Abstract
Objectives
Neuroendocrine and haemodynamic changes were compared between single-lead atrial (AAI) or dual-chamber (DDD) pacing modes in patients with sick sinus syndrome, in a crossover study.
Methods
Inpatients scheduled for their first pacemaker implantation were screened for the following inclusion criteria: sick sinus syndrome; intact atrioventricular conduction; normal QRS interval. All study patients were implanted with a dual-chamber pacemaker, programmed for AAI or DDD pacing mode. Patients were allocated randomly to AAI followed by DDD pacing or to DDD followed by AAI pacing, each mode being applied for 72 h. Echocardiographic, electrocardiographic and neuroendocrine parameters were tested at the end of each pacing mode.
Results
From 152 inpatients screened for inclusion, 28 were selected for treatment. Plasma levels of atrial natriuretic peptide (ANP), endothelin, aldosterone and angiotension II were significantly lower, and aortic flow velocity–time integral was significantly higher, in AAI mode than in DDD mode. Aortic pre-ejection interval, interventricular mechanical delay and QRS duration were significantly higher in DDD than in AAI mode.
Conclusions
In patients with sick sinus syndrome, DDD pacing mode can induce neuroendocrine system activation, and left ventricular dysfunction and dyssynchrony. These findings discourage the routine use of DDD pacing in patients with sick sinus syndrome.
Keywords
Introduction
In patients with sick sinus syndrome, bradycardia can be treated with either a single-lead atrial pacemaker (AAI) or a dual-chamber pacemaker (DDD). Although AAI and DDD modes are both conventional physiological pacing modes for preserving atrioventricular synchrony, use of the DDD mode may lead to some degree of unnecessary pacing in the right ventricular apex (RVA), changing the electrical activation and contraction pattern of the ventricles. Clinical studies have observed left ventricular (LV) dyssynchrony during RVA pacing using echocardiography techniques.1,2 Moreover, RVA pacing may result in ventricular remodelling and decreased left ventricular ejection fraction.3–5 The choice of AAI or DDD pacing mode influences not only the mechanical systolic sequence, but also components of the neuroendocrine system such as the renin–angiotensin–aldosterone system (RAAS), the endothelin system and atrial natriuretic peptide (ANP), which are markers of clinical heart failure6,7 and atrial fibrillation.8,9 Although some related observations have been reported, 10 our group is the first to investigate all these factors together in the field of pacemaker. Classically, activation of the RAAS starts with renin production and culminates in the formation of angiotension II (a peptide that acts as a vasoconstrictor and also as a stimulator of aldosterone secretion). 11 It appears that angiotensin II contributes to the development of cardiac, vascular and renal damage. 11 Notably, aldosterone is also implicated in arterial and tissue remodelling and injury. 12 The endothelin system is an important compensatory mechanism that is activated during left ventricular dysfunction. 13 Elevated plasma levels of endothelin have been reported in association with heart disease, hypertrophy and atrial fibrillation. 14 ANP is a key regulator in the homeostasis of water excretion and has emerged as an important prognostic marker for symptomatic chronic heart failure; 15 research has demonstrated that elevated plasma ANP levels correlate with New York Heart Association functional class. 6 Although many observations on this subject have been reported, some important questions about mode selection and neuroendocrine changes remain to be elucidated. The aim of the present study was to find out whether AAI or DDD was the better physiological pacing mode, in terms of neuroendocrine and haemodynamic changes.
Patients and methods
Study population
Between May 2011 and November 2012, inpatients scheduled for their first pacemaker implantation at our institution were screened for the following study inclusion criteria: symptomatic bradycardia; documented sino-atrial block or sinus arrest with pauses >2 s or sinus bradycardia <40 beats/min for >1 min while awake; PR interval ≤0.20 s; QRS interval <0.12 s. Exclusion criteria are listed, together with a flow diagram of the study protocol, in Figure 1. Randomization for pacing mode was performed by the use of sealed envelopes, before pacemaker implantation.
Flow diagram showing the protocol for a crossover study comparing neuroendocrine and haemodynamic changes between single-lead atrial (AAI) or dual-chamber (DDD) pacing modes in patients with sick sinus syndrome.
Ethical considerations
The study was approved by the Research Ethics Committee of Tianjin Medical University and conducted in accordance with the Declaration of Helsinki. Patients volunteered to participate in the study and were included after they had given written informed consent.
Implantation and programming of pacemakers
A bipolar atrial lead was implanted in the right atrium and the ventricular lead was implanted in the RVA. An atrial pacing test was performed at 130 beats/min in all patients and 1 : 1 atrioventricular conduction was required for the study. The atrioventricular delay was progressively shortened until RVA pacing occurred without fusion, which was confirmed by the left bundle branch block pattern on the electrocardiogram. The pacing device was programmed in AAI or DDD mode. The study used a crossover design in which the patients were randomly allocated to receive either AAI or DDD mode pacing first; 72 h later the treatments of the two groups were switched. Thus, each patient received one period of AAI followed by one period of DDD mode pacing, or one period of DDD followed by one period of AAI mode pacing. Plasma concentrations of neuroendocrine factors and haemodynamic parameters of the patients were evaluated at the end of each 72-h period of AAI or DDD mode pacing.
Echocardiography and electrocardiography
Echocardiography and electrocardiography were performed at the end of the right ventricular pacing (DDD mode with shorted atrioventricular delay) and intrinsic ventricular conduction (AAI) pacing modes. Patients underwent M-mode echocardiography, two-dimensional echocardiography, pulsed wave (PW) Doppler examination and electrocardiography to measure the QRS wave interval. The left ventricular ejection fraction (LVEF) was assessed with the biplane Simpson equation. Mitral inflow velocities were obtained at the mitral leaflet tips to measure early (E) diastolic and atrial (A) wave velocities, from which the E/A ratio was calculated. In the parasternal short axis, the delay between the motion of the septal and posterior walls at the level of the papillary muscle was measured as the septal-to-posterior wall motion delay (SPWMD). A sequence lasting >130 ms was regarded as an indicator of intraventricular dyssynchrony. 16 The intervals between the onset of QRS and the beginning of ejection at the aortic and pulmonary valve levels, obtained by PW Doppler examination, were defined as the aortic pre-ejection interval (APEI) and the pulmonary pre-ejection interval (PPEI), respectively. Interventricular mechanical delay (IVMD) was defined as the interval between APEI and PPEI. An IVMD value ≥40 ms is regarded as an indicator of interventricular dyssynchrony. 16 APEI ≥40 ms reflects prolonged left ventricle activation, left ventricle dyssynchrony and delayed LV ejection. 17 Continuous wave Doppler echocardiographic measurement of aortic flow velocity–time integral (AoVTI) was used as a surrogate of stroke volume.
Neuroendocrine factor measurement
At the end of each pacing mode, plasma neuroendocrine factors were measured, as follows. After overnight fasting, 5-ml venous blood samples were collected from patients between 08:00 h and 10:00 h, following 20 min of supine rest. The blood samples were placed immediately into ice-chilled tubes containing aprotinin and/or ethylenediaminetetra-acetic acid, and centrifuged at 2000
Statistical analyses
Prism® 5 (GraphPad Software, La Jolla, CA, USA) software was used to analyse all data. Results were presented as mean ± SD.The paired t-test was used, all tests performed were two-sided and a P-value of <0.05 was considered to be statistically significant.
Results
Baseline characteristics of the study population
Of the 152 patients screened for inclusion, 28 were selected for study treatments and data analysis; patient distribution and exclusion criteria are shown in Figure 1. The mean age of the included patients was 61.10 ± 5.99 years and there were 16 (57%) men. Of the patients, diabetes mellitus was present in seven (25%), coronary heart disease in 12 (43%) and hypertension in seven (25%). Oral therapies prescribed for those with hypertension included standard regimens of calcium channel antagonists and statins.
Diastolic and systolic function
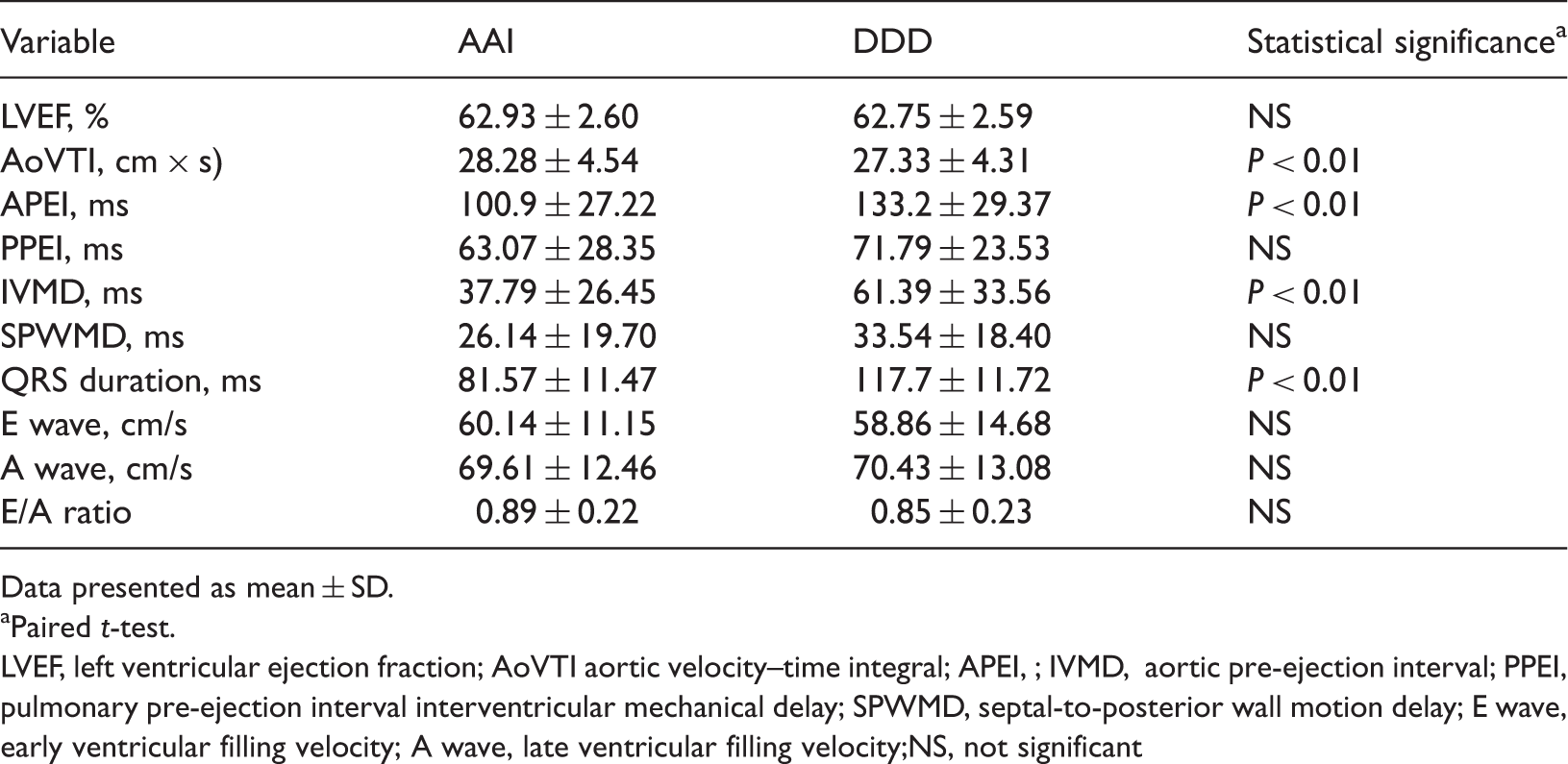
With respect to LV diastolic function, there were no differences in transmitral E and A velocities and E/A ratio between AAI and DDD modes (Table 1). The systolic parameter AoVTI was significantly greater in AAI than in DDD mode (P < 0.01) (Table 1; Figure 2, A), whereas LVEF did not differ between the two pacing modes (Table 1).
Comparison of haemodynamic parameters and QRS interval between single-lead atrial (AAI) and dual-chamber (DDD) pacing modes in patients with sick sinus syndrome. AoVTI, aortic flow velocity time–integral; APEI, aortic pre-ejection interval; IVMD, interventricular mechanical delay, ** P < 0.01, paired t-test. Comparison of electrocardiographic and echocardiographic findings between single-lead atrial (AAI) and dual-chamber (DDD) pacing mode in 28 patients with sick sinus syndrome. Data presented as mean ± SD. Paired t-test. LVEF, left ventricular ejection fraction; AoVTI aortic velocity–time integral; APEI, ; IVMD, aortic pre-ejection interval; PPEI, pulmonary pre-ejection interval interventricular mechanical delay; SPWMD, septal-to-posterior wall motion delay; E wave, early ventricular filling velocity; A wave, late ventricular filling velocity;NS, not significant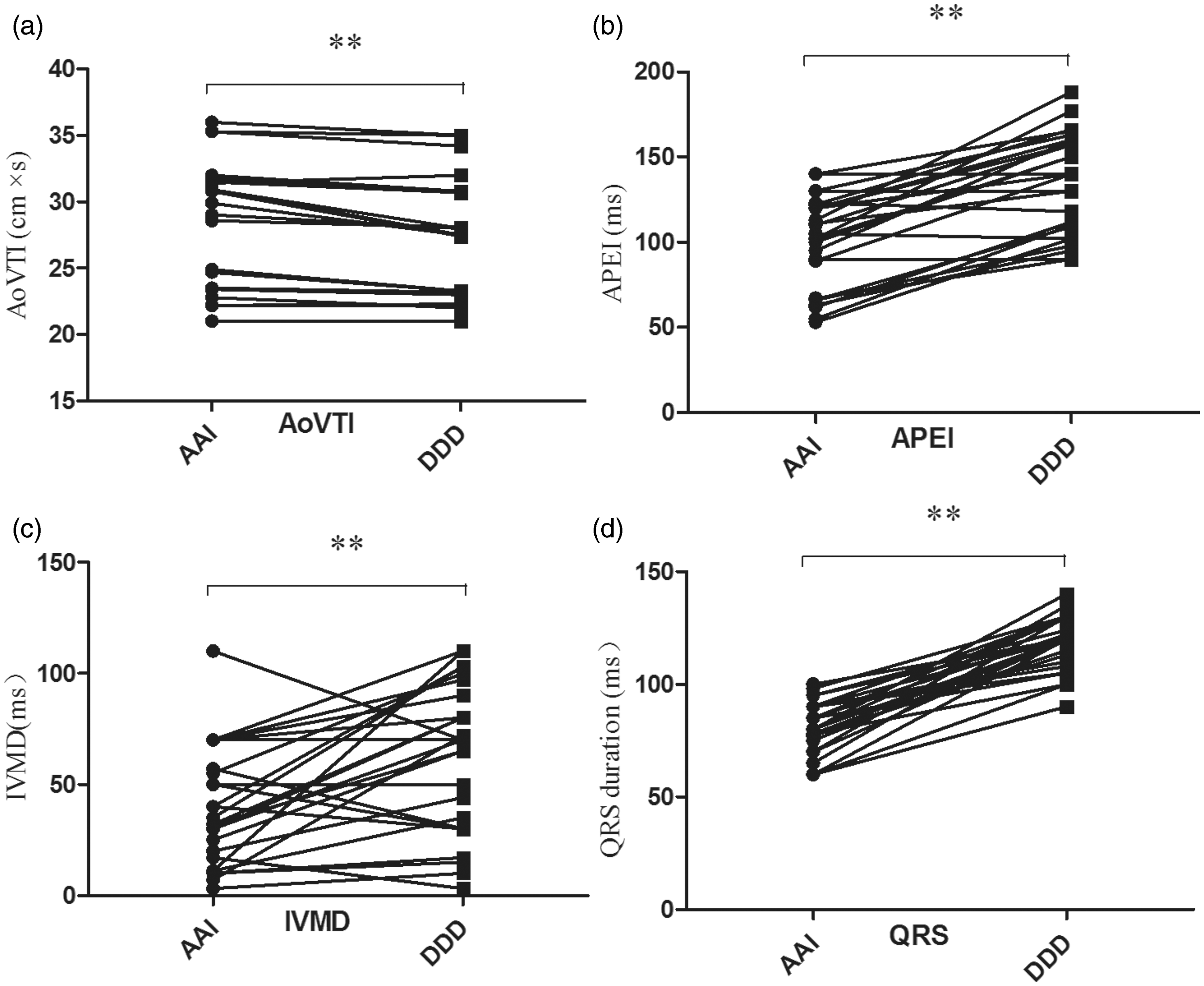
Dyssynchrony
Electrical and echocardiographic indicators of dyssynchrony are shown in Table 1 and Figure 2. QRS duration was significantly greater in DDD than in AAI mode (P < 0.01), 18 The echocardiographic parameters IVMD and APEI were significantly greater in DDD than in AAI mode (P < 0.01 for both parameters). SPWMD did not differ significantly between the pacing modes.
Neuroendocrine factors
Values for ANP, endothelin, angiotensin II and aldosterone were significantly higher in DDD mode than in AAI mode (P < 0.01 for all differences) (Table 2 and Figure 3).
Comparison of neuroendocrine factors between single-lead atrial (AAI) and dual-chamber (DDD) pacing modes in patients with sick sinus syndrome. ANP, atrial natriuretic protein; ET, endothelin; ALD, aldosterone; AngII, angiotensin II. ** P < 0.01, paired t-test. Comparison of neuroendocrine factors between single-lead atrial (AAI) and dual-chamber (DDD) pacing mode in 28 patients with sick sinus syndrome. Data presented as mean ± SD. Paired t-test.

Discussion
This is the first study that correlates both plasma levels of neuroendocrine factors and haemodynamic changes with AAI versus DDD pacing mode. The results suggest that AAI pacing may be a better choice than DDD pacing for patients with sick sinus syndrome. Conventional dual-chamber pacing is performed by implanting separate leads in the right atrial appendage and RVA. Although several large randomized clinical trials have suggested that RVA pacing may result in LV systolic dysfunction and heart failure,19–23 the RVA is still the most commonly used pacing position because it can be easily reached and allows chronically stable positioning and stimulation thresholds. DDD pacing with a very short atrioventricular interval and >99% ventricular apex pacing has been reported to increase the incidence of atrial fibrillation. 24 In this study, all patients had preserved atrioventricular conduction. In DDD pacing mode, a shortened atrioventricular delay was also programmed in order to mimic ventricular pacing. It is easy to observe electrical dyssynchrony by electrocadiographic examination, as the QRS duration is significantly increased in DDD mode, compared with in AAI mode; indeed we found this in our present study. To detect mechanical dyssynchrony in different pacing modes, we used echocardiography.
Animal studies have demonstrated obvious changes in LV electrical activation and mechanical dyssynchrony induced by RVA pacing.25–28 In clinical trials, LV dyssynchrony was associated with a poor haemodynamic outcome and major cardiac events.29,30 An observational study in patients with congenital heart block (paced for 10 years) demonstrated that RVA pacing was associated with increased LV dyssynchrony, adverse LV remodelling and decreased cardiac output, compared with matched controls. 21 Among patients with atrial fibrillation who had atrioventricular node ablation and RVA pacing, 49% developed LV dyssynchrony during a period of 3–4 years. 31 A significant reduction in LVEF was observed in patients who developed dyssynchrony, whereas no change in LVEF occurred in those who did not develop dyssynchrony. 31 Dyssynchrony is an important topic in the study of advanced heart failure, and today’s pacemaker engineers and designers seek to diminish both intra- and interventricular dyssynchrony.
In the present randomized study, we attempted to quantify LV dyssynchrony by means of echocardiography. SPWMD and IVMD are useful parameters in the context of cardiac resynchronization therapy. 32 IVMD (which if elevated indicates interventricular dyssynchrony) was found to be significantly higher in DDD than in AAI pacing, in the present study. SPWMD, which signifies a delay in posterior wall contraction in comparison with the septum, is a useful marker of intraventricular asynchrony; it also predicts LV reverse remodelling and is associated with long-term clinical improvement after cardiac resynchronization therapy.33,34 However, SPWMD did not differ between AAI and DDD pacing modes in the present study population. Aksoy et al. 18 commented that the presence of regional wall motion abnormalities was a major restriction on SPWMD. They found that measurements were unreliable, particularly in patients in whom the anterior septum was akinetic. As a consequence of this, the reliability of SPWMD for predicting the response to dyssynchrony could not be described as being particularly strong. 18
The deleterious effects of RVA pacing may be related to an abnormal electrical and mechanical activation pattern and neuroendocrine activation. During RVA pacing, the electrical wave front propagates slowly through the ventricular myocardium rather than through the His–Purkinje conduction system (which is similar to what occurs during left bundle branch block); this increases interventricular dyssynchrony. RVA pacing also alters the intraventricular activation sequence. In the present study, APEI and IVMD were higher in DDD than in AAI mode (Table 1), which indicates that mechanical intraventricular dyssynchrony was greater in DDD mode than in AAI mode.16,18 Unsurprisingly, AoVTI, a surrogate of stroke volume, was lower during DDD than during AAI pacing.
Elevated ANP has been reported in experimental pacing models of congestive heart failure.35–37 Small studies have shown that natriuretic peptides are elevated in patients during pacing.38–40 These changes occur even after short periods of pacing and seem to be more pronounced during VVI (i.e., ventricular inhibited) than DDD pacing. 41 This may be because VVI pacing leads to atrioventricular dissociation. In addition, the plasma level of ANP declines after cardiac resynchronization therapy and may therefore serve as a quantitative marker to evaluate the response to this therapy. 42 This was confirmed by our data.
The principal focus of our present study was the assessment of levels of ANP, endothelin, angiotensin II and aldosterone during AAI and DDD pacing, to determine whether these neuroendocrine factors are correlated with unnecessary right ventricular pacing in patients with sick sinus syndrome. The results showed that concentrations were significantly higher during DDD than during AAI mode, regardless of whether AAI or DD pacing was applied first. This indicates that right ventricular pacing may result in the adverse effect of neuroendocrine activation.
To conclude, In the DDD setting, the activation of ANP and the RAAS, as well as increased endothelin levels, together impairs cardiovascular homeostasis in patients with sick sinus syndrome. The DDD pacing mode induced not only neuroendocrine system activation but also left ventricular dysfunction and left ventricular dyssynchrony, in the short term. These findings discourage the routine use of DDD pacing in patients with sick sinus syndrome.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
