Abstract
Objective
To evaluate the feasibility of using serum microRNA (miR)-221 as a noninvasive diagnostic and prognostic biomarker in epithelial ovarian cancer (EOC).
Methods
In this retrospective study, real-time reverse transcription–polymerase chain reaction was used to measure miR-221 expression in serum samples from patients with EOC and healthy age-matched controls. Correlations between serum miR-221 levels and clinicopathological factors and prognosis were explored.
Results
Serum miR-221 was upregulated in patients with EOC (n = 96) compared with healthy controls (n = 35). The level of serum miR-221 expression was significantly associated with International Federation of Gynecology and Obstetrics stage, and tumour grade. Furthermore, multivariate analysis of overall survival showed that high serum miR-221 expression was an independent unfavourable prognostic factor in EOC.
Conclusions
These findings indicate that serum miR-221 may have a role as a novel diagnostic and prognostic marker, and may have potential as a therapeutic target in EOC.
Keywords
Introduction
Ovarian cancer is the most lethal of he common gynaecological malignancies, with over 204 000 new cases and 125 000 deaths reported annually worldwide, accounting for 4.0% of all cancer cases and 4.2% of all cancer deaths in women. 1 Although the prognosis for women with ovarian cancer has improved slightly over the past 25 years, 5-year survival rates remain <50%. 2 The events that initiate ovarian cancer and lead to disease progression are poorly understood. Novel biomarkers, with high sensitivity and specificity, are urgently needed as diagnostic tools and targets for therapy in ovarian cancer.
MicroRNAs (miRs) are endogenous, small, noncoding RNAs, approximately 22 nucleotides in length, that negatively regulate gene expression via RNA interference machinery. 3 Reserach has shown that miRs can act as either tumour suppressors or oncogenes by regulating processes such as proliferation, the cell cycle, apoptosis, invasion and metastasis. 4 Growing evidence has indicated the possible use of miR expression profiles to distinguish between normal and neoplastic tissues, leading to the identification of new diagnostic and/or prognostic markers. 5 It has been confirmed that miRs are present in human serum and plasma in a remarkably stable form that is protected from endogenous RNase activity, 6 and that circulating miRs have the potential to serve as noninvasive diagnostic biomarkers for various cancers. 7
One of the first members of the miR family to be described, miR-221, is located on chromosome X and has been reported to be upregulated in various tumour types, including ovarian cancer.8–10 However, the precise role of serum miR-221 expression in ovarian cancer has not been elucidated. In the present study, serum levels of miR-221 were investigated in patients with epithelial ovarian cancer (EOC), and the feasibility of using serum miR-221 as a noninvasive diagnostic and prognostic biomarker for EOC was evaluated.
Patients and methods
Patients
Patients with primary EOC who were treated at the Second Hospital of Shandong University, Jinan, China, between July 2005 and October 2007, were included in this retrospective study. In order to be eligible for study inclusion, patients had to be 18–70 years of age, to have an Eastern Co-operative Oncology Group performance status of 0 or 1, and to have adequate haematological, hepatic, renal and cardiac function. In addition, patients who had a previous or secondary malignancy, were pregnant and/or had previously undergone radiotherapy, chemotherapy or immunotherapy were excluded from the study.
All patients received standard surgery aimed at providing maximal tumour resection (including hysterectomy, bilateral salpingo-oophorectomy, pelvic and/or para-aortic lymphadenectomy and omentectomy). After resection, platinum-based chemotherapy was administrated for at least six cycles. Surgical staging was determined, based on the International Federation of Gynecology and Obstetrics (FIGO) criteria. Tumour grade was designated as low or high, according to criteria developed at the University of Texas MD Anderson Cancer Center. 11 The age of the patient and the histological type of the tumour were also recorded.
Ethical approval was obtained from the Institutional Review Board of the Second Hospital of Shandong University, Jinan, China. Written informed consent for the use of serum samples was obtained from each study participant.
Measurement of miR-221 Expression
Before surgery, 5-ml blood samples were obtained from all patients by peripheral venous puncture. Blood samples from healthy age-matched volunteers (recruited from the local population in Jinan, China) were used as controls. Immediately after collection, blood samples were centrifuged at 2800 g for 10 min at 4°C to spin down the blood cells; the serum supernatant was then transferred into fresh tubes, stored in liquid nitrogen and kept frozen at −80°C until RNA extraction.
For RNA extraction, frozen serum samples were thawed and transferred into Eppendorf tubes. Then, 50 fmol mmu-miR-295 mimics (Qiagen, Valencia, CA, USA) were added to 100 µl of serum and incubated at 37°C for 5 min. RNA isolation was carried out using a total RNA purification kit for sera (Norgen Biotek, Thorold, Canada) according to the manufacturer’s instructions. Serum RNA preparations were quantified using a DU® 800 spectrophotometer (Beckman Coulter, Brea, CA, USA), then pretreated with RNase-free DNase I (Promega, Madison, WI, USA) to eliminate potential DNA contamination.
Expression of miR-221 was measured using real-time reverse transcription–polymerase chain reaction (RT–PCR) as previously described, with minor modification.12,13 Briefly, 20 µl of reverse transcription reaction mixture containing 40 ng of total RNA, 5 × reverse transcription buffer, pH 7. (Qiagen), 10 mM of each deoxynucleotide triphosphate (Takara Biotechnology, Dalian, China), 5 U/µl of RNase inhibitor (Takara) and 0.25 µl of antisense looped primer for miR-221 (5′-GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACGAAACCCA-3′) (Qiagen) underwent cycles of 16°C for 15 min, 42°C for 60 min and 85°C for 5 min.
Subsequently, real-time quantification was performed using the QuantiTect® SYBR® Green PCR kit (Qiagen) and the Applied Biosystems 7300 sequence detection system (Applied Biosystems, Foster City, CA, USA) according to the manufacturers’ instructions; mmu-miR-295 was selected as the internal normalization control.14–16 The cycle threshold (CT), which was defined as the number of PCR cycles required for the fluorescent signal to be higher than a threshold indicating baseline variability, was recorded. miR-221 expression levels were then normalized against the mmu-miR-295 expression levels using the equation 2−Δ C T , where ΔCT = (CT miR-221−CT mmu-miR-295); 17 thus the ΔCT value correlated negatively with the serum level of miR-221.
Follow-up Period
All patients were followed up until March 2012. Overall survival was defined as the time interval between the start of treatment and the date of death or the end of the follow-up period.
Statistical Analyses
Data were expressed as the mean ± SD. Student’s t-test was used to analyse differences in miR-221 expression levels between patients and controls, and between patients grouped according to clinicopathological features. Discriminant function analysis was performed to ascertain how many patients would be correctly classified on the basis of serum miR-221 levels. The association between expression levels of miR-221 and overall survival was analysed using the Spearman correlation coefficient. Multivariate analysis of prognostic factors was performed using the Cox regression model.
All statistical analyses were carried out using SPSS® software, version 17.0 (SPSS Inc., Chicago, IL, USA). A two-sided P-value <0.05 was considered to be statistically significant.
Results
A total of 96 patients with primary EOC with a mean (±SD) age of 58.6 ± 6.2 years were included in the study. Of these, nine tumours were FIGO stage I, 23 were stage II, 44 were stage III and 20 were stage IV.
Blood samples from 35 healthy age-matched volunteers were used as controls.
Serum miR-221 in EOC
The mean (± SD) ΔCT value was 5.66 ± 0.71 in the 96 samples from patients with EOC and 7.35 ± 0.82 in healthy controls (P < 0.001). Thus, serum miR-221 was significantly upregulated in patients with EOC.
Change in polymerase chain reaction cycle threshold (ΔCT), which negatively correlates with serum microRNA-221 (miR-221) expression, according to clinicopathological features in patients with epithelial ovarian cancer.

Data presented as mean ± SD.
Student’s t-test.
International Federation of Gynecology and Obstetrics criteria.
CT = (CT miR-221−CT mmu-miR-295), where CT is the polymerase chain reaction cycle threshold.
NS, no statistically significant between-group differences (P ≥ 0.05).
Discriminant function analysis of serum microRNA-221 expression in patients with epithelial ovarian cancer.
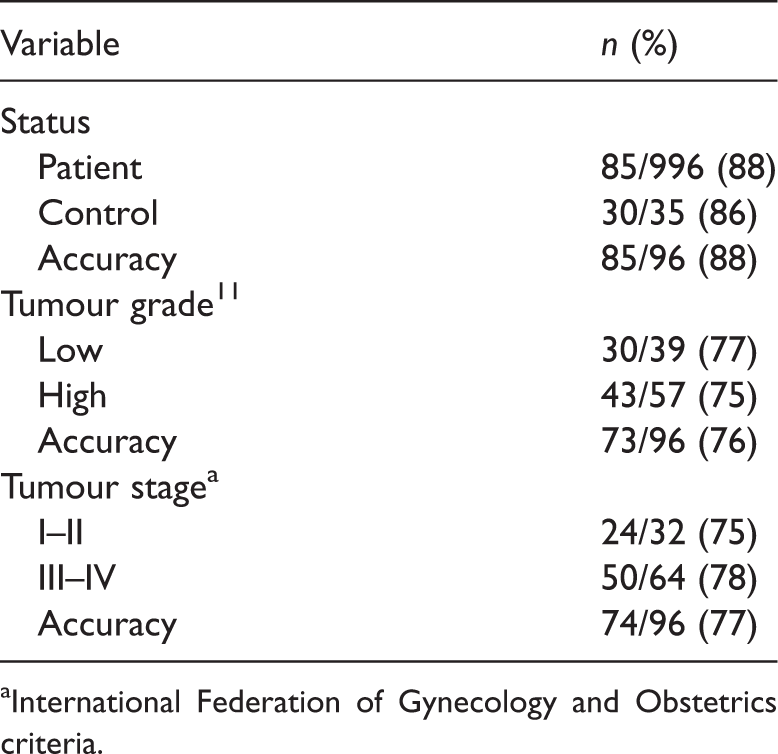
International Federation of Gynecology and Obstetrics criteria.
Prognosis
At the end of the study period, 39 of the 96 patients were still alive, including 18 patients with no evidence of ongoing disease. A total of 51 patients died during the study period as a result of EOC; six patients died from other causes. Spearman correlation coefficient analysis showed a significant correlation between serum miR-221 levels (ΔCT) and overall survival (r = 0.512, P = 0.005).
Univariate and multivariate survival analyses of prognostic factors in patients with epithelial ovarian cancer (n = 96).
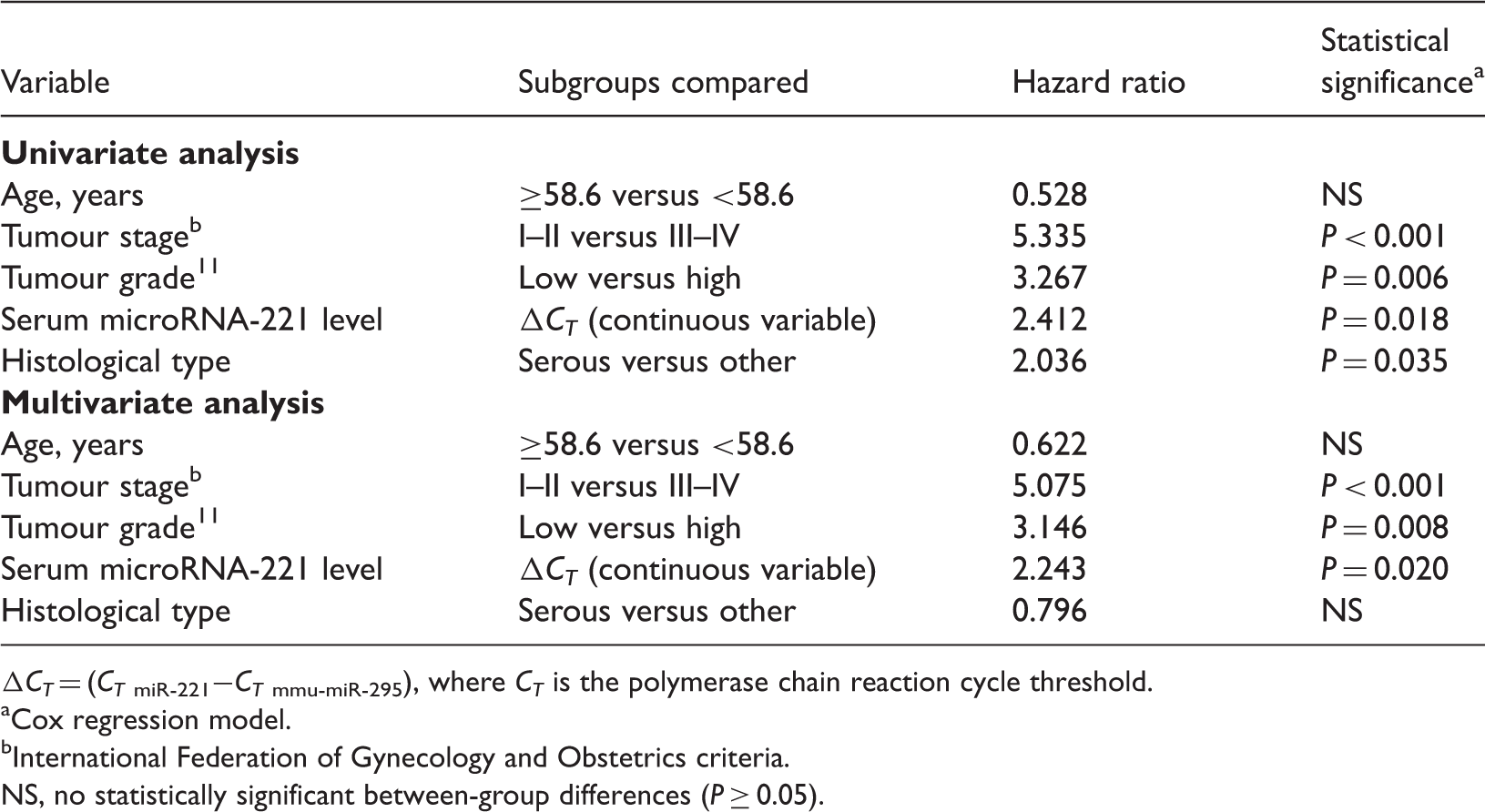
CT = (CT miR-221−CT mmu-miR-295), where CT is the polymerase chain reaction cycle threshold.
Cox regression model.
International Federation of Gynecology and Obstetrics criteria.
NS, no statistically significant between-group differences (P ≥ 0.05).
Discussion
Ovarian cancer is a common gynaecological malignancy and is a leading cause of cancer-related death. 2 Elucidation of factors affecting the behaviour of ovarian cancer is crucial in improving the prognosis of patients with this condition. The discovery of miRs has broadened the understanding of carcinogenesis. It has been proposed that there are over 1000 miRs in the human genome, 18 and these miRs have been reported to play important functions in vivo, including roles in cancer development, angiogenesis and immune response.19–21 Although many miRs are expressed in tissues and tumour cells, the development of such miRs as biomarkers requires tissue collection by invasive methods, rather than the more convenient approach of studying peripheral blood. In the present study it was demonstrated that patients with EOC had significantly elevated levels of serum miR-221 compared with healthy controls, and that serum miR-221 expression was associated with FIGO stage, tumour grade and overall survival. These results suggest that the serum miR-221 level may serve as a useful diagnostic and prognostic biomarker for EOC.
Overexpression of miR-221 has been confirmed in many types of cancer including gastric cancer, 22 kidney and bladder cancer, 23 colorectal cancer 24 and breast cancer. 25 Studies have also reported that miR-221 was upregulated in serum/plasma in several malignancies. Li et al. 16 reported that miR-221 was overexpressed in human hepatocellular carcinoma serum samples, and that a high level of miR-221 expression was correlated with tumour size, cirrhosis, tumour stage and overall survival. Guo et al. 26 demonstrated different circulating miR-221 levels in patients with natural killer/T-cell lymphoma and controls, and a reverse correlation between plasma miR-221 level and patient survival after treatment. Kanemaru et al. 5 found that serum levels of miR-221 were significantly increased in patients with malignant melanoma, and were associated with tumour stage and thickness. Moreover, they observed a tendency for miR-221 levels to decrease after surgical removal of the primary tumour and to increase again at recurrence. Therefore miR-221 may play an important role not only in tumour initiation but also in the development and progression of malignancies.
The molecular mechanisms that link miR-221 overexpression to tumorigenesis and development are not well understood. Several functional targets have been corroborated, including mRNA associated with p27/kip1 and p57/kip2 (two important cell-cycle inhibitors and tumour suppressors),27,28 c-Kit (a common oncogene encoded on chromosome 4q11–21), 29 Bmf (which belongs to the Bcl-2 family and may be important for sensing intracellular damage and triggering apoptosis) 30 and the tumour suppressor, PTEN. 31 However, there is not a one-to-one connection between miRs and target mRNAs; an miR can have over 100 targets 32 and one mRNA can be regulated by a variety of miRs. Therefore further elucidation of the molecular characteristics of miR-221 remains an important aspect of future investigations.
Early detection of EOC is essential to improve patient survival. Although several biomarkers associated with EOC carcinogenesis and tumour development have been identified, none of them show 100% sensitivity and specificity. In the present study, serum miR-221 was significantly upregulated in patients with EOC, but since miR-221 is also overexpressed in various other malignancies, this marker may not have high specificity for EOC. However, using serum miR-221 as the sole variable, tumour grade and tumour stage were correctly classified in 76% and 77% of patients, respectively, showing that the serum miR-221 level is a discriminant factor in patients with EOC.
The progression of EOC is a complicated process, but miR-221 may serve as an important node in this large and intricate network. The identification of new genes and biomarkers is an ongoing task and warrants further investigation.
In conclusion, the present study showed that serum miR-221 levels are elevated in patients with EOC compared with controls and that serum miR-221 expression is associated with FIGO stage and tumour grade. Furthermore, a higher plasma miR-221 level correlated with a less favourable long-term outcome. These findings indicate that serum miR-221 may have a role as a novel diagnostic and prognostic marker, and may have potential as a therapeutic target in EOC.
Footnotes
Declaration of Conflicting Interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
