Abstract
Objective
To investigate the effect of the poly(ADP-ribose) polymerase-1 (PARP-1) inhibitor PJ34 on the proliferation and invasiveness of ovarian cancer C13* cells and the role of nuclear factor-κB (NF-κB).
Methods
Proliferation of C13* cells was measured using a 3 -(4,5-dimethylthazol-2-yl)-2,5-diphenyl tetrazolium bromide assay after incubation with PJ34 at different concentrations and for different treatment durations. In addition, expression of PARP-1 and the NF-κB p65 subunit after treatment with PJ34 was measured using Western blot and immunocytochemistry. The effect of PJ34 on cell invasiveness was examined using a transwell invasion assay.
Results
PJ34 inhibited proliferation of C13* cells in a time- and dose-dependent manner. PJ34 treatment was also associated with a dose-dependent decrease in PARP-1 and NF-κB p65 expression and attenuated invasiveness of C13* cells. PARP-1 expression was positively correlated with NF-κB p65 expression.
Conclusion
The PARP-1 inhibitor PJ34 can markedly inhibit the proliferation and invasiveness of C13* cells, possibly due to PARP-1-mediated attenuation of NF-κB activity.
Introduction
Ovarian cancer is a very common gynaecological malignancy and has one of the highest mortality rates of all cancers. 1 It can metastasize extensively at an early stage, but the underlying mechanisms are not clear. 2 Tumour invasion and metastasis are two important causes of treatment failure and death; inhibition of these processes is likely to be the key to improving the survival rate of patients with ovarian cancer. Poly(ADP-ribose) polymerase (PARP) is a monomeric protease widely present in the nuclei of most eukaryotic cells that is associated with the occurrence and development of a variety of diseases. 3 It is known to activate nuclear factor-κB (NF-κB) through a variety of pathways, which can lead to increased expression of NF-κB-dependent genes such as oncogenes, cell adhesion molecules, matrix metalloproteinases and growth factors. 4 PARP gene knockout or the application of PARP inhibitors can inhibit NF-κB activity, thus lowering NF-κB-dependent gene expression.5–8
Current reports on PARP have mostly concentrated on colon cancer, 9 breast cancer, 10 liver cancer 11 and malignant lymphoma; 12 few studies have investigated the inhibition of PARP expression and its relationship with biological activity in ovarian cancer. In the present study, the effects of the PARP-1 inhibitor PJ34 on the expression of PARP-1 and the NF-κB p65 subunit and on cell proliferation and invasion were examined in ovarian cancer C13* cells, and the possible underlying mechanisms were explored.
Materials and methods
Cell culture
The ovarian cancer epithelial cell line C13* was provided by Meirong Du, Institute of Obstetrics and Gynaecology, Fudan University, Shanghai, China. The cells were cultured in complete medium consisting of RPMI 1640 medium plus 10% HyClone fetal bovine serum (FBS) (Thermo Fisher Scientific, Rockford, IL, USA), 100 U/ml penicillin and 100 µg/ml streptomycin at 37℃ in an incubator supplemented with 5% CO2. Cells were passaged after digestion with 0.25% HyClone trypsin (Thermo Fisher Scientific) and were used in the following experiments once they had reached the logarithmic growth phase.
MTT assay
The effect of PJ34 on in vitro proliferation of C13* cells was measured using an assay based on the tetrazolium salt 3 -(4,5-dimethylthazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT). Briefly, 100 µl of a suspension of C13* cells at a concentration of 5 × 104 cells/ml of complete medium were seeded into each well of a 96-well culture plate. After incubation at 37℃ overnight, the medium was replaced with complete medium containing 0 (control), 2.5, 5.0 or 10.0 µmol/l of the PARP inhibitor PJ34 (Merck, Darmstadt, Germany). No cells were added to one well per plate as a blank. After 24, 48 or 72 h of treatment, cells in each well were incubated with 20 µl MTT (Merck) for an additional 4 h. After removal of the supernatant fraction, the cells were incubated with 150 µl dimethyl sulphoxide whilst being shaken for 10 min. The absorption at 490 nm for each well was then measured using an automatic plate reader. Each experiment was performed five times. The inhibition rate was calculated as: inhibition rate (%) = (1 – mean absorption for experimental cells/mean absorption for control cells) × 100%
Western blot assay
The effects of PJ34 on the expression of PARP-1 and NF-κB by C13* cells were measured using a Western blot assay. C13* cells were treated with 0 (controls), 2.5, 5.0 or 10.0 µmol/l PJ34 for 24 h and lysed in radioimmunoprecipitation lysis buffer with phenylmethylsulphonyl fluoride. Total protein was extracted using a protein extraction kit (Shenneng Bo cai Biotechnology, Shanghai, China). The total protein concentration in the resulting lysate was then measured using the bicinchoninic acid method, with the optical density values being measured at 562 nm. Next, 60 µg of total protein from each sample was subjected to sodium dodecyl sulphate–polyacrylamide gel electrophoresis and transferred onto polyvinylidene difluoride membranes. The membranes were then blocked with 5% skimmed milk for 1 h and incubated with 1 000-fold diluted rabbit anti-human PARP-1 and NF-κB p65 antibodies and mouse anti-human β-actin antibody (Spring Bioscience, Pleasanton, CA, USA) at 4℃ overnight. After being washed three times for 10 min with Tris-buffered saline with Tween (TBST) (pH 7.5), the membranes were incubated with the appropriate horseradish peroxidase-labelled goat anti-rabbit and goat anti-mouse immunoglobulin G secondary antibodies (1 : 10 000) (ZSGB-Bio, Beijing, China) at room temperature for 1 h. After being washed again three times for 10 min with TBST, the proteins were visualized by incubation with an enhanced chemiluminescence chromogenic reagent kit (Millipore, MA, USA) and imaged using the LAS-3000 imaging system (Fujifilm Medical Systems, Stamford, CT, USA). The band intensity was measured using the AlphaImager 2200 image analyser software (ProteinSimple, Santa Clara, CA, USA) and the expression of PARP-1 and NF-κB p65 was normalized to that of β-actin.
Immunocytochemistry
The effects of PJ34 on the expression of PARP-1 and NF-κB by C13* cells were also measured using immunocytochemistry. C13* cells were grown on glass coverslips in complete medium and treated with 0 (control) or 5 µmol/l PJ34 for 24 h. After washing with phosphate buffered saline (PBS) (pH 7.4), the cells were fixed with 4% paraformaldehyde and permeabilized with 0.2% Triton-X100 for 30 min in a humidified tissue culture incubator at 37℃. The cells were incubated with rabbit anti-human antibodies against PARP-1 or NF-κB p65 (1 : 200) at 4℃ overnight. One glass coverslip was incubated with PBS instead of primary antibody as a negative control. The cells were then incubated with the appropriate goat anti-rabbit immunoglobulin G secondary antibodies for 30 min at 37℃. Between each step, the glass coverslips were washed with PBS. The cells were stained with haematoxylin and viewed using light microscopy. Positive staining for PARP-1 appeared as tan or brown granules mainly in the nucleus or cytoplasm. Positive staining for NF-κB appeared as brownish-yellow granules located mainly in the cytoplasm but also in the nucleus. Positive expression of PARP-1 and NF-κB was quantified using ImageJ software (National Institutes of Health, Bethesda, MA, USA).
Transwell invasion assay
The effect of PJ34 on the invasiveness of C13* cells was investigated using a transwell migration assay. The upper chambers of transwell migration chambers (Corning, Shanghai, China) were coated with BD Matrigel matrix (BD Biosciences, San Jose, CA, USA) at 4℃ and air dried. Confluent C13* cells were treated with 0 (controls) or 5 µmol/l PJ34 for 24 h. After aspiration of the cell medium, the cells were incubated with 0.1% bovine serum albumin (BSA)-free medium at 37℃ for 30 min, digested, and resuspended in 0.1% BSA-free medium to form a 2 × 105 cells/ml suspension. Then 200 µl of cell suspension was added to each Matrigel-coated upper chamber and 600 µl of complete medium containing 20% FBS was added to the lower chamber. After being cultured for 24 h, cells in the Matrigel were removed using a cotton swab and cells that had migrated onto the other side of the membrane were fixed with 4% paraformaldehyde and observed under light microscopy. The number of cells in eight randomly selected high-power fields (× 400 magnification) were counted for each specimen.
Statistical analyses
Data were expressed as the mean ± SD of at least three experiments. Differences between two independent samples were analysed using a two-sample t-test for independent samples and differences between groups were analysed using single factor variance analysis. A P-value of < 0.05 was considered to be statistically significant. All statistical analyses were performed using SPSS software version 18.0 (SPSS Inc., Chicago, IL, USA).
Results
Effects on proliferation of C13* cells
The dose- and time-dependent effects of PJ34 on the proliferation of C13* cells are shown in Figure 1. PJ34 significantly inhibited C13* cell proliferation at all concentrations and durations of treatment (P < 0.05 for all).
Dose- and time-dependent effects of PJ34 on the proliferation of C13* cells relative to controls. Data presented as mean ± SD.
Effects on expression of PARP-1 and NF-κB p65
The effects of PJ34 on the expression of PARP-1 and NF-κB relative to the expression of β-actin in C13* cells as measured by Western blot are shown in Figure 2. The expression of PARP-1 and NF-κB in C13* cells treated with 2.5, 5.0 and 10.0 µmol/l PJ34 for 24 h was significantly lower than in control cells (P < 0.05 for all). In addition, expression of PARP-1 was positively correlated with expression of NF-κB p65 (P < 0.05).
Western blot and expression of poly(ADP-ribose) polymerase-1 (PARP-1) and nuclear factor-κB (NF-κB) p65 protein relative to β-actin in C13* cells treated with PJ34 and controls. Data presented as mean ± SD.
Expression of PARP-1 and NF-κB p65 was also seen to be affected by PJ34 treatment using immunocytochemistry (Figures 3 and 4). When quantified, PARP-1 expression was 58.03 ± 1.43 in control cells and 28.41 ± 1.38 in cells treated with 5 µmol/l PJ34 (P < 0.05). Similarly, NF-κB p65 expression was 46.39 ± 3.96 in control cells and 24.11 ± 0.29 in cells treated with 5 µmol/l PJ34 (P < 0.05).
Immunocytochemistry of poly(ADP-ribose) polymerase-1 expression in C13* cells treated with 0 (control) or 5 µmol/l PJ34. Immunocytochemistry of nuclear factor-κB p65 expression in C13* cells treated with 0 (control) or 5 µmol/l PJ34.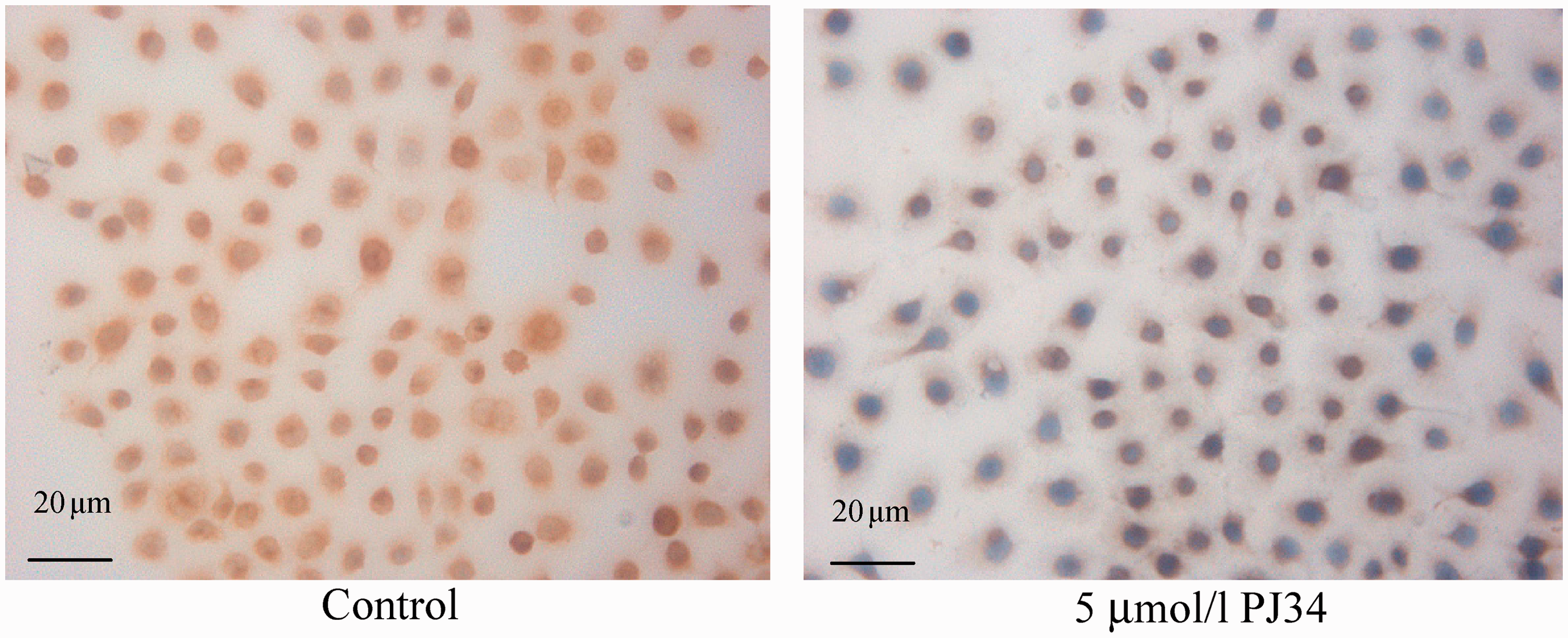

Effects on invasive ability
Using the transwell migration assay, the number of C13* cells that invaded the lower chamber was 91.20 ± 9.26 for control cells, but was only 44.20 ± 4.49 for cells treated with 5 µmol/l PJ34 (P < 0.05) (Figure 5).
Migrating C13* cells after treatment with 0 (control) or 5 µmol/l PJ34.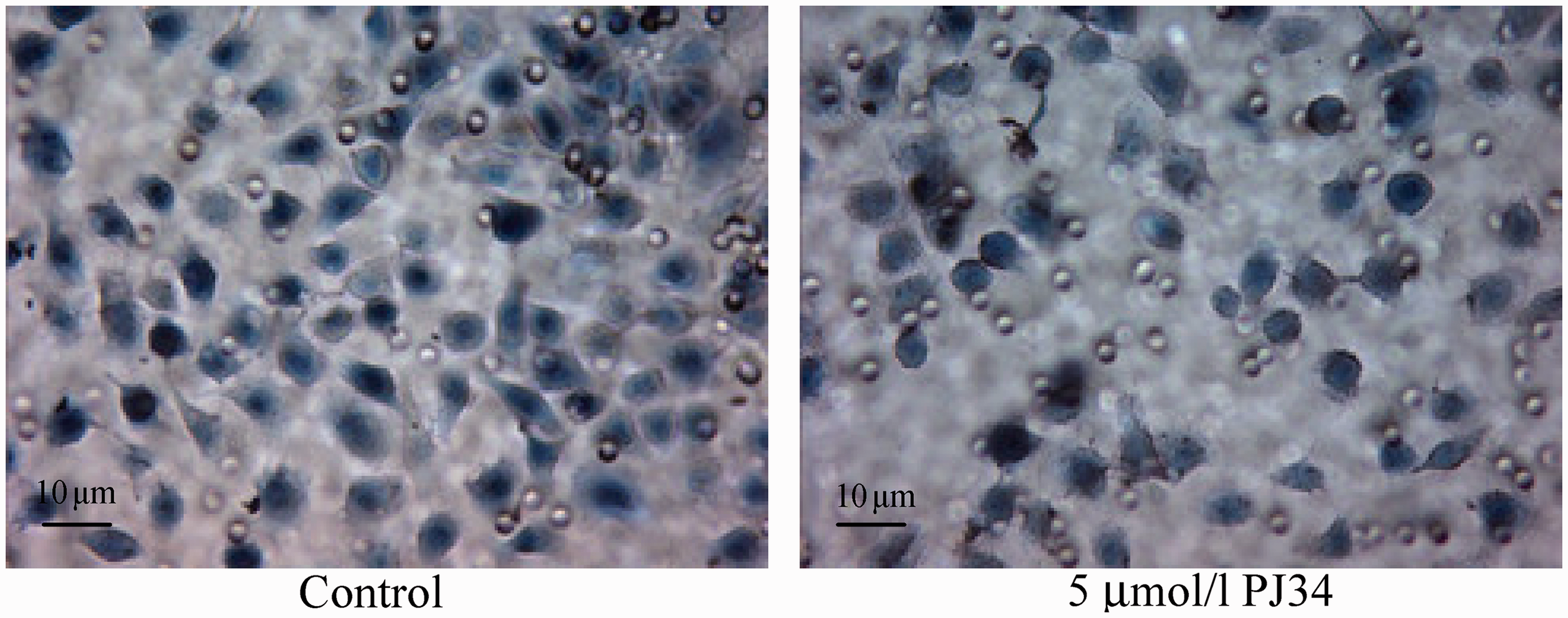
Discussion
Progress in molecular biology, especially in the areas of cell proliferation, differentiation and apoptosis regulation mechanisms, has led to the development of molecular targeted therapy. The roles of PARP and NF-κB have been a particular focus for study, but little has been published on their expression and interaction in ovarian cancer.
The protease PARP is present in the nuclei of most eukaryotic cells. It is involved in poly-ADP glycosylation and controls post-translational modification of many proteins and enzymes in the nucleus, thereby regulating a number of intracellular molecular events, including DNA repair, gene transcription, cell cycle progression and cell death. Since its discovery, PARP has attracted the attention of many researchers. Among the members of the PARP family, PARP-1 has the most typical structure and has been a particular focus for study.
We have previously shown that the PARP-1 inhibitor PJ34 can significantly lower PARP-1 expression, inhibit cell proliferation, promote apoptosis and enhance the sensitivity to cisplatin in C13* cells. 13 However, the effect of PJ34 on the invasiveness of ovarian cancer cells has not been studied. Martin-Oliva et al. 14 reported that treatment with PARP-1 inhibitors significantly inhibited the growth of skin cancer. Huang et al. 15 reported that PJ34 inhibited the proliferation of liver cancer HepG2 cells and the growth of subcutaneously transplanted HepG2 cells a nude mouse model. In addition, it has been shown that that PARP inhibitors can inhibit the formation of blood vessels. Rajesh et al. 16 reported that PARP inhibitors PJ34 and 3-aminobenzamide inhibited migration of vascular endothelial growth factor (VEGF)-induced human umbilical vein endothelial cells (HUVECs), and Pyriochou et al. 17 demonstrated that PJ34 could inhibit HUVEC migration induced by VEGF, the nitric oxide donor DETA-NO or BAY41-2272. Therefore, it is possible that PARP-1 inhibitors may have inhibitory effects on ovarian cancer cell proliferation and invasion.
The exact mechanisms of invasion and metastasis in ovarian cancer are not yet fully elucidated. NF-κB, in addition to being involved in a variety of physiological and pathological processes such as cell proliferation and apoptosis, also seems to have an important role in tumour formation, transfer and invasion. 18 NF-κB is a ubiquitous inducible nuclear transcription factor. In the resting state, NF-κB presents as a dimer of p50 and p65 and exists in cytoplasm in an inactive state by combining with its inhibitory protein IκB. It has been shown that NF-κB has an important role in the occurrence and development of colorectal cancer by promoting the growth of tumour cells through regulating the expression of certain oncogenes (such as c-myc), by promoting colorectal cancer proliferation through upregulating cyclooxygenase-2 expression, and by promoting colorectal metastasis through upregulating the expression of proteins such as intercellular adhesion molecule-1 (ICAM-1).19–21 Ko et al. 22 have shown that blocking NF-κB transcriptional activity reduced invasion and metastasis and the matrix adhesion capacity of breast cancer cells. Therefore, inhibition of the NF-κB signalling pathway has a potential role in the treatment of cancer.
Interactions between PARP and NF-κB have been studied. Genovese et al. 23 showed that PARP inhibitors can inhibit the degradation of IκB and reduce NF-κB activity. In addition, PARP and NF-κB have synergistic coactivation effects. Hassa et al. 24 showed that PARP-1 could directly interact with NF-κB to synergistically activate transcriptional factors independent of enzymatic activity and DNA-binding ability. Stilmann et al. 25 also reported that PARP-1 is involved in the process of NF-κB activation. It is possible that PARP forms a complex with NF-κB to increase NF-κB activity, which in turn may upregulate the expression of NF-κB-dependent genes. Furthermore, regulation of ICAM-1 and P-selectin expression by PARP in the inflammatory process has been shown to be mediated by the activation of NF-κB.26,27
In the present study, the effects of the PARP-1-specific inhibitor PJ34 on cell growth and invasiveness and their relation to NF-κB activity were investigated in ovarian cancer C13* cells. It was shown that PJ34 is able to significantly inhibit ovarian cancer C13* cell proliferation in a dose- and time-dependent manner. Immunocytochemical analysis demonstrated that expression of PARP-1 and NF-κB in C13* cells was significantly decreased after treatment with PJ34. This effect was confirmed on Western blot analysis and was shown to be dose-dependent. On in vitro invasion assay, the number of PJ34-treated C13* cells invading the transwell chamber membrane was significantly reduced compared with control cells, suggesting that the capacity for cell invasion and metastasis is inhibited by PJ34. Thus, PARP-1 seems to be involved in the proliferation, invasion and metastasis of C13* cells, and these effects may be mediated by changes in NF-κB activity.
In summary, in the present study PJ34 significantly reduced PARP-1 expression and inhibited growth, invasion and metastasis in ovarian cancer C13* cells. These inhibition effects appear to be related to PARP-1 inhibition resulting in a decrease in NF-κB activity. These data suggest that PARP-1 may play a central role in ovarian cancer cell proliferation, invasion and metastasis, and could have an important role in the clinical treatment and postoperative prognosis of ovarian cancer. Further in-depth studies are required to elucidate the detailed molecular mechanisms underlying the inhibitory effects of PARP-1 inhibitors on ovarian cancer cell growth and invasion and to confirm the inhibitory effects of PARP-1 inhibitors on the proliferation and invasion of ovarian cancer in animal models.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This study was sponsored by the Shandong Provincial Natural Science Foundation (ZR2009CM104) and Shandong Province Technology Development Projects (2012GSF11821).
