Abstract
Objective
A disintegrin and metalloprotease (ADAM)10 has been implicated in the progression of various solid tumours. Little is known, however, about its role in hepatocellular carcinoma (HCC). The aim of the present study was to evaluate the protein and transcript level expression of ADAM10 in HCC patients.
Methods
Samples of HCC and adjacent noncancerous liver tissue were taken during liver resection surgery. Immunostaining was used to measure ADAM10 protein expression levels and quantitative reverse– transcription polymerase chain reaction was used to measure ADAM10 mRNA expression levels. Levels of ADAM10 were compared, and a survival analysis undertaken.
Results
In total, 98 HCC patient samples were studied. There were significant associations between protein levels of ADAM10 and tumour grade, amount of tumour differentiation, tumour size and the presence of metastasis. Furthermore, ADAM10 protein expression was significantly associated with shortened patient survival.
Conclusions
ADAM10 is strongly expressed in a large proportion of HCC cases, which is in agreement with findings in other tumour entities. Expression of ADAM10 may serve as a useful molecular marker for HCC.
Introduction
Hepatocellular carcinoma (HCC) is one of the most frequently occurring tumours worldwide, affecting more than 500,000 people each year.1,2 Primary liver cancer is the fifth most common malignancy in men and the ninth in women.1,2 As with other cancers, HCC tends to show early invasion into blood vessels as well as intrahepatic metastasis. 3 Although there have been developments in surgical strategies and in clinical care, the overall outcome for HCC patients remains very poor as HCC is commonly detected at a late stage when therapeutic options are limited. 4 Thus, there is a need to understand the molecular mechanism of this cancer and to develop sensitive and specific molecular markers and novel therapies. Some molecular markers have been reported for diagnosis of HCC. However, they do not provide information on the prognosis of the tumour. 5
A disintegrin and metalloproteinase (ADAM)10 belongs to the disintegrin and metalloproteinase (ADAM) gene family. This family is a class of type I transmembrane proteins that participate in a wide range of physiological functions. They can degrade the extracellular matrix and control cell adhesion and movement through regulation of intercellular adhesion, protease activity and cell activities that are closely related to the metastasis of human tumours.6,7 Some members of the ADAM family, ADAM9, ADAM10 and ADAM17, are closely involved in tumorigenesis and the development and metastasis of tumours.8–10 ADAM10 has been reported to play important roles in cell migration, tumour development and metastasis by proteolytic shedding of cell surface proteins.11,12 , In addition, an increasing body of evidence indicates that ADAMs are involved in promoting tumour formation and progression of HCC.13–15 These findings strongly imply that ADAM10 plays an essential role in tumour migration and invasion.
Overexpression of ADAM10 has been demonstrated in colon cancer 16 and oral squamous cell cancer 17 However, the relationship between the expression of ADAM10 in HCC and patient prognosis has not been established. In this study, ADAM10 transcription and protein expression was studied to evaluate its diagnostic or prognostic value in HCC.
Materials and methods
Patients and tissue samples
All patients gave written informed consent to participate in the study. This study was approved by the Ethics Committee of Jilin University, Changchun, Jilin Province, China.
Consecutive patients with HCC, diagnosed according to World Health Organization criteria, 18 who underwent liver resection between June 2008 and August 2012 at the First Hospital of Jilin University, Changchun, China, participated in this study. None of the patients had received chemotherapy or radiotherapy before surgery. During the surgical procedure, samples of malignant liver tissue and samples from adjacent noncancerous tissue (>5 cm away from the tumour site, cirrhosis tissue was excluded) were taken. All specimens were fixed in 10% neutral buffered formalin.
Diagnosis and staging of HCC was performed according to the tumour/lymph nodes/metastasis (TNM) classification system: each of the three elements, tumour, lymph nodes and metastasis, are graded for each patient and the combination of TMN values correspond to a stage (0 to IV) of an individual cancer, with 0 being the least serious and IV the most serious. 19 For analysis of survival time, the date of surgery was used as the beginning of the follow-up period. Patients who died from diseases other than HCC or from unexpected events were excluded from the analysis. Follow-up was continued until June 2013.
Real-time quantitative reverse transcription–polymerase chain reaction (RT–PCR)
Total RNA was isolated from 50 mg formalin-stabilised liver tissue using the RNeasy Mini Kit (Qiagen GmbH, Hilden, Germany) according to the manufacturer’s instructions. Then, 1 µg RNA was reverse transcribed into cDNA using a Primescript™ RT reagent kit (Takara Biotechnology (Dalian) Co. Ltd, Dalian, China) according to the manufacturer’s instructions. Real-time quantitative PCR was performed incorporating SYBR® Green fluorescent dye (Takara Biotechnology (Dalian) Co. Ltd), on a Rotor-Gene 3000 real-time PCR apparatus (Corbett Research Pty Ltd, Sydney, Australia). The primer sequences were as follows: ADAM10, forward: 5'-CTG CCCAGCATCTGACCCTAA-3', reverse: 5'-TTGCCATCAGAACTGGCACAC-3'. An internal control, β-actin, was used to evaluate the relative expression of ADAM10; primer sequences for β-actin were, forward: 5'-GATCATTGCTCCTC CTGAGC-3' and reverse: 5'-ACTCCTGCTTGCTGATCCAC-3'. The PCR conditions were an initial 2 min at 95℃ for predenaturation, followed by 40 cycles of denaturation at 95℃ for 10 s and annealing/extension at 60℃ for 20 s. The amplification specificity was checked by a melting curve analysis. The PCR products, a band of 205 bp for β-actin and a band of 189 bp for ADAM10 were visualized by 2% gel electrophoresis. The 2−ΔΔCT method 20 was used to calculate the relative abundance of the target gene expression using Rotor-Gene Real-Time Analysis Software, version 6.1.81 (Corbett Research Pty Ltd) with the target gene mRNA level normalized to the β-actin mRNA level. All experiments were performed in triplicate and a mean value was calculated.
Immunohistochemistry
Formalin-stabilised liver tissue specimens were embedded in paraffin and cut into 3 µm sections for use in immunohistochemistry. Sections were dewaxed in xylene, rehydrated in alcohol in descending percentage, and blocked for endogenous peroxidase and avidin/biotin activities with 3% bovine serum albumin in 0.01 M phosphate buffered saline (PBS, pH 7.2). Sections were incubated with mouse monoclonal antibody against human ADAM10 (Santa Cruz Biotechnology, Santa Cruz, CA, USA) at a dilution of 1:1000 overnight at 4℃. Samples were then washed in 0.01 M PBS (pH 7.2) for 5 min before incubation with biotin-labelled rabbit anti-mouse antibody (1:100; ZSGB-Bio, Beijing, China) for 2 h at 37℃. After washing with PBS three times, the immunostain was visualized with a streptavidin–peroxidase reaction system (Wuhan Boster Biological Technology Ltd, Wuhan, China), and developed with diaminobenzidine hydrogen peroxide (Wuhan Boster Biological Technology Ltd). The intensity of the staining was graded as: 0, if no immunoreactive cells were observed (negative); 1+, if the proportion of immunoreactive cells was <25%; 2+, if the proportion of immunoreactive cells was 25–75%; 3+, if the proportion of immunoreactive cells was >75%. Samples graded as 0 or 1+ were classified as negative, and samples graded as 2+ or 3+ were classified as positive for ADAM10.
Statistical analyses
The Statistical Package for Social Sciences, SPSS® version 14.0 (SPSS Inc, Chicago, IL, USA) was used to analyse the study results. Student’s t-test was used to compare differences between group means. The relationship between ADAM10 protein expression and clinical and pathological variables was analysed using Pearson’s χ 2 test. The relationship between ADAM10 protein expression and patient survival was analysed using univariate analysis (Kaplan–Meier). A P-value <0.05 was considered statistically significant.
Results
Characteristics of the 98 patients with hepatocellular carcinoma enrolled in this study, and the pathological features and level of a disintegrin and metalloproteinase (ADAM)10 expression in samples of their tumour.
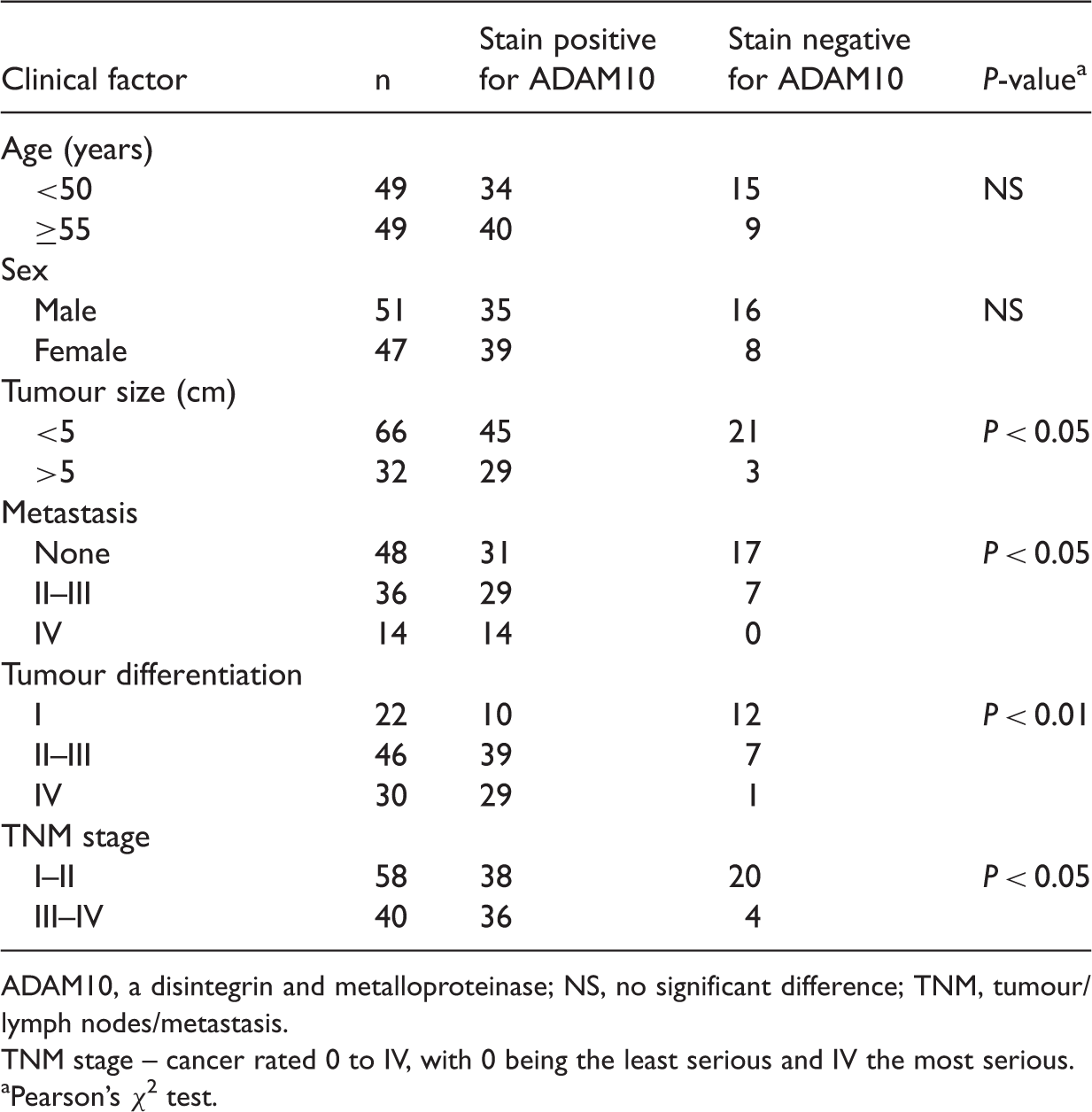
ADAM10, a disintegrin and metalloproteinase; NS, no significant difference; TNM, tumour/lymph nodes/metastasis.
TNM stage – cancer rated 0 to IV, with 0 being the least serious and IV the most serious.
Pearson’s χ 2 test.
Analysis of ADAM10 mRNA expression in HCC tissue and adjacent non-tumorous liver tissue showed that ADAM10 mRNA expression was significantly higher in cancerous tissue than in non-cancerous tissue samples (mean difference 3.1 times higher, range 0.8–11.8; P < 0.01) Figure 1.
Relative expression of a disintegrin and metalloproteinase (ADAM)10 in hepatocellular carcinoma (HCC) and adjacent healthy tissue samples from 98 patients requiring surgical hepatectomy. P < 0.05, HCC tissue versus adjacent healthy tissue (Student’s t-test).
Expression of ADAM10 was absent or sporadic in non-tumorous liver tissues (Figure 2A). The ADAM10-positive cells in HCC tissues showed unequivocal staining in the cytoplasm and cell membrane. Areas representing apparent invasive fronts appeared to be the most intensely stained, and strongly positively stained tumour cells could be seen in areas adjacent to vessels (Figure 2B). Of the 98 patients with HCC, 74 (75.6%) were evaluated as ADAM10-positive. The distribution of tumour cells with ADAM10 immunoreactivity was diffuse or focal within the same liver sample.
Cellular distribution of a disintegrin and metalloproteinases (ADAM)10 expression in samples of hepatocellular carcinoma (HCC) and samples of adjacent non-tumorous liver tissue: A, No ADAM10 staining was seen in nontumorous liver tissues; B, ADAM10-positive staining was seen in HCC samples.
Testing the association between ADAM10 immunostaining with the clinicopathological parameters of the patients with HCC showed no significant differences with regard to patient sex or age. The ADAM10-positive tumours were of larger size, were poorly differentiated, had a higher TNM stage and were more likely to have metastasis than the ADAM10-negative tumours (P < 0.01, Table 1).
The relationship between ADAM10 protein expression and patient survival was explored with a Kaplan–Meier analysis. Patients with ADAM10-positive HCC had a significantly poorer outcome than those with ADAM10-negative HCC (P < 0.01, Figure 3).
Kaplan–Meier survival curves for 98 patients with hepatocellular carcinoma (HCC) who underwent surgical hepatectomy and provided liver tissue samples for a disintegrin and metalloproteinases (ADAM)10 expression analysis. Intensity of ADAM10 staining was graded as: 0, if no immunoreactive cells were observed (negative); 1+, if the proportion of immunoreactive cells was<25%; 2+, if the proportion of immunoreactive cells was 25–75%; 3+, if the proportion of immunoreactive cells was >75%. Samples graded as 0 or 1+ were classified as negative, and samples graded as 2+ or 3+ were classified as positive for ADAM10.
Discussion
In this study, the association between the expression pattern of ADAM10 and the grade of HCC malignancy was characterized, as well as the prognostic value of ADAM10 in HCC.
Expression of ADAM10 was highest in tumour tissues with poor differentiation, metastases and those that were invasive, which is in agreement with previous reports that ADAM10 expression was increased in some tumours.16,17 Our results show that ADAM10 expression level was correlated with the tumour size, its differentiation level and TNM stage. In addition, the Kaplan–Meier survival analysis revealed that ADAM10 expression was significantly linked to a poor prognosis and a shorter survival time after surgical resection in HCC patients. To our knowledge, this study is the first to report that ADAM10 expression was increased in HCC samples compared with healthy, control tissue, and that the ADAM10 expression level was correlated with tumour size, differentiation level and TNM stage.
The designation of ADAMs is derived from their two transmembrane domains, which possess a disintegrin and a metalloprotease function. 21 ADAMs have the potential to regulate the remodelling of extracellular matrix proteins and to influence cell migration.2,22,23 There are many examples of expression and upregulation of proteolytic ADAMs in tumour tissues and cancer cell lines.24,25 Among them, ADAM10 is known to be involved in tumour progression.11,12,16,17,26 It has been reported that ADAM10 can cleave collagen type IV in the basement membrane, which is relevant to tumour metastasis. 11 Murai et al. showed that the cleavage of CD44 catalyzed by ADAM10 contributed to the migration and invasion of glioblastoma tumour cells. 12 Lee et al. reported that ADAM10 was upregulated in melanoma metastases compared with primary melanomas. 26 In another study, Gavert et al. reported that the expression of ADAM10 was detected at the invasive front of human colorectal tumour tissues. 16 Ko et al. demonstrated an overexpression of ADAM10 at the mRNA level in oral squamous cell carcinoma. Gaida et al. showed that ADAM10 expression was upregulated in pancreatic ductal adenocarcinoma and was involved in migration and invasion. 27 Carloni et al. showed that ADAM10 contributes to cancer cell fusion. 28 In the current study, our data showed that at the mRNA and protein level, ADAM10 was significantly upregulated in HCC compared with adjacent normal tissue. This suggests that ADAM10 has diagnostic value.
The limitations of the study include: (1) expression of ADAM10 in cirrhotic adjacent non-cancerous tissue was not measured; (2) alternative classifications systems such as the BCLC classification or the CLIP score were not used to verify ADAM10 as a predictive marker; (3) serial staining with both anti-alkaline phosphatase and peroxidase, which would have strengthened the findings, was not carried out. Although the present study has shown that ADAM10 may have diagnostic value in HCC, the detailed molecular mechanisms for the role of ADAM10 in HCC remains unclear. These should be examined in further studies.
In conclusion, our data showed that ADAM10 is overexpressed in HCC tissues, which is consistent with findings in other tumour entities. In addition, we have shown that ADAM10 may be an independent prognostic marker of overall survival following hepatectomy, which could provide a theoretical basis for the application of immunohistochemical or quantitative RT–PCR ADAM10 detection in biopsy materials for diagnostic HCC pathology.
Footnotes
Declaration of conflicting interest
The authors had no conflicts of interest to declare in relation to this article.
Funding
The authors gratefully acknowledge the financial support provided by the Health Bureau of Jilin (209Z0198).
