Abstract
Objective
To investigate the relationship between the serum concentration of high mobility group box 1 protein (HMGB1) and oxidative stress in patients with atrial fibrillation (AF).
Methods
Patients with AF (paroxysmal or persistent) and matched control subjects were recruited. Serum HMGB1 concentration and malondialdehyde (MDA) and superoxide dismutase (SOD) activity were determined.
Results
Serum hs-CRP and HMGB1 concentrations and MDA activity were significantly higher in patients with persistent AF (n = 33) or paroxysmal AF (n = 53) than in controls (n = 30). Serum SOD activity was significantly lower in both patient groups than in controls. In the patient group, HMGB1 concentration was significantly positively correlated with MDA activity (r = 0.535), and negatively correlated with SOD activity (r = −0.491). MDA, SOD, hs-CRP and HMGB1 were significant independent predictors of AF.
Conclusions
Increased oxidative stress may contribute to increased HMGB1 concentrations in patients with AF. Inhibition of oxidative stress may provide a potential therapeutic strategy for AF.
Introduction
Atrial fibrillation (AF) is the most common sustained arrhythmia seen in clinical practice. The prevalence of AF is strongly age-dependent, affecting ∼1% of people aged <65 years and 5% of those >65 age years.1,2 Studies suggest that inflammation and oxidative stress are involved in the development of AF,3–5 but the underlying pathophysiological mechanisms remain unclear.
High mobility group box 1 protein (HMGB1), a nonchromosomal nuclear protein, is released by necrotic and apoptotic cells, as well as by activated innate immune cells (such as macrophages and monocytes). 6 HMGB1 may act as a proinflammatory cytokine in cardiovascular diseases including AF,7–9 and plays a critical role in the pathogenesis of sepsis. 10 The incidence of paroxysmal AF is high in critically ill patients with sepsis, suggesting that HMGB1 may contribute to the genesis of paroxysmal AF in these patients. 11 Serum concentrations of HMGB1 are significantly increased in patients with both paroxysmal and persistent AF, 12 contributing to the growing body of evidence of a pathogenic link between HMGB1 and AF. 13
The aim of the present study was to investigate the relationship between HMGB1 and oxidative stress in patients with AF.
Patients and methods
Study population
The study recruited consecutive patients with AF attending the Department of Cardiology, Huangshi Central Hospital, Huangshi, China for initial diagnosis and treatment between 1 May 2011 and 28 Feb 2013. Age- and sex-matched individuals without AF were recruited from the general population of Huangshi, China as controls. All participants provided a detailed medical history and underwent a physical examination including routine biochemical testing and 12-lead surface electrocardiogram. Valvular functions, left ventricular size (left ventricular end-diastolic dimension, LVEDD) and function (left ventricular ejection fraction, LVEF), together with left atrial diameter (LAD; measured via parasternal long-axis view) were evaluated by transthoracic echocardiography in all participants. Exclusion criteria for all participants were: (i) aged <20 years or >75 years; (ii) structural heart disease; (iii) coronary artery disease; (iv) left ventricular ejection fraction <45%; (v) other types of arrhythmia; (vi) surgery or stroke within 6 months of study entry; (vii) history of infection, chronic inflammatory, hepatic or malignant disease, chronic renal failure, autoimmune disease, abnormal thyroid function or electrolyte imbalance; (viii) use of anti-inflammatory drugs such as corticosteroids and nonsteroidal anti-inflammatory drugs (excluding aspirin).
The study protocol was approved by the Medical Ethics Committee of Huangshi Central Hospital and the study was conducted according to the guidelines outlined in the Declaration of Helsinki. All study participants provided written or verbal informed consent.
AF classification
Duration of AF was determined by patient-reported, well-defined, abrupt-onset palpitation with subsequent electrocardiographic evidence of AF at the time of presentation, and was classified according to published guidelines. 14 The study did not consider investigating the duration of paroxysmal or persistent AF in detail as this information could not be accurately recorded. However, AF lasting ≤7 days was defined as new onset and was considered recurrent after two or more episodes; recurrent AF was designated paroxysmal if it terminated spontaneously, and persistent when sustained >7 days (this designation may include permanent AF). Initial diagnostic AF was therefore either paroxysmal or persistent. Lone AF was defined as AF occurring in the absence of structural heart disease and hypertension.
Sample collection and biochemical investigation
Venous blood (2 ml) was collected from all participants between 07.00 h and 08.00 h, after an overnight fast. Serum was prepared using standard methods and stored at −70℃ until use (∼1 month later). High-sensitivity C-reactive protein (hs-CRP) was quantified with a Hitachi 912 Analyzer (Roche Diagnostics, Basel, Switzerland) and HMGB1 concentration was determined using a commercially available enzyme-linked immunosorbent assay (ELISA) kit (HMGB1 ELISA kit II; Shino-Test Corporation, Tokyo, Japan), according to the manufacturer’s protocol. Malondialdehyde (MDA) and superoxide dismutase (SOD) activity (indices of oxygen free radicals and lipid superoxide levels, respectively) were quantified using commercial assay kits, according to the manufacturer’s; both were from Nanjing Jiancheng Bioengineering Institute, Nanjing, China.
Study definitions
The study used the following definitions. Alcohol use: equivalent to ≥50 ml/day white wine in the 3 months prior to enrolment; hyperlipidaemia: low-density lipoprotein cholesterol ≥4.14 mmol/l; hypertension: systolic blood pressure ≥140 mmHg or diastolic blood pressure ≥90 mmHg; diabetes: one of (i) two fasting plasma glucose levels ≥7.0 mmol/l, (ii) two 2-h postprandial plasma glucose readings ≥11.1 mmol/l after a glucose load of 75 g, (iii) two casual glucose readings ≥11.1 mmol/l, or (iv) taking oral hypoglycaemic drugs or parenteral insulin.
Statistical analyses
Data were presented as mean ± SD or n (%). Proportions were compared with χ2-test or Fisher’s exact test. Comparisons among groups were made using one-way analysis of variance or Welch’s t-test, with Student–Neuman–Keuls or Dunnett’s T3 tests for post-hoc multiple comparisons. Pearson’s correlation coefficient was used to assess the relationship between serum HMGB1 concentrations and other parameters. Multiple logistic regression analysis was used to assess the risk factors for AF. Statistical analyses were performed with SPSS® version 16.0 (SPSS Inc., Chicago, IL, USA) for Windows®. P-values < 0.05 were considered statistically significant.
Results
Demographic and clinical characteristics of patients with paroxysmal or persistent atrial fibrillation and control subjects included in a study to determine the relationship between serum concentration of high mobility group box 1 protein and oxidative stress.
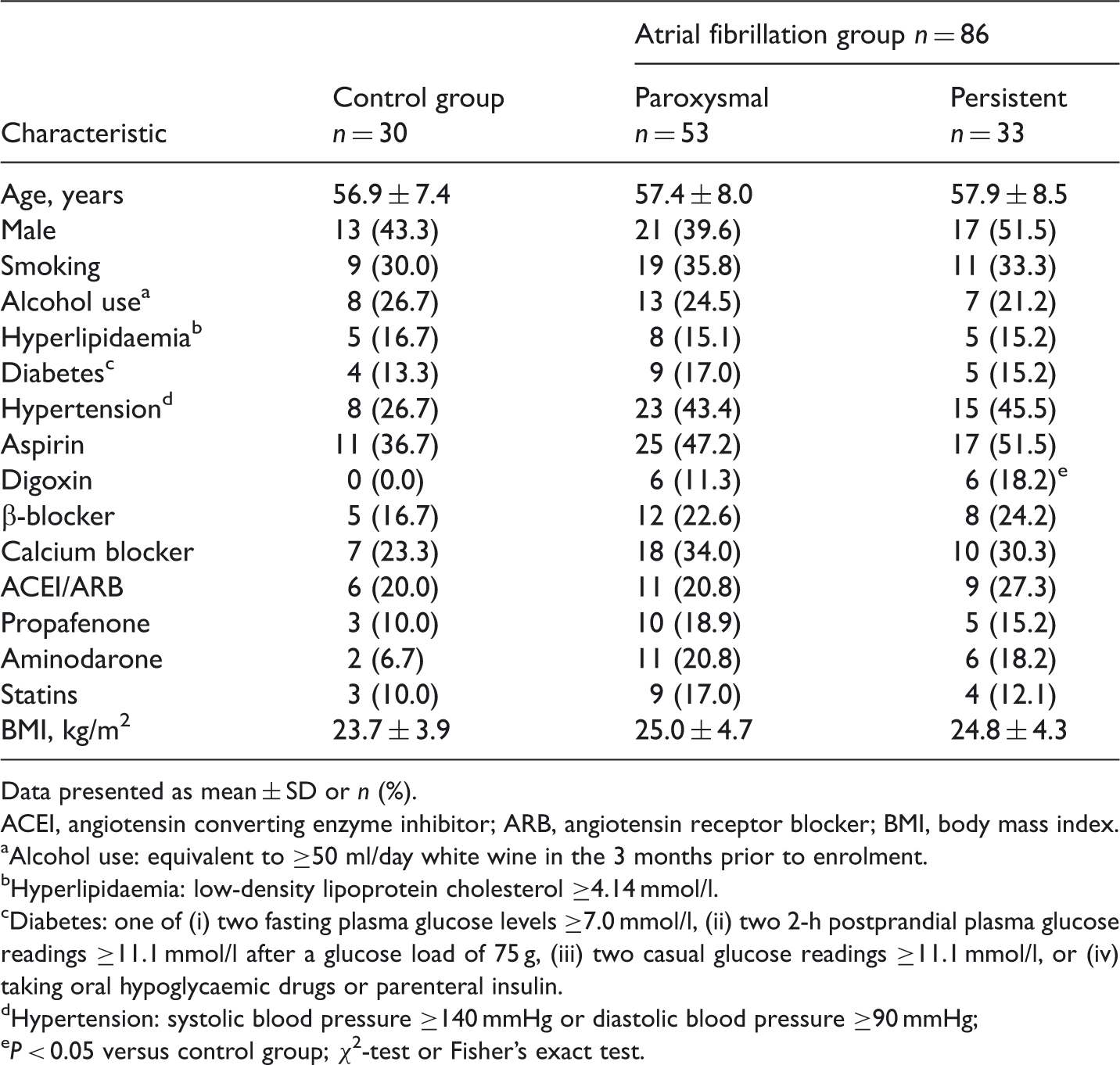
Data presented as mean ± SD or n (%).
ACEI, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker; BMI, body mass index.
Alcohol use: equivalent to ≥50 ml/day white wine in the 3 months prior to enrolment.
Hyperlipidaemia: low-density lipoprotein cholesterol ≥4.14 mmol/l.
Diabetes: one of (i) two fasting plasma glucose levels ≥7.0 mmol/l, (ii) two 2-h postprandial plasma glucose readings ≥11.1 mmol/l after a glucose load of 75 g, (iii) two casual glucose readings ≥11.1 mmol/l, or (iv) taking oral hypoglycaemic drugs or parenteral insulin.
Hypertension: systolic blood pressure ≥140 mmHg or diastolic blood pressure ≥90 mmHg;
P < 0.05 versus control group; χ2-test or Fisher’s exact test.
Echocardiographic parameters and serum oxidative stress/inflammatory markers in patients with paroxysmal or persistent atrial fibrillation and control subjects.

Data presented as mean ± SD.
LAD, left atrial diameter; LVEDD, left ventricular end-diastolic dimension; LVEF, left ventricular ejection fraction; hs-CRP, high-sensitivity C-reactive protein; HMGB1, high mobility group box 1 protein; MDA, malondialdehyde; SOD, superoxide dismutase.
P < 0.05 versus control group, bP < 0.05 versus paroxysmal AF group; all data were normally distributed; one-way analysis of variance with Student–Neuman–Keuls for post-hoc multiple comparisons.
In the total patient group (n = 86) HMGB1 concentration was significantly positively correlated with hs-CRP concentration (r = 0.507, P < 0.05) and MDA activity (r = 0.535, P < 0.05), and negatively correlated with SOD activity (r = −0.491, P < 0.05). There were no significant correlations between HMGB1 and any other factor.
Malondialdehyde (odds ratio [OR] 1.705; 95% confidence intervals [CI] 1.308, 2.545), SOD activity (OR 0.578; 95% CI 0.263, 0.996), hs-CRP (OR 1.530; 95% CI 1.287, 1.951) and HMGB1 concentrations (OR 1.832; 95% CI 1.487, 2.454) were all significant independent predictors for AF.
Discussion
The present study found that the serum HMGB1 concentration was significantly higher in patients with paroxysmal or persistent AF compared with healthy control subjects. In addition, the serum HMGB1 concentration correlated positively with the hs-CRP concentration, which was also significantly increased in patients compared with controls. hs-CRP is an important predictor of successful cardioversion and subsequent maintenance of sinus rhythm in patients with AF. 3 The current data were consistent with studies demonstrating that HMGB1 expression was closely correlated with hs-CRP in patients with AF,12,15 suggesting possible cross-talk between HMGB1 and hs-CRP. HMGB1 functions as a proinflammatory cytokine, upregulating the expression of interleukin (IL)-1, IL-6, tumour necrosis factor-α, and macrophage inflammatory proteins-1α and -1β, thus reinforcing the inflammatory process6,16,17 Upregulation of HMGB1 and other proinflammatory cytokines in patients with AF may be part of the pathophysiological process of this disease.3–5
A growing body of evidence suggests that oxidative stress may cause structural and electrical remodelling of the atrium, and play an important role in the pathogenesis of AF.5,18,19 In the current study, patients with AF exhibited significantly increased serum MDA activity (a reactive oxygen species) and significantly decreased SOD activity (a key antioxidant enzyme) compared with controls. Oxidative stress may be involved in the release of proinflammatory cytokines including HMGB1: hydrogen peroxide has been shown to stimulate macrophages and monocytes to release HMGB1. 20 In the present study, HMGB1 concentrations were positively correlated with MDA and negatively correlated with SOD activity, in patients with AF. Consistent with the findings of others,5,6,20 the present data provide evidence of cross-talk between oxidative stress and HMGB1, such that oxidative stress may reinforce the effect of inflammation on the pathogenesis of AF.
The current study is limited by the small and ethnically homogenous study cohort. Further studies with larger cohorts are required, to elucidate the precise mechanisms underlying our observations.
In conclusion, the present study shows that serum HMGB1 concentrations are significantly higher in patients with AF than control subjects, and are positively correlated with MDA activity and negatively correlated with SOD activity in these patients. Oxidative stress may contribute to the increased HMGB1 concentration in patients with AF. Inhibition of oxidative stress may provide a potential therapeutic strategy for AF through preventing the expression of HMGB1.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This study was partially supported by a grant from the National Natural Science Foundation of China (No. 81100146), grant 111023 from the Fundamental Research Funds for the Central Universities and the Specialized Research Fund for the Doctoral Program of Higher Education of China (No. 20110141120060) and the Subject of Health Department of Huangshi City of China (No. 2012-139).
