Abstract
Objective
To investigate the migratory, metabolic and proliferative ability of rabbit fibrochondrocytes in meniscal fragments of different sizes under an in vivo subcutaneous environment.
Methods
Adult, male, New Zealand white rabbit menisci were harvested and the inner two-thirds of each were minced into different sizes: 0.5 × 0.5 × 0.5 mm (group A); 1.0 × 1.0 × 1.0 mm (group B); 2.0 × 2.0 × 2.0 mm (group C). Meniscal fragments were packed into a fascia sheath, then implanted subcutaneously. At weeks 1, 2 and 3 postimplantation, meniscal fragments were harvested; cell migration and viability were then evaluated by confocal laser scanning microscopy and haematoxylin and eosin staining. Aggrecan was detected using Safranin O and Fast Green staining, and protein levels of types I and II collagen, and proliferating cell nuclear antigen (PCNA), were evaluated by immunohistochemistry.
Results
Protein levels of types I and II collagen and PCNA, and fibrochondrocyte migration, were highest in group A compared with groups B and C, but aggrecan staining remained the same across the groups.
Conclusion
There was an inverse relationship between the migratory, metabolic and proliferative abilities of rabbit fibrochondrocytes and meniscal fragment size.
Introduction
Meniscal injuries are a common and important source of knee dysfunction and disability.1,2 Only the outer one-third of the mature meniscus has a blood-vessel supply that is able to transport the factors necessary for self repair. 3 As a consequence, tears in the inner two-thirds of the mature meniscus (the avascular zone) are less likely to heal than injuries in the outer one-third. 3 Most surgical treatments of meniscal tears in the avascular zone involve the partial or whole resection of the meniscus, which frequently leads to long-term degenerative joint changes and osteoarthritis. 4
Attempts to increase the local blood supply, 5 and techniques such as meniscal rasping, 6 synovial flap implantation 7 and fibrin clots or glues,8,9 have been investigated. However, more research is needed to clarify the clinical potential of these and related techniques. Although the clinical treatment of meniscal defects using tissue engineering implants has greatly improved, broader application of these implants is limited by some intrinsic obstacles.
Research has demonstrated that mincing articular cartilage or meniscus into fragments greatly increases the ability of these tissues to repair defects.10–12 This technique has been applied in human articular cartilage repair and has shown promising clinical results,13,14 but the mechanism of action of this technique remains unclear. The meniscal regeneration potential of the mincing method likely depends on the size of the implanted meniscal fragments, which could influence the activation state of the cells and their ability to migrate, although this has not been tested directly. The present study investigated the migratory, metabolic and proliferative abilities of fibrochondrocytes in meniscal fragments of three different sizes, in an in vivo subcutaneous implantation model in rabbits; confocal laser scanning microscopy, and histological and immunohistochemical evaluations, were undertaken.
Materials and methods
Animals
This study was conducted in the Department of Orthopaedics, First Affiliated Hospital of South China University, Hengyang, Hunan Province, China. The experiment was performed with the approval of the Ethics Review Committee of the First Affiliated Hospital of South China University (reference no. nhfy2010020). Care for the animals in this study was undertaken in accordance with the Guiding Directives for Human Treatment of Laboratory Animals, issued by the National Ministry of Science and Technology on 13 September 2006 (available at: http://www.most.gov.cn/fggw/zfwj/zfwj2006/200609/t20060930_54389.htm).
Thirty skeletally mature (age range 7–8 months) male New Zealand white rabbits with a mean weight of 2.8 kg (range 2.5–3.1 kg) were used in this study; the animals were obtained from Guangdong Medical Laboratory Animal Centre, Foshan, China. Animals were cared for in single cages under temperature- and light-controlled environmental conditions, with a 12-h light–dark cycle, in the Laboratory Animal Centre of South China University (Hengyang, Hunan Province, China). All rabbits received a standard laboratory diet and water ad libitum.
Harvesting of the menisci
Surgical procedures were performed according to a reported method. 15 Briefly, after general anaesthesia was induced with an intravenous injection of pentobarbital sodium (20–25 mg/kg), each rabbit was placed in the supine position. Surgery was were performed on the bilateral knees in each rabbit. After the limbs were disinfected, ∼5 ml of 1% lidocaine was injected subcutaneously, at the anterior and medial regions of the knee. A parapatellar longitudinal medial approach was used to expose the knee joint and the patella was laterally dislocated. Medial menisci of the bilateral knees were resected.
Experimental groups
Each meniscus was cut into two equal parts from the body of the meniscus with four parts in each rabbit, three of which were randomly chosen for use in the experiments. The outer one-third of each meniscus part was removed; the inner two-thirds were randomly minced into fragments of different sizes with a no. 11 surgical blade: 0.5 × 0.5 × 0.5 mm (group A); 1.0 × 1.0 × 1.0 mm (group B); 2.0 × 2.0 × 2.0 mm (group C). Three rectangles of fascia sheath approximately 15 × 15 mm were harvested from the distal medial thigh through the incision. The meniscal fragments were packed into the fascia sheath, then sutured with 3-0 nylon sutures. The meniscal fragment/fascia sheath packages were than implanted into the subcutaneous pockets located in the lateral thigh.
Ten rabbits were randomly euthanized each week at 1, 2 and 3 weeks after meniscal fragment implantation. The meniscal specimens were harvested. At each time point, specimens from two animals were analysed using confocal laser scanning microscopy; specimens from the remaining eight animals were analysed histologically and immunohistochemically.
Confocal laser scanning microscopy
To assess cell migration in the meniscal fragments, the superficial fascia sheath and fibrous scar were removed from the specimens. Next, specimens were stained using a Viability/Cytotoxicity Assay Kit for Animal Live & Dead Cells (30002; Biotium, Hayward, CA, USA). Briefly, samples were washed once at room temperature in 0.01 M phosphate-buffered saline (PBS; pH 7.2–7.4) for 5 min and labelled for 30 min at room temperature with 6 µl of EthD-III and 3 µl of calcein AM, in 3 ml of 0.01 M PBS (pH 7.2–7.4) from the kit. Specimens were washed three times, for 5 min each, in 0.01 M PBS (pH 7.2–7.4) at 20–25℃. Images were obtained using a confocal laser scanning microscope (LSM510; Carl Zeiss Instruments, Thornwood, NY, USA). Calcein AM is well retained within live cells, producing an intense uniform green fluorescence in live cells. EthD-III enters cells with damaged membranes and binds to nucleic acids, producing a bright-red fluorescence in dead cells.
Histological and immunohistochemical evaluation
Meniscal fragment samples used for histological evaluation were fixed for 24 h in 4% formalin and graded concentrations of dehydrated alcohol. Each specimen was embedded in paraffin wax and cut into 5-µm sections, which were stained with haematoxylin and eosin for standard histological evaluation. Additional sections were stained with haematoxylin followed by Safranin O and Fast Green (Sigma-Aldrich, St Louis, MO, USA) at room temperature. Safranin O was used to stain aggrecan; Fast Green was used as a counterstain to Safranin O because it labels collagen. Briefly, sections were deparaffinized and rehydrated using distilled water, then stained with haematoxylin for 10 min. After being washed in running tap water for 10 min, sections were stained with 0.05% Fast Green for 5 min, then rinsed quickly with 1% acetic acid solution for ≤10–15 s, followed by staining in 0.1% Safranin O solution for 5 min. The extent of staining with Safranin O was evaluated using a semiquantitative 3-point scoring system: 0, no staining; 1, faint staining; 2, dense staining.
Immunohistochemistry was performed following a standard protocol for paraffin wax-embedded sections. After sections were deparaffinized and rehydrated in a graded series of alcohol, they underwent antigen retrieval in 10 mM Tris/1 mM ethylenediaminetetra-acetic acid (pH 9.0) by heating in a microwave oven at 98℃ for 20 min. Sections were then washed in running tap water for 10 min, followed by intrinsic peroxidase blocking with 0.3% hydrogen peroxide in methanol for 10 min, which was part of a mouse- and rabbit-specific horseradish peroxidase/3,3′-diaminobenzidine (DAB) ABC immunohistochemistry detection kit (ab64264; Abcam®, Cambridge, UK). After tissue sections were blocked using a protein block from the same kit (ab64264; Abcam®), they were incubated with either antirabbit type I collagen antibody (1 : 800 dilution, ab6308; Abcam®), antirabbit type II collagen antibody (1 : 400 dilution, AF5710; Novus Biologicals, Cambridge, UK) or antirabbit proliferating cell nuclear antigen (PCNA) antibody, purchased in a prediluted state (ab912; Abcam®), overnight at 4℃. Sections were then washed three times for 5 min each time in 0.01 M PBS (pH 7.2–7.4) and incubated with biotinylated goat antipolyvalent secondary antibody (1 : 200 dilution, ab64264; Abcam®) at room temperature for 20 min. After washing three times for 5 min each time in 0.01 M PBS (pH 7.2–7.4), sections were then incubated with streptavidin peroxidase from the detection kit (ab64264; Abcam®) for 20 min at 37℃. After washing three times for 5 min in 0.01 M PBS (pH 7.2–7.4), sections were incubated with a mixture of 20 µl 2.5% DAB chromogen and 1 ml DAB substrate from the detection kit (ab64264; Abcam®) for 10 min at room temperature. Finally, sections were counterstained with haematoxylin, dehydrated through ethanol and xylene, and mounted/sealed with neutral gum. As a negative control, one section of each series was stained with 0.01 M PBS (pH 7.2–7.4), which substituted for the primary antibody. Sections were viewed and photographed using an Olympus AX70 microscope (Olympus, Tokyo, Japan).
An observer unaware of the treatment groups graded the meniscal area for staining patterns. The intensity of immunostaining for type I collagen, type II collagen and PCNA was evaluated using a semiquantitative 4-point scoring system: 0, no staining; 1, weak staining; 2, moderate staining; 3, strong staining. In the sections stained for PCNA, four fields were randomly chosen to be scored and all fibrochondrocytes were counted in the four chosen fields. When ≥30% of the fibrochondrocytes in a specimen were positively stained with ≥2 (i.e. moderate-to-strong staining on the above-defined scoring system) for PCNA, the specimen was scored as positive; in other cases, the specimen was scored as negative. 16 PCNA staining was used a marker of cell proliferation.
Statistical analyses
All statistical analyses were performed using the SPSS® statistical software package, version 17.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Scores produced for the Safranin O staining and immunostaining for types I and II collagen were presented as mean ± SD. Statistical analyses were undertaken using one-way analysis of variance and Fisher’s protected least-significant difference post hoc test. PCNA immunopositivity was recorded as the number of animals showing positive results and was assessed with the χ2-test. A P-value <0.05 was considered statistically significant.
Results
Results from the confocal laser scanning microscopy evaluation (Figure 1) showed that at 1 week after meniscal fragment implantation, some fibrochondrocyte migration to the surface of the meniscal fragments was observed in all three groups. At 2 weeks after meniscal fragment implantation, higher numbers of fibrochondrocytes had migrated to the meniscal fragment surface in all three groups. At some locations on the edges of the meniscal fragments in groups A and B, fibrochondrocytes had almost completely migrated to the surface, leaving blank cell-free zones within the implant; the blank cell-free zones appeared to be wider in group A compared with groups B and C. At 3 weeks after meniscal fragment implantation, the blank cell-free zones became wider in all three groups, as higher numbers of fibrochondrocytes accumulated at the meniscal fragment surface.
Representative fibrochondrocyte migration, observed under confocal laser scanning microscopy, in meniscal fragments from adult male New Zealand White rabbits in group A (A, D, G), group B (B, E, H), and group C (C, F, I) at 1 week (A–C), 2 weeks (D–F) and 3 weeks (G–I) after meniscal fragment implantation. Group A meniscal fragments were 0.5 × 0.5 × 0.5 mm; group B meniscal fragments were 1.0 × 1.0 × 1.0 mm; group C meniscal fragments were 2.0 × 2.0 × 2.0 mm. M, meniscal fragments; F, fibrous tissue; thin arrows indicate locations of fibrochondrocyte accumulation; wide arrows indicate cell-free zones where fibrochondrocytes were no longer located.
In haematoxylin and eosin-stained sections, fibrochondrocyte migration to the surface increased in a time-dependent manner (Figure 2). At 1 week after meniscal fragment implantation, fibrochondrocytes had accumulated at the surface of the meniscal fragments in groups A and B, but cell accumulation was not obvious in group C. At 2 and 3 weeks after meniscal fragment implantation, fibrochondrocyte accumulation at the surface could be seen in all three groups. Higher numbers of fibrochondrocytes could be observed at the meniscal fragment surface in group A than in groups B and C; and group B showed better fibrochondrocyte migration than group C.
Representative haematoxylin and eosin stained sections of meniscal fragments showing fibrochondrocyte migration in adult male New Zealand White rabbits: group A (A, D, G), group B (B, E, H), and group C (C, F, I) at 1 week (A–C), 2 weeks (D–F), and 3 weeks (G–I) after meniscal fragment implantation. Group A meniscal fragments were 0.5 × 0.5 × 0.5 mm; group B meniscal fragments were 1.0 × 1.0 × 1.0 mm; and group C meniscal fragments were 2.0 × 2.0 × 2.0 mm.
In sections stained with Safranin O and Fast Green at 3 weeks after meniscal fragment implantation, meniscal fragments were positively stained with Safranin O, which confirmed the presence of aggrecan (Figure 3; staining at weeks 1 and 2 not shown). There were no significant differences between the three groups in terms of the extent of Safranin O staining at 3 weeks after meniscal fragment implantation (Table 1).
Representative Safranin O and Fast Green staining (A, E, I) and immunohistochemical staining for proliferating cell nuclear antigen (PCNA) in fibrochondrocytes (B, F, J) and type I collagen (C, G, K) and type II collagen (D, H, L) in the extracellular matrix of meniscal fragments from adult male New Zealand White rabbits in group A (A–D), group B (E–H), and group C (I–L) at 3 weeks after meniscal fragment implantation. Group A meniscal fragments were 0.5 × 0.5 × 0.5 mm; group B meniscal fragments were 1.0 × 1.0 × 1.0 mm; and group C meniscal fragments were 2.0 × 2.0 × 2.0 mm. Comparison of the extent of histological staining with Safranin O for aggrecan and the immunohistochemical staining of type I and type II collagen and proliferating cell nuclear antigen (PCNA) in meniscal fragments from adult male New Zealand White rabbits (groups A, B and C) at 3 weeks after meniscal fragment implantation. Data presented as mean ± SD or n of animals (PCNA). Group A meniscal fragments were 0.5 × 0.5 × 0.5 mm; group B meniscal fragments were 1.0 × 1.0 × 1.0 mm; group C meniscal fragments were 2.0 × 2.0 × 2.0 mm. P < 0.05 versus group C, one-way analysis of variance and Fisher’s protected least-significant difference post hoc test for Safranin O staining, and types I and II collagen immunostaining; χ2-test for PCNA immunopositivity.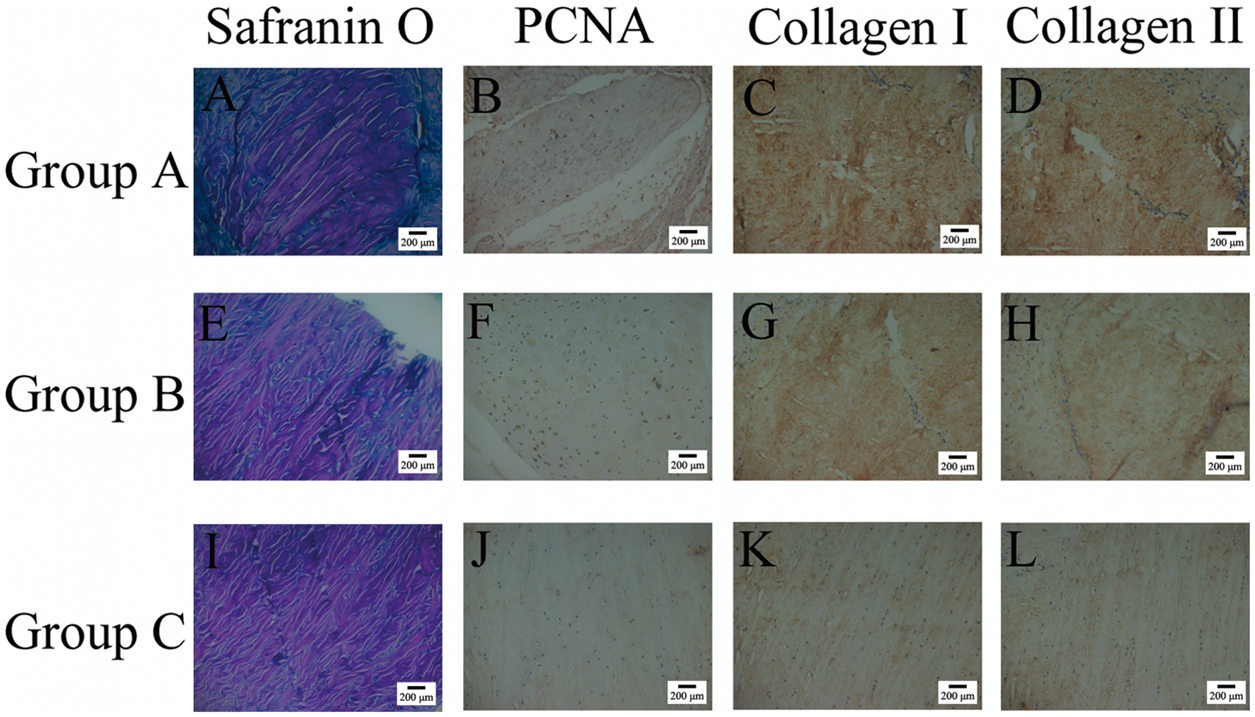

At 3 weeks after meniscal fragment implantation, the intensity of immunostaining of type I and type II collagen was significantly greater in groups A and B compared with group C (P < 0.05 for both comparisons) (Table 1, Figure 3; staining at weeks 1 and 2 not shown). The rate of PCNA immunopositivity was greater in group A than in groups B and C; and the difference was significant between groups A and C (P < 0.05).
Discussion
The meniscus is an inhomogeneous structure that, depending on the distance from the peripheral edge, shows important differences in composition, cellularity and vascularity. Data from canine studies show that the outer one-third of the meniscus is vascular and is composed of type I collagen. 17 Research undertaken in the rabbit shows that cells in this region are fibroblast-like, 18 whereas the inner two-thirds of the meniscus is avascular and is comprised of type I and type II collagen; this region contains fibrochondrocyte-like cells, and has a higher aggrecan content than the outer region. 18
Avascular meniscal and articular cartilage injury cannot heal spontaneously. However, when these tissues were minced into fragments of a small size, the ability to repair increased dramatically.10–14 When Lu et al. 10 implanted minced cartilage fragments (which had been mixed with scaffold materials without the use of cell culture) into cartilage defects, neocartilage was produced. When this minced-cartilage technique was compared with the technique of autologous chondrocyte implantation (ACI) in vivo, the findings demonstrated the superiority of cartilage fragment implantation over ACI techniques. 11
DeNovo® NT Natural Tissue Graft (Zimmer, Warsaw, IN, USA) is a new cartilage regeneration and repair product consisting of cartilage tissue pieces obtained from juvenile allograft donor joints. The cartilage is manually minced under aseptic conditions and no enzymatic digestion or biological manipulation is performed. The minced cartilage can be implanted in the cartilage defects and fixed by fibrin adhesive through a one-step arthroscopic technique. Because of the minimal manipulation of tissue, the Denovo® NT graft is classified as a 361 hTC/P product, and as such it does not require FDA premarketing approval. 13 This product is already available in the USA, for use in cartilage repair procedures and has shown promising clinical results. 14 As for meniscus repair, Kobayashi et al. 12 implanted autogenous meniscal fragments, wrapped in a fascial sheath, into rabbit meniscal defects and demonstrated the formation of homogenous fibrocartilage-like tissue.
Mincing the meniscus increases the surface area of the tissue. 10 Consequently, cells are better able to migrate to the meniscal surface. In the current study, fibrochondrocytes were able to migrate from within the minced cartilage fragments to the meniscal surface of each fragment. We observed an inverse relationship between tissue fragment size and efficiency of the fibrochondrocyte migration, using confocal laser scanning microscopy, and haematoxylin and eosin-stained histological sections.
In the current study, group A, which had the smallest meniscal fragment size (0.5 × 0.5 × 0.5 mm), showed the strongest intensity of immunostaining for type I collagen and type II collagen, and the highest rate of PCNA immunopositivity, compared with groups B (meniscal fragment size, 1.0 × 1.0 × 1.0 mm) and C (meniscal fragment size, 2.0 × 2.0 × 2.0 mm). Types I and II collagen are the main components of the meniscal extracellular matrix (ECM) and are synthesized by fibrochondrocytes located in meniscal tissue. 17 The increased immunostaining for types I and II collagen, and the higher rate of PCNA immunopositivity in this current study, suggest a higher rate of metabolic activity and cell proliferation of the fibrochondrocytes in the smaller meniscal fragments. Our current study has demonstrated that tissue fragmentation appears to have activated cell migration, metabolism and proliferation; and indicated that there was an inverse relationship between the migratory, metabolic and proliferative ability of the fibrochondrocytes and the meniscal fragment size.
Cartilage repair studies have shown that integrative repair requires the migration and proliferation of viable cells into the defect, as well as cell-mediated ECM biosynthesis, including collagen production and cross-linking.19,20 In conclusion, these current findings in a rabbit model suggest that the repair of avascular meniscal injuries, using minced meniscal fragments, might result in a greater level of healing compared with other techniques, and that this approach might offer a potential treatment for avascular meniscal injury repair.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This work was supported by grants from the Provincial Science and Technology Department of Hunan (no. 2012SK3156) and the Provincial Science Foundation of Hunan (no. 10JJ3094).
