Abstract
Objective
To compare the pharmacokinetics of the extended-release MMX
Methods
Using an open-label, randomized, three-period crossover, Latin square design, healthy male or female volunteers received single doses of 6 mg Uceris
Results
The study included 12 subjects. The 9 mg Uceris
Conclusion
Uceris was associated with a similar extent (AUC) of systemic exposure to budesonide compared with that following Entocort. However, for Uceris, the pharmacokinetic profile was delayed, a pattern consistent with greater colonic delivery of the active substance.
Introduction
Conventional treatments for mild-to-moderate ulcerative colitis (UC) include 5-aminosalicylates and systemic glucocorticoids.1–3 Nonsystemic (or locally acting) glucocorticoid therapies directly targeting the inflamed mucosa in inflammatory bowel disease have the potential for considerable safety advantages over comparable systemic treatments. Budesonide is a potent glucocorticoid with low systemic bioavailability (10–15%) due to extensive presystemic inactivation in which the drug is metabolized via the cytochrome P450 (CYP) enzyme CYP3A4 into two principle metabolites, 6β-hydroxybudesonide and 16α-hydroxyprednisolone.4,5 These metabolites have negligible glucocorticoid activity compared with the parent drug. 4 As a consequence, budesonide has only modest effects on the hypothalamic–pituitary–adrenal axis.6,7 As an example, a rectally-administered budesonide enema was found to have comparable efficacy to a rectally-administered prednisolone enema in UC, but was associated with significantly less plasma cortisol suppression. 8
Oral budesonide, administered as a plain formulation, is completely absorbed high in the gastrointestinal (GI) tract, making this formulation unsuitable for the treatment of UC, which requires delivery of active drug distally to the entire colon for optimal therapeutic effect. Entocort® EC (Astra Zeneca, London, UK) is a budesonide formulation containing granules that are coated to prevent dissolution in gastric pH. This formulation releases budesonide as it passes through the small intestine (pH > 5.5), delivering the steroid to the distal ileum and allowing treatment of terminal ileal or right-sided colonic Crohn‘s disease.5,9,10 However, in contrast to Crohn's disease, UC typically involves the colon (and usually the left colon): 11 a pattern of disease that requires more distal distribution of budesonide than can be achieved by Entocort EC®.
An extended-release multimatrix (MMX
The GI transit and pharmacokinetics of Uceris® have been evaluated using γ-scintigraphy of 153Sm-labelled tablets containing 9 mg of budesonide. 13 The appearance of budesonide in the systemic circulation was almost exclusively (96%) associated with the absorption of the dose in the colon.
The efficacy of the once-daily tablet formulation of Uceris® in patients with active mild-to-moderate UC has been evaluated in the COlonic RElease budesonide (CORE) I and CORE II studies.14,15 These similarly designed studies were randomized, double-blind, double-dummy, placebo-controlled trials, using combined clinical and endoscopic remission at 8 weeks as the primary endpoint. Pooled analyses of the CORE I and CORE II studies demonstrated that the remission rates for patients treated with 9 mg Uceris® and 6 mg Uceris® once daily was 17.7% (P = 0.0002) and 10.9% (P = 0.0692), respectively, compared with 6.2% for placebo. 16 In the CORE I study, significantly higher rates of symptom resolution were achieved in patients treated with 9 mg Uceris® (28.5%) and 6 mg Uceris® (28.9%) versus placebo (16.5%; P < 0.05). 14 Similarly, in the CORE II study, significantly higher rates of symptom resolution were achieved in patients receiving 9 mg Uceris® (23.9%) versus placebo (11.2%; P < 0.05), but not in patients treated with 6 mg Uceris® (13.8%). 15
The objective of the current study was to compare the pharmacokinetics of two doses (6 and 9 mg) of the extended-release Uceris® formulation of budesonide with that of 9 mg Entocort® EC (supplied as 3 × 3 mg capsules).
Patients and methods
Study design
This was a single-center, open-label, single-dose, phase 1, randomized 3-period crossover study designed to describe the pharmacokinetics of a new extended-release formulation of budesonide using the MMX® technology (Uceris®). Randomization followed a Latin square design ensuring balance (within sex) for sequence. Pharmacokinetics data from this formulation were to be contrasted with data from controlled ileal release budesonide provided as 3 × 3 mg capsules (Entocort® EC). The study also included an evaluation of the pharmacokinetics of 6 mg Uceris®.
This phase 1 study was conducted by Cross Research SA (principal investigator: Antonio Rusca MD FMH, Phase 1 Unit, Arzo, Switzerland) between 5 March and 19 April 2007. It was performed in accordance with the relevant guidelines of the Declaration of Helsinki (1964). Study subjects were informed of the potential benefits and risks of study participation, and entered the study after providing written informed consent. The study protocol, and all the other relevant documentation, were reviewed and approved by an independent Ethics Committee (Comitato Etico Cantonale, Canton Ticino) on 16 January 2007 (reference number: 1863). The Federal Health Authorities (Swissmedic) assigned the reference number 2007DR1050 to the study on 27 February 2007.
Study population
Healthy male or female volunteers, aged 18–55 years, body mass index (BMI) 18–29 kg/m2, were eligible to participate in the study. Subjects were recruited from the local population of Arzo, Switzerland; they had no history of inflammatory bowel disease or other GI disease. Subjects were on normal balanced diets (caloric intake between 1600 and 3500 kcal/day), with no history of medication (prescription or over-the-counter) in the 2 weeks prior to the study and no history of recreational drug use, or excessive alcohol, caffeine, or tobacco consumption. All women of childbearing potential used an acceptable method of birth control for ≥1 month before dosing, and had a negative serum pregnancy test at screening. Subjects were excluded if they had a history of renal, hepatic, GI, cardiovascular, respiratory, skin, hematologic, endocrine or neurological disease or clinically relevant abnormalities on physical examination or evaluation of laboratory tests including electrocardiogram (ECG), biochemistry and hematology.
Procedures
Study treatments
Study treatments were administered under fasting conditions (≥10 h since previous meal) with a washout interval of ≥5 days between each study period. The randomization sequence was determined using a computer-generated schedule that included two blocks of six treatment sequences and was balanced by sex. Prior to each study period, subjects were admitted to the clinical research laboratory (Phase 1 Unit, Cross Research SA, Arzo, Switzerland), where they stayed for 36 h after dosing. On each admission to the laboratory, recent medical history was reviewed (including use of alcohol, drugs, and concomitant medications). An alcohol breath test, urine drug screening, and a urine pregnancy test (when applicable) were performed. During confinement, subjects were not permitted to smoke more than five cigarettes per day and coffee, tea, and xanthine-containing products (i.e. cola, chocolate, etc.) were not allowed 72 h prior to each drug administration and during confinement. Grapefruit and grapefruit juice were forbidden 24 h before first drug administration until the end of the study. Alcohol was forbidden 36 h before each drug administration and during confinement. Study medication was administered with 240 ml of water at 08.00 h ± 1 h under fasting conditions. Subjects were instructed to swallow the dose forms (tablets or capsules) whole (without chewing).
Blood collection and analysis
Blood samples were collected using a cannula placed intravenously. Samples were obtained at the following timepoints: prior to dosing (0 h); at 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 14, 16, 18, and 20 h postdose; on day 2 at 24, 30, and 36 h post-dose. Blood samples (8 ml) were collected in polypropylene heparinized tubes, stored on ice, and centrifuged (2500
Samples were packed in solid CO2 prior to shipping for analysis by Pharmakin GmbH, Neu-Ulm, Germany. Plasma budesonide concentrations were determined using validated liquid chromatography–mass spectrometry/mass spectrometry methods, with a 50 pg/ml lower limit of quantitation (LLOQ). At LLOQ, inter- and intra-assay precision were 4.88% and 6.35%, respectively. Values below the limit of quantification (<50 pg/ml) were not plotted; the late appearance of material in the systemic circulation was particularly evident for the concentration profiles for 9 mg Uceris®. Values below the limit of quantification were treated as zero during pharmacokinetic analysis. For clarity, individual plasma concentration profiles for 6 mg Uceris® are not presented, to allow direct comparison of dosing with 9 mg Uceris® and 9 mg Entocort® EC.
Pharmacokinetic analysis (noncompartmental analysis) was performed with Kinetica software, version 4.4 (Thermo Electron, Waltham, MA, USA). Maximum concentration (Cmax), time to maximum concentration (Tmax), and the time of first appearance (Tlag) were obtained from the concentration data. These data were used to determine the area under the concentration–time curve (AUC), including AUC0–last, and, where appropriate, AUC0–∞, and half-life (t½). Mean transit time (MTT) was calculated using the ratio of the first moment of the AUC and AUC itself (AUMC/AUC0–∞). Mean arrival time (MAT) was derived from the relationship, MTT = MRT + MAT, where MRT indicates mean residence time. Since MRT requires intravenous administration (not done in this study), an externally-derived estimate (3.1 h) from a previous study was used. 17
Safety assessments
Safety assessments included adverse events; physical examinations and 12-lead ECG examinations, which were performed at screening and final visit; vital signs performed on day 1 at predose (0 h) and at 4 and 16 h postdose, and on day 2 at 24 and 36 h post-dose; and routine laboratory tests (hematology, blood chemistry, urinalysis) performed at screening and final visit.
Statistical analyses
This was an exploratory pharmacokinetic study. Accordingly, the sample size was selected without consideration of formal power calculations for testing bioequivalence, however a sample size of 12 subjects was considered sufficient to provide a description of the pharmacokinetic response. A total of 13 subjects were enrolled to ensure that 12 subjects completed the study procedures. Pharmacokinetic analyses were conducted for the 12 subjects who completed the study (per protocol population). Safety assessments considered all enrolled subjects.
Descriptive statistical summaries of the pharmacokinetic data, calculations of point estimates and confidence intervals for analysis of bioequivalence were prepared, together with summaries of demographic variables and safety data. In keeping with the usual approach for bioequivalence studies, point estimates and confidence intervals were provided for AUC and Cmax. Other pharmacokinetic parameters were not formally tested. Statistical analyses were performed using SAS® software, version 9.1.2 (SAS Institute, Cary, NC, USA), of log-transformed data in analysis of variance models appropriate for the Latin square design of the study.
Results
A total of 20 healthy subjects were screened to identify 13 eligible subjects who were randomized to a treatment sequence and received study drug. One subject withdrew for personal reasons (not specified) prior to completing study procedures, resulting in 12 subjects completing the study and composing the per protocol population, of which all subjects were Caucasian and six (50%) were male. Mean ± SD age was 37 ± 10 years and BMI was 23.0 ± 3.1 kg/m2.
Inspection of individual subject plasma drug concentration profiles (Figure 1) shows that administration of 9 mg Uceris® was associated with a delay in the appearance of budesonide in the systemic circulation, with the consequence that plasma concentrations over the early postdosing interval (0–6 h) appeared to be lower for 9 mg Uceris® than for 9 mg Entocort® EC. In contrast, plasma drug concentrations observed ≥12 h after dosing tended to be greater following 9 mg Uceris® treatment compared with those following 9 mg Entocort® EC treatment (Figure 1; Table 1). Median Tlag (the time point at which budesonide was first observed above the LLOQ) occurred 6 h after dosing for both 9 mg Uceris® and 6 mg Uceris® but only 1 h after dosing with 9 mg Entocort® EC (Table 1). Mean MAT was longer for 9 mg Uceris® and 6 mg Uceris® compared with 9 mg Entocort® EC (Table 1).
Systemic budesonide plasma concentration profiles for individual study subjects (S1–S12), following administration of 9 mg Entocort® EC (orange) or 9 mg Uceris® (blue). Values below the lower limit of quantification (<50 pg/ml) were not plotted, which was particularly evident in early samples collected for the 9 mg Uceris® plasma profiles. Mean arrival times (MATs), for Entocort® EC and Uceris®, respectively, are provided in the upper right of each profile. For subject S3, the MAT could not be calculated for 9 mg Entocort® EC. For subject S9, the asterisk indicates that the value was influenced by a poorly defined elimination phase for 9 mg Uceris®. Pharmacokinetic parameters after administration of a single dose of 6 mg Uceris®, 9 mg Uceris® or 9 mg Entocort® EC in 12 healthy study subject. Data presented as mean (SD) or median (range). Cmax, maximum concentration; AUC, area under the concentration–time curve; t½, half-life; MAT, mean arrival time; Tmax, time to maximum concentration; Tlag, time of first appearance.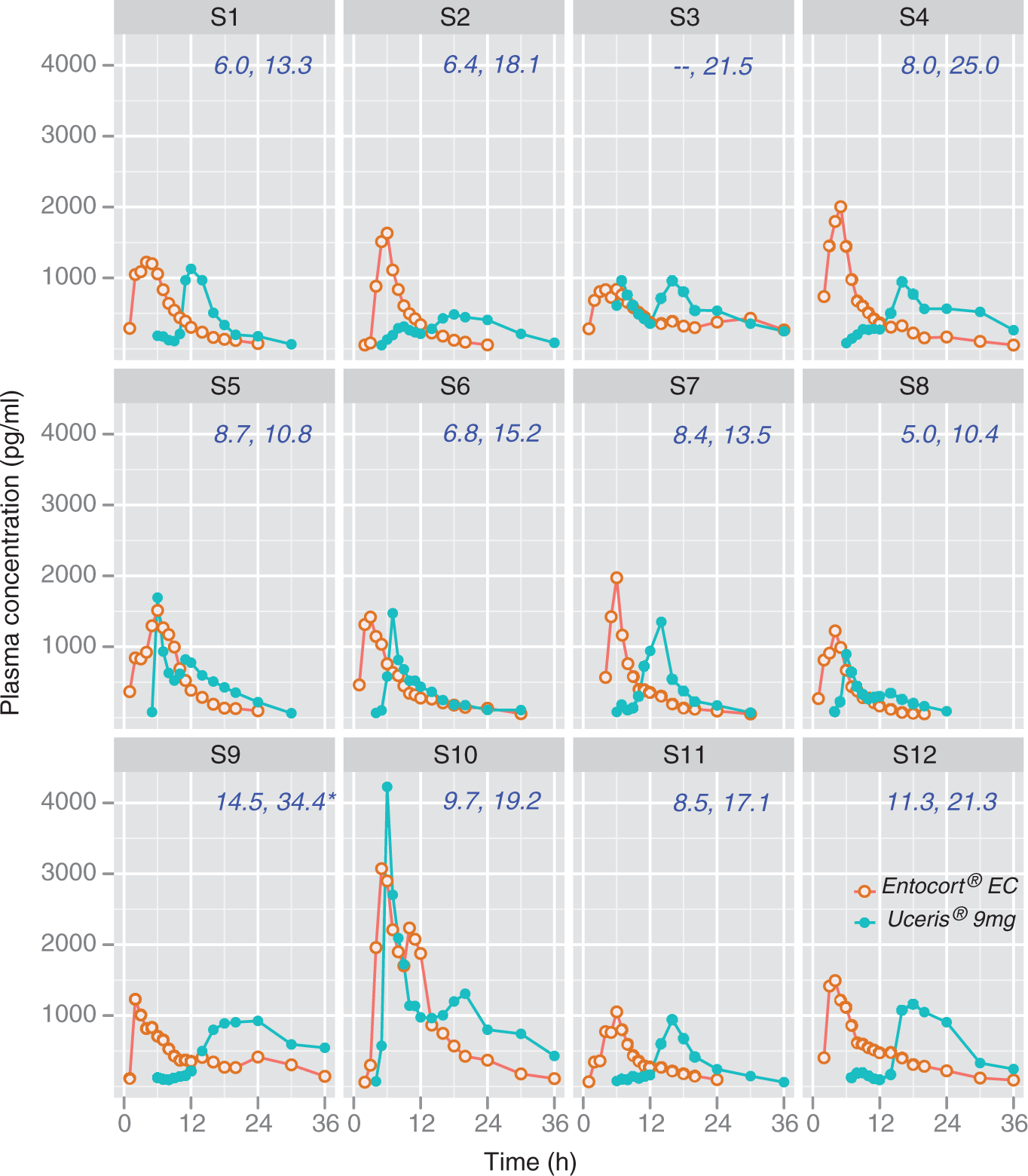

Systemic exposure following 9 mg Uceris® administration was comparable with that following 9 mg Entocort® EC. The ratio of the geometric mean Cmax (Uceris®/Entocort® EC) was 79%, with a 90% confidence interval (CI) around this estimate of 63–100%. For AUC0–last, the comparable ratio was 91% (90% CI 77%, 108%). Median Tmax was longer for 9 mg Uceris® and 6 mg Uceris® compared with 9 mg Entocort® EC. The mean ± SD terminal half-life (where calculable) was at 7.5 ± 2.9 h (n = 11) for 9 mg Uceris® and 7.7 ± 1.8 h (n = 11) for 9 mg Entocort® EC.
Three subjects experienced adverse events during the study, including two events of moderate upper respiratory tract infection (6 mg Uceris® and 9 mg Entocort® EC) and one event of mild headache (9 mg Entocort® EC). Study drug was otherwise well tolerated with no meaningful effects on vital signs, ECG, laboratory parameters, or physical examination findings.
Discussion
This study showed that the pharmacokinetics of budesonide following administration of Uceris® differed from those following administration of Entocort® EC. The primary difference between the two budesonide formulations was observed in the lag time (Tlag), or the time between oral administration of the dose form and first appearance of budesonide in the systemic circulation.
The study demonstrated that the pharmacokinetic parameters describing exposure to systemic budesonide were similar following administration of the two dose forms, Uceris® and Entocort® EC. In this study, which was designed with an arbitrary number of subjects and without prior knowledge of pharmacokinetic variability of the test formulations, the Uceris® formulation of budesonide did not exhibit bioequivalence with Entocort® EC at the same dose using conventional regulatory criteria for equivalence. The point estimate for the ratio of log-transformed AUC0–last (Uceris®/Entocort® EC) was 91% (90% CI 77%, 108%). For Cmax, the corresponding data were 79% (90% CI 63%, 100%).
The processes leading to systemic availability of budesonide take substantially longer for Uceris® compared with Entocort® EC. The MAT, calculated using an external estimate of MRT from another study of budesonide, in which the drug was given as an intravenous bolus, 17 was 16.9 h for Uceris® and 8.5 h for Entocort® EC.
These current data are consistent with the different dissolution and release characteristics of the two formulations of budesonide. As expected, the multimatrix structure of the Uceris® tablet, which is designed to release the active ingredient more distally in the GI tract, had an appreciably greater Tlag and Tmax, and a somewhat reduced Cmax, with an extended ‘tail’ observed in the pharmacokinetic profile, when compared with Entocort® EC.
A high degree of variability in the pharmacokinetics of these formulations is consistent with the substantial contribution that first-pass metabolic processes play in the appearance of budesonide in the systemic circulation. 13 Single doses of both Uceris® and Entocort® EC were well tolerated in this trial.
The current study was limited by the fact that the study was not powered to test bioequivalence. Additionally, the pharmacokinetic sample schedule, which involved no observations after 36 h post-dose, led to difficulties in full characterization of the terminal phase, in some subjects. Application of the results of this study in a healthy subject population to patients with UC may be limited by differences in GI transit, the pH profile in the GI tract, and CYP enzyme activity, all of which may influence the pharmacokinetics of budesonide for these formulations.
The current study provided pharmacokinetic data suggesting that the extended-release formulation of budesonide with MMX® technology (Uceris®) undergoes in vivo dissolution in a more distal region of the GI tract compared with the Entocort® EC formulation, which is consistent with the release mechanisms of these formulations as demonstrated in a previously-conducted γ-scintigraphy study. 13 This difference resulted in a substantially delayed lag time, MAT, and Tmax for Uceris® compared with Entocort® EC. Systemic budesonide exposure (AUC) and Cmax were comparable for Uceris® and Entocort® EC.
Footnotes
Acknowledgements
The authors would like to thank Dr Edward Schweizer, MD, Paladin Consulting Group, Princeton, NJ, USA for his contributions in developing the initial draft of the manuscript. Funding for the development of the manuscript was provided by Santarus Inc., San Diego, CA, USA.
Funding
This study was funded by Cosmo Technologies Ltd, Dublin, Ireland.
Declaration of conflicting interest
Dr Andrew Nicholls is a consultant to Santarus Inc., San Diego, CA, USA. Drs Yun Hardiman, Raúl Harris-Collazo and Michael Huang are full-time employees of Santarus Inc., San Diego, CA, USA. Richard Jones and Dr Luigi Moro are full-time employees of Cosmo Technologies Ltd, Ireland.
