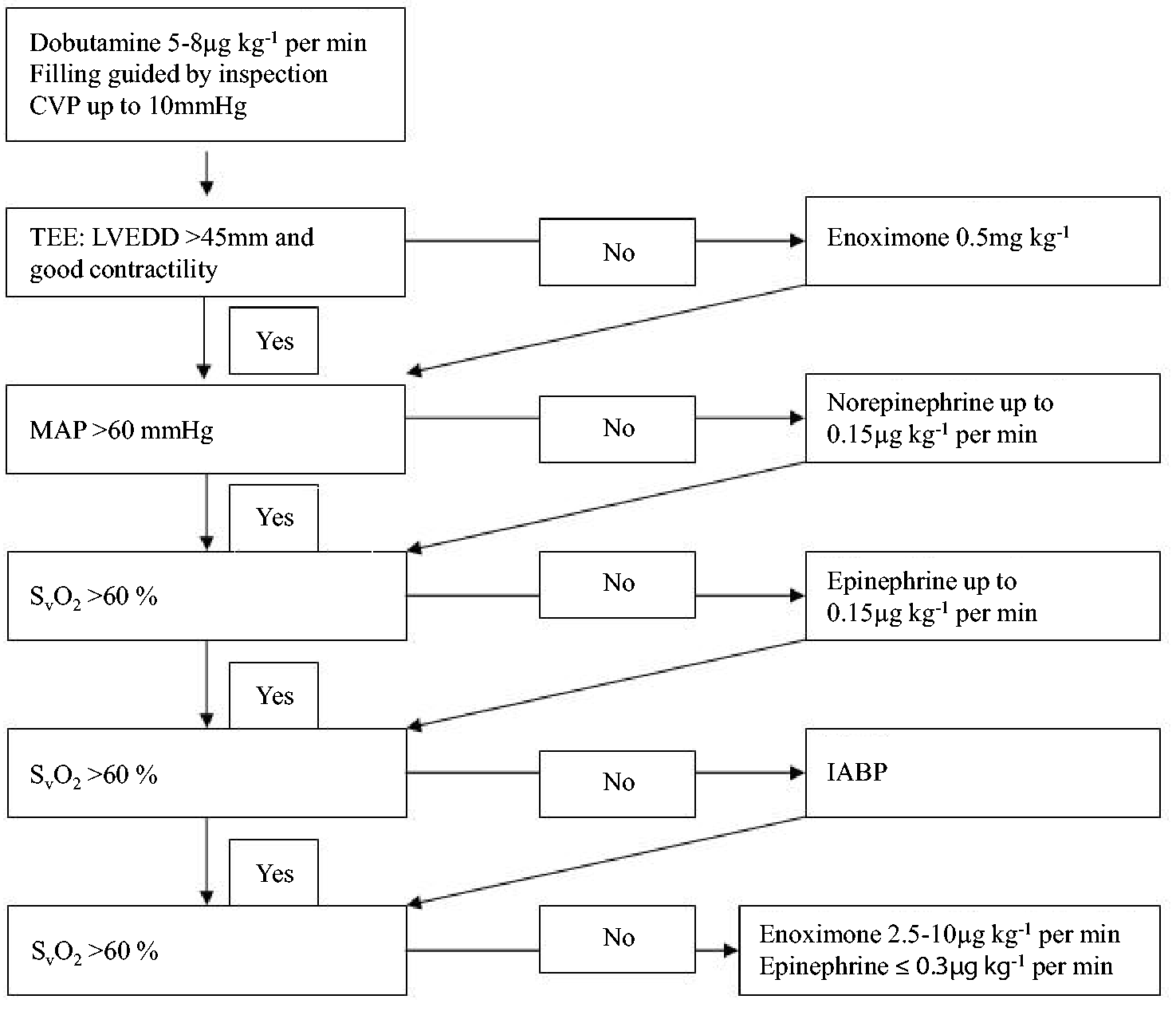
Haemodynamic management
Haemodynamic goals were defined according to guidelines 6 as follows: heart rate 80–100 beats min–1, mean arterial pressure 65–85 mmHg, central venous pressure 8–12 mmHg (at positive end-expiratory pressure 5 cm H2O), pulmonary artery occlusion pressure 12–15 mmHg, cardiac index >2.5 l min−1 per m2, stroke volume index >30 ml m–2, mixed venous oxygen saturation >65%, diuresis ≥0.5 mlkg−1 per h and haemoglobin >8 g dl−1. The study drug (or placebo) was administered as a supplementary medication and did not influence routine haemodynamic management.
ICU management
After chest closure, each patient was transferred to the intensive care unit (ICU) under sedation, intubation and mechanical ventilation. Patients were kept sedated with 1–3 mgkg−1 per h propofol i.v. and piritramid boli i.v. (Janssen) until the following criteria were met: haemodynamic goals; temperature 36–37℃; haemodynamically insignificant fluid losses via chest drain (<100 mlh−1). If mechanical ventilation was required for >12 h, analgosedation was changed to 0.01–0.2 mgkg−1 per h midazolam i.v. (Roche, Germany; to achieve a Richmond Agitation Sedation Scale score between 0 and −1) 21 combined with 0.15–0.7 µgkg−1 per h sufentanil i.v. (to achieve a Behavioural Pain Scale score < 6). 22 Mechanical ventilation was gradually reduced according to a daily algorithm 23 for which extubation required adequate respiratory efforts: FiO2 < 0.4; PaO2 > 75 mmHg; SpO2 > 95%; PaCO2 < 55 mmHg; no acidosis or alkalosis.
Discharge criteria from the ICU were sufficient spontaneous respiration with PaO2 > 65 mmHg; SpO2 > 95% at a FiO2 < 0.4 by facemask; haemodynamic stability without inotropic or vasopressor support; no significant arrhythmias; haemoglobin>8 g dl−1; chest tube drainage <50 mlh−1; diuresis ≥0.5 mlkg−1 per h; adequate neurological status.
Primary and secondary endpoints
The primary endpoints were SOFA scores (ranging between 4 and 24) on postoperative days 0–3. Secondary endpoints were haemodynamic parameters (described above); need for vasoactive medication; need for haemodialysis; ICU length of stay; 30-day survival; 6-month survival; health-related and quality-of-life at 6 months postoperation.
Data collection
Data, i.e. haemodynamic, need for vasoactive medication, etc., were collected at the following timepoints: day before surgery; after induction of anaesthesia; immediately prior to going on CPB; 10 min after CPB separation and after chest closure; 30 min after arrival in ICU; two further measurements at 6-h intervals; every 12 h until 08.00 h on postoperative day 3 or until discharge from the ICU, whichever occurred first.
The SOFA scores were measured upon arrival in the ICU (POD 0) and on postoperative days 1–3 (POD 1–3). All of the aforementioned haemodynamic parameters and medications (actual rates and cumulated doses) were registered at each data collection point. Each patient completed a health-related quality-of-life questionnaire (EQ5D) 24 6 months post operation.
The following serious adverse events were recorded: reintubation due to pneumonia and/or tracheobronchitis as diagnosed by Centers for Disease Control and Prevention/American Thoracic Society criteria (http://www.cdc.gov/pneumonia/); reintubation due to lung oedema; emergence of a sepsis (diagnosed using the Surviving Sepsis Campaign Guidelines 25 ); renal insufficiency requiring continuous renal replacement therapy; reoperation; readmission to hospital associated with the operation; persistent haemodynamic instability with the need of implantation of a VAD; cardiopulmonary resuscitation; death..
Statistical analyses
Results were expressed as mean ± SD, median (interquartile range [IQR]) or n (%). After assessment of a normal distribution of data, differences between groups for interesting clinical parameters were tested using Student’s t-test or nonparametric Wilcoxon–Mann–Whitney test. Changes in variables over time were analysed using a nonparametric analysis of longitudinal data in a two-factorial design. Frequencies were tested by exact Χ2-test on contingency tables. For cases of small samples, greater differences in sample sizes, large but unbalanced groups, data sets containing ties or sparse data, tests were carried out using an exact version. Kaplan–Meier survival curves for the time to discharge from ICU were plotted and compared using the log-rank test after checking that the proportional hazard precondition was met.
According to the hospital’s previous patient admission rate, sample size calculation was based on an expected reduction of the SOFA score between 12 points (IOR10–14) and 8 points (IOR 6–10), resulting in an effect size of 0.6745. With Power of 80% and a two-sided α of 5%, the sample size calculation for the Wilcoxon–Mann–Whitney test resulted in a minimum sample size of 39 patients per group. Statistical analyses were performed using SPSS® version 19 (SPSS Inc., Chicago, IL, USA). A two-tailed P-value < 0.05 was considered statistically significant. All tests were conducted for exploratory data analysis; therefore, no adjustments for multiple testing were made.
Results
In total, 37 patients met the inclusion criteria and were randomly assigned to receive levosimendan (n = 19) or placebo (n = 18). Four patients dropped out of the study prior to receiving the study medication due to a change in the operative status (i.e. from elective to urgent), so that 33 patients (levosimendan n = 17, placebo n = 16) completed the study protocol (Figure 2). The study was discontinued prematurely, due to reasons explained in the Discussion.
Consort flowchart of patients with left ventricular ejection fraction <30% undergoing elective coronary artery bypass surgery receiving either levosimendan (12.5 mg, 0.1 µgkg–1 per min, via central line) or placebo immediately after induction of anaesthesia as add-on medication to a goal-orientated treatment algorithm.
There were no statistically significant differences between groups in patient and surgical characteristics, intraoperative data and primary endpoints (Tables 1–3). For the primary endpoints, no significant differences in SOFA scores (over the whole time course, the maximum and the last SOFA score) were found in relation to baseline (Figure 3). For the secondary endpoint of additional use of inotropic/vasoactive drugs, significantly lower requirements for epinephrine (duration and use after 24 h) and nitroglycerine (use after 24 h) were observed in the levosimendan group compared with the placebo group (Table 4); use of norepinephrine, dobutamine and enoximone did not differ between groups. However, in the levosimendan group, significantly fewer patients required nitroglycerine after 24 h (Table 4). No significant between-group differences were observed in any of the other secondary endpoints (Table 3 and Figure 4).
Sequential organ failure assessment (SOFA) scores in patients with left ventricular ejection fraction <30% undergoing elective coronary artery bypass surgery receiving either levosimendan (12.5 mg, 0.1 µgkg−1 per min, via central line) or placebo immediately after induction of anaesthesia as add-on medication to a goal-orientated treatment algorithm. Data presented as mean ± SD. There were no statistically significant differences between the groups (P = 0.34; nonparametric analysis of longitudinal data in a two-factorial design). POD, postoperative day. Intraoperative haemodynamics in patients with left ventricular ejection fraction <30% undergoing elective coronary artery bypass surgery receiving either levosimendan (12.5 mg, 0.1 µgkg−1 per min, via central line) or placebo immediately after induction of anaesthesia as add-on medication to a goal-orientated treatment algorithm: (a) mean arterial pressure, (b) pulmonary artery occlusion pressure, (c) cardiac index, (d) stroke volume index, (e) heart rate, (f) mixed venous oxygen saturation and (g) central venous pressure. Data presented as median and interquartile range. There were no statistically significant differences between the groups (nonparametric analysis of longitudinal data in a two-factorial design). CPB, cardiopulmonary bypass; POD, postoperative day. Patient characteristics and surgical procedures in patients with left ventricular ejection fraction <30% undergoing elective coronary artery bypass surgery and receiving either levosimendan (12.5 mg, 0.1 µgkg−1 per min, via central line) or placebo, immediately after induction of anaesthesia as add-on medication to a goal-orientated treatment algorithm. Data presented as mean ± SD or n (%). Student’s t-test, or exact Χ2-test, respectively. ACE, angiotensin-converting enzyme; ASA, American Society of Anesthesiology; BMI, body mass index; CABG, coronary artery bypass graft; COPD, chronic obstructive pulmonary disease; ECG, electrocardiogram; LVEF, left ventricular ejection fraction; NYHA, New York Heart Association
26
Baseline haemodynamic data after anaesthesia induction and before start of study medication and procedure characteristics in patients with left ventricular ejection fraction <30% undergoing elective coronary artery bypass surgery receiving either levosimendan (12.5 mg, 0.1 µgkg−1 per min, via central line) or placebo immediately after induction of anaesthesia as add-on medication to a goal-orientated treatment algorithm. Data presented as mean ± SD. Student’s t-test. CI, cardiac index; HR, heart rate; MAP, arterial pressure; PAOP, pulmonary artery occlusion pressure; PAP, pulmonary artery pressure; SOFA, Sequential Organ Failure Assessment; SvO2, mixed venous oxygen saturation. Postoperative data in patients with left ventricular ejection fraction <30% undergoing elective coronary artery bypass surgery receiving either levosimendan (12.5 mg, 0.1 µgkg−1 per min, via central line) or placebo immediately after induction of anaesthesia as add-on medication to a goal-orientated treatment algorithm. Data presented as mean ± SD, median (interquartile range) or n (%). Student’s t-test, nonparametric Wilcoxon–Mann–Whitney test, or exact Χ2-test, respectively. IABP, intra-aortic balloon pump; ICU, intensive care unit; LVEF, ejection fraction; POD, postoperative day; SOFA, Sequential Organ Failure Assessment; VAD, ventricular assist device. Postoperative inotropic and vasoactive drug requirements in patients with left ventricular ejection fraction <30% undergoing elective coronary artery bypass surgery receiving either levosimendan (12.5 mg, 0.1 µgkg−1 per min, via central line) or placebo immediately after induction of anaesthesia as add-on medication to a goal-orientated treatment algorithm. Data presented as n or n (%). *P < 0.05; exact Χ2-test.



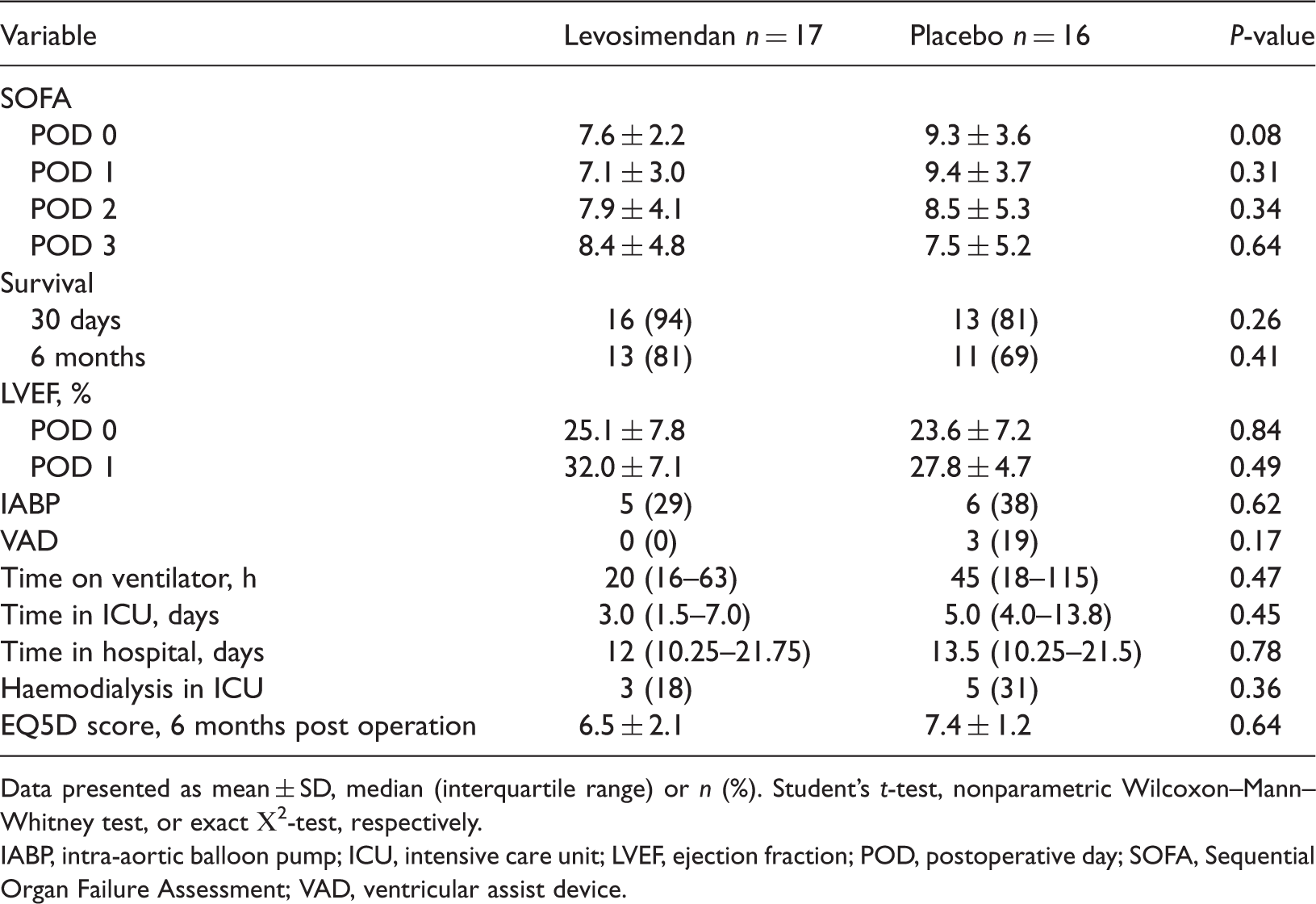

This study observed a nonsignificant trend towards shorter ICU length of stay in the levosimendan group compared with the placebo group (patients discharged up to POD 2: nine of 17 (53%) patients versus three of 16 (19%) patients, respectively; Table 5). However, Kaplan–Meier analysis of overall discharge did not result in any significant difference (P = 0.311; Figure 5).
Kaplan–Meier curves for cumulative discharge from the intesive care unit in patients with left ventricular ejection fraction <30% undergoing elective coronary artery bypass surgery receiving either levosimendan (12.5 mg, 0.1 µgkg−1 per min, via central line) or placebo immediately after induction of anaesthesia as add-on medication to a goal-orientated treatment algorithm. There were no statistically significant differences between the groups (P = 0.311; log-rank test). Cumulative discharge from the intesive care unit in patients with left ventricular ejection fraction <30% undergoing elective coronary artery bypass surgery receiving either levosimendan (12.5 mg, 0.1 µgkg−1 per min, via central line) or placebo immediately after induction of anaesthesia as add-on medication to a goal-orientated treatment algorithm. Data presented as n (%). There were no statistically significant differences between the groups (exact Χ2-test).

Furthermore, three patients in the placebo group could only be weaned from CPB with the support of a VAD: two patients requiered left VADs and one required a biventricular VAD. Only one of these patients survived at 30 days postoperation, and none survived at 6 months postoperation. Finally, the rates of serious adverse events were significantly reduced in the levosimendan group compared with the placebo group (13 versus 47, respectively, P < 0.05).
Discussion
Patients with severely reduced LVEF in this study did not have a better preservation of organ function after timely perioperative treatment with levosimendan, compared with patients in the control group (who received a vitamin infusion). However, despite the protocol-driven haemodynamic optimization resulting in comparable stroke volume indices and delivery of oxygen in both groups, this study demonstrated a significant reduction in the time and/or cumulative doses of epinephrine and nitroglycerine in the levosimendan group, compared with the control group. There was a nonsignificant trend towards reduced necessity for mechanical VAD and a significantly reduced number of serious adverse events in patients treated with levosimendan, compared with controls.
These preliminary results indicate that levosimendan acts as an inotropic drug in patients with severely reduced LVEF and reduces the need for additional inotropic support, as reported elsewhere.27,28 Consistent with previous studies,17,29,30 the present study observed no increased need for vasoconstrictors and significantly reduced demand for epinephrine and nitroglycerine in the levosimendan group. These effects may be due to either the pharmacodynamics and pharmacokinetics of the long-acting agent levosimendan by omitting an initial bolus and using an overall low continuous infusion rate, 31 or to the goal-directed volume administration applied in this study. Also, concomitant with the significantly lower number of serious adverse events was the finding that perioperative application of levosimendan to prevent a low cardiac output state was feasible and appeared to offer a favourable safety profile, as reported elsewhere. 32
The goal-directed haemodynamic optimization during and after separation from CPB and in the ICU was successful in achieving comparable haemodynamics in both groups, and, due to the observed similar arterial haemoglobin levels in both groups, it can be concluded that all patients had the same state of oxygen delivery to tissues. A meta-analysis has shown that haemodynamic goal-directed therapy can reduce the number of patients with cardiac (e.g. myocardial infarction, arrhythmias, low cardiac output, etc) and non-cardiac (e.g. renal failure, liver insuffciency, etc.) complications after cardiac surgery. 33
Two meta-analyses of randomized controlled studies demonstrated a reduction in mortality with levosimendan.34,35 In the current study, mortality rates in the levosimendan and placebo groups after 30 days and 6 months were comparable with those reported previously, 34 but there were no significant differences in survival or organ dysfunction rates. Only three studies included in these meta-analyses included cardiac surgery patients with preoperative severely reduced LVEF,16,30,36 but none was designed to detect organ-protective effects because levosimendan was only administered as a rescue medication for low cardiac output after separation from CPB. Consistently with the current results, in all three studies levosimendan effectively reversed low cardiac output by increasing cardiac index and reducing systemic vascular resistance; however, only Levin et al. 30 was able to demonstrate reduced postoperative mortality with levosimendan (8.7% versus 25% with dobutamine). Interestingly, the mortality rate in the study by Levin et al. 30 is similar to our findings.
Surprisingly, and although not powered for in this study, a highly significant decrease in serious adverse events was observed in the levosimendan group compared with the control group. This may account for the observed trend for reduced demand of mechanical VAD in the levosimendan group. However, it may be that goal-directed therapy optimization with enoximone and/or epinephrine, which were administered more frequently in the placebo group than in the levosimendan group (Table 4), increases the occurrence of serious adverse events and therefore negatively influences recovery. 37
The present study had several limitations. First, the study concept was to give levosimendan to patients with severely impaired myocardial function, as they have the highest need of perioperative inotropic pharmacological support and prevention of mortality.38,39 Here, levosimendan certainly showed its inotropic potency in reducing the need for conventional inotropic medication, but the nonsignificant differences in the need for haemodialysis and duration of mechanical ventilation may be a result of similar arterial oxygenation levels in the treatment and control groups, the low overall number of patients and/or the fact that patients possibly were not able to take advantage of the organ-protective mechanisms of levosimendan because too much irreversible organ damage had occurred preoperatively. Secondly, the SOFA score may be not sensitive enough to detect organ- protective mechanisms of levosimendan due to reduced assay sensitivity. This study did not perform serial evaluations of cardiac troponin, other enzymic markers or brain natriuretic peptide as it was mainly interested in extracardiac organ dysfunction, although these have some predictive value in relation to organ failure.40,41 Thirdly and most importantly, due to difficulties in patient recruitment, this study did not include the intended number of patients and so did not achieve sufficient power. The review board was asked for permission to perform a blinded interim analysis after the inclusion of 37 patients because the calculation on the basis of the actual differences in SOFA scores between groups revealed that at least 236 patients would have to be included in each group to show a significant difference in the primary endpoint. Therefore, the study was discontinued prematurely.
In summary, timely administration of the long-acting inotrope levosimendan prior to surgical trauma is feasible and has a favourable safety profile; its administration may prevent low cardiac output syndrome in patients with severely reduced LVEF undergoing cardiac surgery. Levosimendan may also reduce the demand for epinephrine and nitroglycerine and decrease the rate of serious adverse events. Nevertheless, despite having mortality rates comparable with those presented by others when administering levosimendan, this underpowered study did not detect differences in preservation of organ function.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Trial registry number
This trial is registered with the European Clinical Trials Database (EudraCT 2007-004674-49).
Acknowledgements
The authors are grateful to anaesthesiologists, intensivists, cardiac surgeons and nurses at Charité – Universitätsmedizin Berlin for their help with this study.
