Abstract
Objectives
To compare the efficacy, safety and other clinical benefits of the levonorgestrel-releasing intrauterine system (LNG-IUS) and gonadotropin-releasing hormone analogues (GnRH-a) in women with endometriosis.
Methods
A systematic search was carried out using the Cochrane Central Register of Controlled Trials, PubMed, MEDLINE™ and EMBASE databases for all randomized controlled trials (RCTs) that evaluated the use of the LNG-IUS and GnRH-a in premenopausal women with endometriosis.
Results
Five RCTs studies were identified. A meta-analysis showed that, in women with endometriosis, both the LNG-IUS and GnRH-a reduced pain visual analogue scale scores (weighted mean difference [WMD] 0.03 [95% confidence interval [CI] −0.53, 0.59]), serum levels of CA125 (WMD −12.29 [95% CI −29.90, 3.32]), and American Society of Reproductive Medicine staging scores (WMD 1.10 [95% CI −27.98, 30.18]). Psychological and general wellbeing index scores were increased (WMD 1.50 [95% CI −6.19, 9.19]). Levels of low-density lipoprotein cholesterol were also significantly reduced in patients treated with the LNG-IUS (WMD 39.30 [95% CI 6.74, 71.86]).
Conclusions
The LNG-IUS had clinical efficacy equivalent to that of GnRH-a but may have some clinical advantages over GnRH-a in the treatment of endometriosis-associated symptoms. These observations will require further verification in additional studies employing larger patient populations.
Keywords
Introduction
Endometriosis, which affects 10–20% of women of reproductive age, is a gynaecological disease characterized by the presence and growth of endometrial tissue outside the uterus. 1 Common symptoms include dysmenorrhoea, dyspareunia, menorrhagia, noncyclical pelvic pain and infertility. 1 The precise pathogenesis of endometriosis is unclear but it is believed to be oestrogen dependent; current conservative medical treatment is hormonally based and predominantly palliative.2,3
Gonadotropin-releasing hormone analogues (GnRH-a) including buserelin, goserelin, histrelin, leuprolide and nafarelin are currently the gold-standard medical treatments for endometriosis. 4 These agents selectively bind to specific receptors on the gonadotropic cells of the anterior pituitary with higher affinity than the native hormone. 5 Activation of these receptors by GnRH-a mediates an immediate increase in secretion of follicle-stimulating hormone and luteinizing hormone, followed by inhibition of gonadotropins and gonadal steroids. 6 GnRH-a are widely used to reduce endometriosis-related pain but are associated with a reduction in bone-mineral density, which limits their use to a period of ≤6 months. 7 Other side-effects of these agents include hot flushes, insomnia, vaginal dryness and emotional lability. 8 In addition, the high cost of GnRH-a further limits their use, especially in developing countries.
The levonorgestrel-releasing intrauterine system (LNG-IUS), first introduced in Finland in 1990, provides an alternative mechanism for the local release of LNG directly into the uterine cavity at a steady rate of 20 µg per day over 5 years. 9 In the presence of high concentrations of LNG, the endometrium becomes atrophic and pseudodecidualization is observed; ovulation is unaffected. 10 Systemic levels of LNG are, however, lower than those associated with oral progestogens,11,12 meaning that the side-effect profile of the LNG-IUS is less severe. Aside from its use as a contraceptive, the LNG-IUS has been reported to improve the pain symptoms associated with endometriosis and to reduce menstrual blood loss. 13
A systematic review of randomized controlled trials (RCTs) comparing the LNG-IUS with GnRH-a has not previously been conducted. The aim of the present study was to perform a meta-analysis to examine the efficacy, safety and other benefits of the LNG-IUS compared with GnRH-a in women with endometriosis, following conservative surgery.
Materials and methods
Search strategy and study selection
A systematic search was carried out in the Cochrane Central Register of Controlled Trials, PubMed, MEDLINE™ (between January 1966 and April 2012) and EMBASE (between January 1985 and April 2012) databases for all RCTs using the following keywords: ‘levonorgestrel-releasing intrauterine system’, ‘LNG-IUS’, ‘endometriosis’, ‘GnRH-a’ and ‘gonadotropin-releasing hormone analogues’. The list of articles retrieved by the search was reviewed by one of the authors (S.L.). Reference lists of selected publications were subjected to a further manual search, to identify further studies of relevance. RCTs were included if they: (i) had a parallel design investigating the safety and clinical efficacy (including effects on pain scores, CA125 levels, American Society of Reproductive Medicine [ASRM] staging scores, HRQoL, cardiovascular risk markers and side-effects) of LNG-IUS versus GnRH-a; (ii) enrolled premenopausal women with endometriosis following conservative surgery who had no plans to become pregnant; (iii) were not quasi-randomized trials. Only original publications were included; conference abstracts and dissertations were not considered. Language restrictions were not applied.
Assessment of study quality
According to existing criteria from the Quality Assessment of Diagnostic Accuracy Study (QUADAS), 14 ten of the 14 items were applicable and were used to assess the quality of studies included in the meta-analysis. Any study that failed to reach these standards was considered to be of low quality. The study quality items included were: (i) verification of diagnosis by a reference standard in all tested cases; (ii) adequate description of selection criteria; (iii) adequate description of test; (iv) appropriate patient spectrum; (v) blinding of test; (vi) prospective study design; (vii) consecutive recruitment; (viii) adequate follow-up period; (ix) available clinical data; (x) explained withdrawal.
Statistical analyses
All statistical analyses were performed using RevMan software (version 5.0.23.0; the Nordic Cochrane Centre, Rigshospitalet, Copenhagan, Denmark), according to statistical guidelines of the Cochrane Menstrual Disorders and Subfertility Review Group. 15 A fixed-effects model (Z-test) was used to calculate the observed efficacies (continuous outcome). The safety profiles of LNG-IUS and GnRH-a were compared by calculating crude odds ratios (ORs) with 95% confidence intervals (CIs). It was anticipated that only a small number of RCTs would be identified; as such, sensitivity analyses, funnel plots and Egger's test (which are used to estimate any potential publication bias) could not be used. Statistical heterogeneity among the studies was calculated using the Q-test (χ2-test) and I2 statistics; P < 0.05 indicated a lack of homogeneity among the studies.
Results
Key characteristics and study findings from five randomized controlled trials, selected for a meta-analysis comparing the levonorgestrel intrauterine system (LNG-IUS) with gonadotropin-releasing hormone analogues (GnRH-a), in women with endometriosis following conservative surgery.

VAS, visual analogue scale; HRQoL, health-related quality of life; PGWBI, Psychological General Well-Being Index Questionnaire; CPP, chronic pelvic pain; TESP, total endometrial severity profile; ASRM, American Society for Reproductive Medicine.
Pooled analysis of three trials (including 126 women with endometriosis16–18) demonstrated that both the LNG-IUS and GnRH-a reduced pain scores as recorded using a visual analogue scale (VAS) and there was no statistically significant difference between treatment groups (weighted mean difference [WMD] 0.03 [95% CI −0.53, 0.59]; Figure 1). No heterogeneity was determined among these studies (χ2 = 2.74, I2 = 27%). Since there were never more than two trials within the other meta-analyses described below, no further heterogeneity analysis was performed.
Results of a meta-analysis of the effects of the levonorgestrel-releasing intrauterine system (LNG-IUS) and gonadotropin-releasing hormone analogue (GnRH-a) on pain scores (measured by a visual analogue scale: 0, no pain; 10, worst pain), in women with endometriosis as determined from three randomized controlled trials. WMD, weighted mean difference; IV, inverse variance; CI, confidence interval; df, degrees of freedom.
In a trial of 44 participants,
19
no significant difference was determined between the LNG-IUS and GnRH-a in reducing serum CA125 levels; the WMD was calculated to be −12.29 (95% CI −27.98, 3.32; Figure 2). Both treatments also decreased ASRM staging scores to a similar extent, as demonstrated by Gomes et al.
17
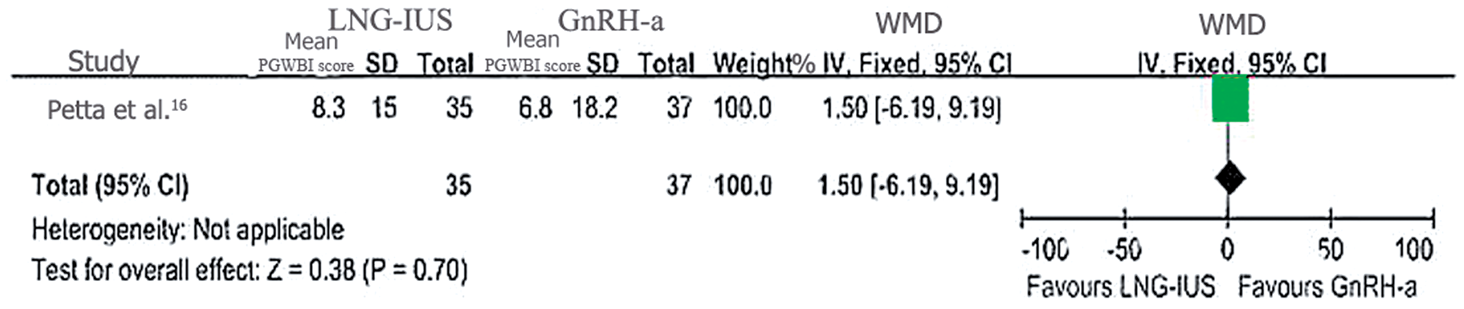
(WMD 1.10 [95% CI −27.98, 30.18]; Figure 3) and improved HRQoL, as measured by the Psychological and General Well-Being Index Questionnaire in the study by Petta et al.
16
(WMD 1.50 [95% CI −6.19, 9.19]; Figure 4).
Effects of the levonorgestrel-releasing intrauterine system (LNG-IUS) and gonadotropin-releasing hormone analogues (GnRH-a) on serum CA125 levels in women with endometriosis, as determined from a randomized controlled trial. WMD, weighted mean difference; IV, inverse variance; CI, confidence interval. Effects of the levonorgestrel-releasing intrauterine system (LNG-IUS) and gonadotropin-releasing hormone analogues (GnRH-a) on American Society for Reproductive Medicine (ASRM) staging scores (stage I, minimal [score 1–5]; stage II, mild [6–15]; stage III, moderate [16–40]; stage IV, severe [>40])
36
in women with endometriosis, as determined from a randomized controlled trial. WMD, weighted mean difference; IV, inverse variance; CI, confidence interval. Effects of the levonorgestrel-releasing intrauterine system (LNG-IUS) and gonadotropin-releasing hormone analogues (GnRH-a) on health-related quality of life (HRQoL), as measured by Psychological and General Well-Being Index Questionnaire (PGWBI) scores (where the higher the score, the better the HRQoL) in women with endometriosis enrolled in a randomized controlled trial. WMD, weighted mean difference; IV, inverse variance; CI, confidence interval.

One of the studies included in this analysis investigated the effects of the LNG-IUS and GnRH-a on levels of cardiovascular risk markers.
18
Serum levels of low-density lipoprotein cholesterol (LDL-C) and total cholesterol (TC) were significantly reduced in patients treated with the LNG-IUS, compared with patients treated with GnRH-a; the WMD for each marker was calculated as 39.30 ([95% CI 6.74, 71.86]; P = 0.02) and 56.40 ([95% CI 13.35, 99.45]; P = 0.01), respectively (Figure 5). Changes in the levels of triglycerides (TG) and high-density lipoprotein cholesterol (HDL-C) failed to reach statistical significance. The frequency of common endometriosis symptoms in patients treated with the LNG-IUS or GnRH-a are shown in Figure 6. Irregular bleeding, simple ovarian cysts and one-sided lower abdominal pain occurred more commonly in the LNG-IUS group (P ≤ 0.03) while vasomotor symptoms and amenorrhea were observed more frequently in the GnRH-a group (P ≤ 0.05).
Effects of the levonorgestrel-releasing intrauterine system (LNG-IUS) and gonadotropin-releasing hormone analogue (GnRH-a) on the cardiovascular risk markers total cholesterol (TC), triglycerides (TG), low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C) in women with endometriosis enrolled in a randomized controlled trial. WMD, weighted mean difference; IV, inverse variance; CI, confidence interval. Common side-effects associated with the use of the levonorgestrel-releasing intrauterine system (LNG-IUS) and gonadotropin-releasing hormone analogue (GnRH-a) in women with endometriosis enrolled in a randomized controlled trial. M–H, Mantel–Haenszel.

Discussion
The clinical consequences associated with endometriosis have considerable impact on patient HRQoL. It is reported that 70–90% of women with chronic pelvic pain have endometriosis, with dysmenorrhea being the most common symptom. 1 Although laparoscopic surgery is considered the first-choice and gold-standard treatment for endometriosis-associated symptoms, the rate of reoperation for pain after conservative surgery may be as high as 40%. 21 In addition, data from a RCT involving 236 patients showed that there was no significant difference between surgery and the LNG-IUS in the improvement of patient HRQoL, after 5 years’ follow-up. 22 Routine use of laparoscopic surgery is not advocated in patients with mild endometriosis, meaning that effective medical therapy is important in these individuals. The main aim of pharmacological therapy in endometriosis is to suppress ovarian activity and, as such, GnRH-a have been considered the standard postoperative medical treatments since the 1990s.6,23 These drugs are, however, expensive and their use should be limited to ≤6 months, due to adverse events such as bone loss, vasomotor symptoms and other symptoms associated with hypooestrogenism.4,16
The LNG-IUS is used worldwide as an effective form of contraception, but its additional benefits in other gynaecological conditions have also been investigated since as far back as 1982. 24 The LNG-IUS has reportedly been associated with reductions in menstrual blood loss, increases in haemoglobin levels and relief of menstrual pain.25–28 More recently, data from RCTs have indicated that the LNG-IUS is effective in relieving endometriosis-related pain.3,16,29 The mechanism of action by which the LNG-IUS relieves pain in endometriosis is still unclear, but it most likely involves both local and systemic effects. After insertion, the LNG-IUS releases high levels of LNG directly to the circumambient endometrium, depleting oestrogen and progesterone receptors, thereby reducing endometrial-cell proliferation. This is subsequently followed by decidualization of the stroma and atrophy of endometrial glands.30–32 Other research has demonstrated that use of the LNG-IUS is associated with an increase in the concentration of LNG in the peritoneal fluid, suggesting that it has a local activity on endometrial lesions. 33 The LNG-IUS has also been shown to reduce pelvic vascular congestion, which may contribute to relief of endometriosis-associated pain. 10 In addition, use of the LNG-IUS in endometriosis has been demonstrated to reduce expression of the progesterone receptor oestrogen receptor-α and the apoptosis marker Fas, which may contribute to its antiproliferative effect. 34 Although no significant difference was found between the LNG-IUS and GnRH-a in the present meta-analysis with respect to pain relief, this is likely to be due to the low number of RCTs included and the consequent small size of the sample. Additional RCTs, enrolling larger patient populations, are required in order to confirm the effect of LNG-IUS on endometriosis-associated pain.
It has been suggested that CA125 is a serum marker of endometriosis. The serum level of CA125 is higher in the majority of patients with endometriosis, compared with women without the condition, and may be related to the proliferative activity of endometriotic epithelial cells. 35 In the present analysis, only one study was identified that compared the effects of LNG-IUS and GnRH-a on CA125 levels. According to the results of this study, the level of CA125 decreased significantly at 6 months after insertion of the LNG-IUS, suggesting that the LNG-IUS may have an effect on endometriosis lesions. 19 There was, however, no significant difference in the reduction of CA125 between the LNG-IUS and GnRH-a, suggesting that perhaps both treatments have similar effects in moderating endometrial proliferation and controlling endometriosis. The precise mechanism by which the LNG-IUS reduces serum CA125 needs further research.In a prospective, noncomparative, observational study involving 334 women with endometriosis, the LNG-IUS was found to improve disease staging in 30.8% of cases after 6 months’ treatment, as measured by the ASRM classification system (stage I, minimal [score 1–5]; stage II, mild [6–15]; stage III, moderate [16–40]; stage IV, severe [>40]), 36 although the mechanism by which it achieves this is still unclear. 3 The present study further indicated the positive role of the LNG-IUS on staging of endometriosis: one RCT was identified in which staging scores decreased following treatment with both the LNG-IUS and GnRH-a. 17 No between-group differences in staging were observed, but since this observation is from a single study only, further investigation of the potential differences between the two treatments is required.
The studies identified as part of the present analysis also showed that women with endometriosis treated with either the LNG-IUS or GnRH-a achieved equivalent improvement in HRQoL scores. 16 In addition, the benefits of the LNG-IUS on lipid profiles were demonstrated. 18 A statistically significant reduction in LDL-C and TC, but not in TG or HDL-C levels, was observed in patients with endometriosis treated with the LNG-IUS compared with GnRH-a. Lipid profiles are important in defining the risk of arteriosclerosis and coronary disease, and LDL-C is possibly the most important parameter in the normalization of dyslipidaemia. 37 This suggests that the effects of the LNG-IUS on LDL-C may be of clinical importance in patients with endometriosis and dyslipidaemia. In addition to these benefits on lipids, several other clinical benefits unrelated to contraception have been reported for the LNG-IUS, including effects on menorrhagia, leiomyomata, adenomyosis and endometrial hyperplasia. 38–41 The present study was, however, unable to identify any RCTs investigating these effects.
The LNG-IUS is generally well tolerated when used as a contraceptive device in women, and any adverse effects are often transient and do not have any detrimental effects on patient satisfaction. 42 In the present study, the most common side-effects identified with LNG-IUS use in endometriosis included irregular menstrual bleeding, abdominal pain and simple ovarian cysts. These adverse events may develop due to impaired tolerance and pelvic discomfort. 43 Despite such adverse events, it has been previously reported that 75% of women using the LNG-IUS are satisfied or very satisfied with the treatment after 12 months’ follow-up. 44 Taken together, these data suggest that insertion of the LNG-IUS may be a convenient and efficacious treatment for endometriosis in the long-term.
In conclusion, the present meta-analysis demonstrated that the LNG-IUS and GnRH-a have equal clinical efficacy in the treatment of endometriosis. Long-term use of GnRH-a may be precluded by a high rate of systemic side-effects but the present study showed that the LNG-IUS was associated with fewer hypo-oestrogenic adverse events. Furthermore, insertion of a single LNG-IUS lasts for 5 years, which renders the treatment more convenient and cost-effective than GnRH-a for patients with endometriosis. The present meta-analysis (and published RCTs reviewed herein) indicate that the LNG-IUS may have clinical advantages over GnRH-a in the treatment of endometriosis-associated symptoms, but this will require further verification in additional studies employing larger patient populations.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
