Abstract
Objective
To examine the effects of caffeic acid phenethyl ester (CAPE; a component of honey bee-hive propolis with antioxidant, anti-inflammatory, antiviral and anticancer properties) on bone regeneration and fibrotic healing in a rat model.
Methods
Male Sprague–Dawley rats (n = 63; mean age 7 weeks; weight 280–490 g) were randomly divided into three groups: A, cranial defect with no bone healing treatment (n = 21); B, cranial defect treated with CAPE (n = 21); C, cranial defect treated with CAPE and β-tricalcium phosphate/hydroxyl apatite (n = 21). Rats were anaesthetized with ketamine (8 mg/100 g) by intraperitoneal injection and a cranial critical size bone defect was created. Following surgery, CAPE (10 µmol/kg) was administered by daily intraperitoneal injection. Seven rats in each group were killed at days 7, 15 and 30 following surgery. Bone regeneration, fibrotic healing and osteoblast activity were evaluated by histopathology.
Results
Statistically significant differences in healing were found between all groups. There were no statistically significant within-group differences between day 7 and 15. At day 30, bone healing scores were significantly higher in groups B and C compared with group A.
Conclusion
CAPE significantly improved bone-defect healing in a rat model, suggesting that CAPE has beneficial effects on bone healing.
Introduction
Despite advancements in oral and maxillofacial surgery and related technology, bone fractures do not always heal successfully. 1 Multiple events occur during bone fracture healing including inflammation, repair and remodelling, and the release of excessive amounts of free oxygen radicals during these processes. Free radicals are highly reactive products that may cause cellular injury or death through their effects on almost all components of the cell including lipids, proteins, DNA, carbohydrates and enzymes. Free oxygen radicals are known to have adverse effects on many processes, including bone fracture healing.2–4 The negative effects of free oxygen radicals appear to be more prominent in early phases of fracture healing, since antioxidants have been shown to be most effective at this time. 5
Caffeic acid phenethyl ester (CAPE), an active component of honey bee-hive propolis, has many biological and pharmacological properties including antioxidant, anti-inflammatory, antiviral and anticancer effects.6–8 CAPE is a specific inhibitor of the nuclear transcription factor, nuclear factor-κB. 9 CAPE has been shown to significantly suppress the lipoxygenase pathway of arachidonic acid metabolism during inflammation. 10 It has also been shown to inhibit HIV-1 integrase 11 and inhibit the proliferation of transformed cells, 12 and it induces apoptosis in transformed fibroblasts. 13 These studies show that CAPE is effective in suppressing the inflammatory compounds that cause fibrotic healing, and suggest that CAPE may have beneficial effects in treating bone fracture healing.
The present study investigated the effects of CAPE on bone fracture healing using an experimental cranial bone model in rats.
Materials and methods
Rat critical size cranial model and surgical procedure
Male Sprague–Dawley rats (n = 63; mean age 7 weeks; weight 280–490 g) were included in this study, which was conducted at the Health Institution of Research Centre, Dicle University, Diyarbakir, Turkey. Animals were individually housed in plastic cages in a controlled environment (21℃; 12-h light–dark cycle) with free access to drinking water and a diet of standard laboratory rat food pellets.
The rats were randomly divided into three groups: group A (control group), critical size cranial model with no treatment (n = 21); group B, critical size cranial model treated with CAPE (10 µmol/kg, daily intraperitoneal injection; n = 21); and group C, critical size cranial model treated with CAPE (10 µmol/kg, daily intraperitoneal injection) and β-tricalcium phosphate/hydroxyapatite bone graft substitute (n = 21).
Animals were anaesthetized with 5 mg/kg ketamine hydrochloride (Ketavett, Ratiopharm, Ulm, Germany) by intramuscular injection at the surgical site. A midline incision was made from the nasofrontal area to the external occipital protuberance along the midsagittal suture. The skin and underlying tissues including the temporal muscle were reflected bilaterally to expose the full extent of the calvaria. A calvarial through-and-through osteotomy 7.0 mm in diameter was trephined into the dorsal portion of the parietal bone on each side of the midsagittal suture using a dental handpiece and a trephine bur (Messeinger, Dusseldorf, Germany) under constant irrigation with sterile saline, and the trephined bone was removed from the surgical field.
Straumann® BoneCeramic (particle size 500–1000 μm; Institut Straumann AG, Basel, Switzerland), a medical grade, particulate form of biphasic calcium phosphate comprising 60% hydroxyapatite and 40% β-tricalcium phosphate with a wide spongiosa structure, a wide interconnected pore system (300–1500 µm) and a crystalline size of 10–60 μm, was moistened with sterile saline for 5 min before being placed into the defect to fill in the defect area.
Periosteum and skin were sutured in two layers using resorbable suture (Vicryl 3.0; Ethicon GmbH and Co. KG, Norderstedt, Germany). Upon completion of the surgical procedure, each animal received a single dose of 100 mg/kg ampicillin antibiotic by intramuscular injection.
Following surgery, seven rats per study group were killed at day 7, 15 and 30, using high dose ketamine. Bone regeneration, fibrotic healing and osteoblast activity were evaluated by histopathology.
All animal procedures were approved by the Animal Research Committee of Dicle University, Diyarbakır, Turkey and performed in compliance with the Medical Research Centre University guidelines for the care and handling of experimental animals.
Histopathological evaluation
Rat calvaria were fixed in 10% formaldehyde solution for 48 h and decalcified with 5% nitric acid. Calvarial specimens were washed with tap water, dehydrated with ascending concentrations of ethylalcohol, cleaned in xylene (Sigma-Aldrich, Steinheim, Germany) and infiltrated with paraffin. Starting from the centre of each specimen, serial sections (5 µm) were cut parallel to the midsagittal suture defect using a microtome, and stained with haematoxylin and eosin.
Tissue samples were assessed by a pathologist using a light microscope (Nicon Eclipse 80i, SEQ Enterprises, South Lakeland, FL, USA). The pathologist was blinded to the treatment group that each specimen came from. A 10-point scale 14 was used for evaluation of bone healing, where tissue was scored according to histopathological findings in the healing zone, as follows: 1, only fibrous tissue; 2, predominantly fibrous tissue; 3, equal amounts of fibrous and cartilage tissue; 4, predominantly cartilage tissue with little fibrous tissue; 5, only cartilage tissue; 6, predominantly cartilage tissue with little immature bone; 7, equal amounts of cartilage and immature bone tissue; 8, predominantly immature bone with little cartilage tissue; 9, healing with immature bone; 10, healing with mature bone.
Statistical analyses
Data were analysed using the SPSS® software package, version 15.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Shapiro–Wilk test and graphical methods were used to test normality of the data. Kruskal–Wallis and Bonferroni-corrected Mann–Whitney U-test were used for between-group comparisons at each time point, and within-group comparisons between each time point. Spearman’s correlation coefficient analysis was performed to analyse correlation between the treatment groups across all three time points. For the Bonferroni-corrected Mann–Whitney U-test analysing differences between the three groups, a P-value < 0.0167 was considered statistically significant. In all other cases, a P-value < 0.05 was considered statistically significant.
Results
Critical size cranial defect healing scores in rats (n = 63) randomly assigned to three treatment groups: A, critical size cranial defect with no treatment (n = 21); B, critical size cranial defect treated with caffeic acid phenethyl ester (CAPE, 10 µmol/kg, daily intraperitoneal injection; n = 21); C, critical size cranial defect treated with CAPE (10 µmol/kg, daily intraperitoneal injection) and β-tricalcium phosphate/hydroxyapatite bone graft substitute (n = 21).

For each group, seven rats were killed per time point (day 7, 15 and 30).
Data presented as mean ± SD.
Between-group comparisons at day 7 (P < 0.05).
Between-group comparisons at day 15 (P < 0.05).
Between-group comparisons at day 30 (P < 0.05).
Group A compared with group B (P < 0.0167).
Group A compared with group C (P < 0.0167).
Group B compared with group C (P < 0.0167).
NS, no statistically significant between-group difference (P ≥ 0.05, Kruskal–Wallis test; P ≥ 0.0167, Bonferroni-corrected Mann–Whitney U-test).
There was a positive relationship between Group C (r = 0.633, P = 0.002) and Group B (P < 0.01; Figure 1). There was no relationship between between Groups A and B (r = 0.350, P = 0.120) or between Groups A and C (r = 0.383, P = 0.087; Figure 1).
Box-whisker plot showing correlation between cranial bone healing scores in rats (n = 63) randomly assigned to three groups: A, critical size cranial defect with no treatment (n = 21); B, critical size cranial defect treated with caffeic acid phenethyl ester (CAPE, 10 µmol/kg, daily intraperitoneal injection; n = 21); C, critical size cranial defect treated with CAPE (10 µmol/kg, daily intraperitoneal injection) and β-tricalcium phosphate/hydroxyapatite bone graft substitute (n = 21). The heavy black horizontal lines for each group show the mean values, the extremities of the box are the 25th and 75th percentiles, and the error bars represent maximum and minimum values.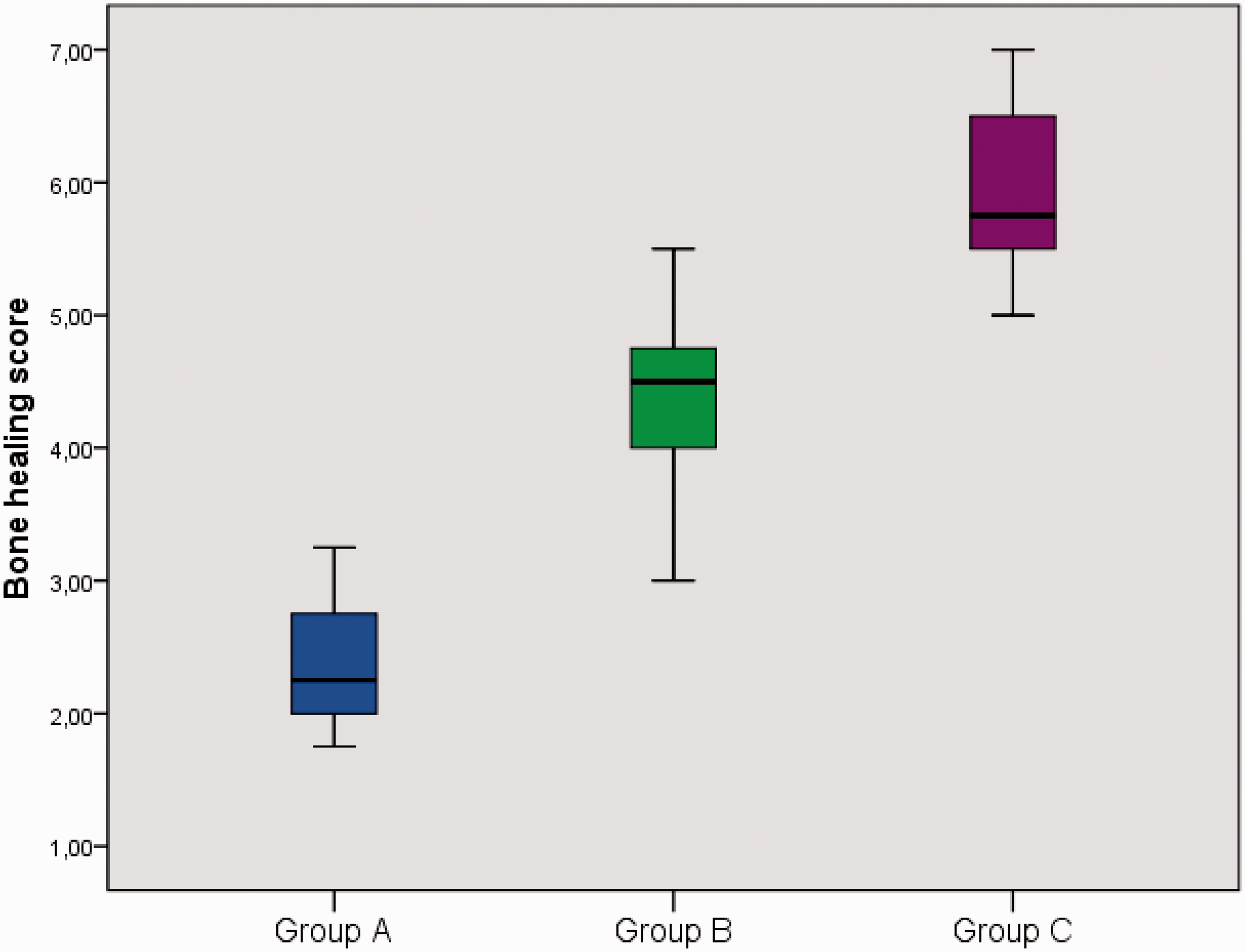
Discussion
The present study demonstrated that treatment with CAPE had a beneficial effect on bone healing in an experimental rat model. Histopathological assessment revealed significantly improved bone healing with CAPE treatment compared with controls without treatment.
Studies that examined the role of molecules involved in oxidative processes related to bone healing, reported results comparable to the findings of the present investigation. Administration of two oxidant molecules, zymosan and nicotine, was shown to result in impaired fracture healing, evidenced by radiological and histopathological findings, 3 and the beneficial effects of the antioxidant vitamin E on fracture healing, have been shown previously.15–17
CAPE exhibits significant cytotoxicity in oral cancer cells. 17 It has free radical scavenging and antioxidant properties that do not require interaction with a receptor, 18 and can be used as a free radical scavenger. 19
The antioxidant, anti-inflammatory, antiviral, and anticancer properties of CAPE have been reported previously. 20 In vivo irradiation has been shown to induce a cascade of inflammatory responses that can be reduced by CAPE treatment, 21 suggesting that CAPE may be useful in treating bone defect healing. The level of oxidative stress is most prominent during the early phases of bone healing. 22
In the present study, significantly improved bone healing was observed in rats treated with CAPE on days 15 and 30 following treatment, compared with the control group, with the greatest difference observed at day 30. These results suggest that CAPE increases bone healing levels in the late period of fracture healing.
In conclusion, the present study showed that treatment with CAPE had beneficial effects on bone healing assessed by histopathological evaluation. Current experimental evidence suggests that CAPE may have potentially beneficial effects in many clinical settings, including maxillo-facial surgery, and in the authors’ opinion, CAPE has a favourable safety profile in experimental studies. Based on the findings in the present study, together with published studies regarding the beneficial effects of CAPE, the authors believe that CAPE has the potential to be used systemically or locally to treat conditions such as bone fracture and bone defect healing. However, we recommend that studies including a greater number of animals and different doses of CAPE should be carried out before clinical trials are designed.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
