Abstract
Eighty patients with osteoarthritis who underwent unilateral total knee arthroplasty were randomly assigned to two groups: the trial group received an intraoperative intra-articular injection of magnesium sulphate and ropivacaine, and the control group received an injection of normal saline. All patients received patient-controlled analgesia with morphine for 48 h post-operatively. It was found that an intra-articular injection of magnesium sulphate and ropivacaine significantly reduced morphine consumption during the 0 - 24 h post-operative period and total 48-h postoperative morphine consumption. Pain scores at rest and during motion in the trial group were significantly lower than in the controls during the first 24 h post-operatively. The time to be able to perform a straight leg raise and to reach a 90° knee flexion was significantly shorter in the trial group compared with the controls. This study demonstrated that an intra-operative intra-articular magnesium sulphate and ropivacaine injection reduced the use of post-operative morphine.
Keywords
Introduction
Good pain relief is important for postoperative rehabilitation for total knee arthroplasty (TKA) and it may influence the overall post-operative outcome. At present, post-TKA analgesia includes continuous epidural infiltration, peripheral nerve block, patient-controlled analgesia (PCA), continuous intra-articular infusion of analgesics and local intra-articular analgesic injection. Among them, continuous epidural infiltration and femoral and/or sciatic nerve block improve post-operative pain control and reduce consumption of narcotics 1 at the expense of other potential problems, such as epidural bleeding (with prophylactic anticoagulation therapy), infection, urinary retention, diminished muscle control and nerve damage.1–4 PCA administration of opioids is often associated with nausea and vomiting, respiratory depression, drowsiness, pruritus, reduced gut motility and urinary retention. 5 Continuous intra-articular infusion of analgesics is associated with large effusion of the surgical wound, and may provide direct access for infectious agents. 6 Providing analgesia locally in the area of the surgical site, with minimal systemic side-effects, is an attractive option. 7 It has been reported that intra-operative magnesium sulphate reduces analgesic requirements and improves post-operative analgesia.8.9 Thus, the present prospective, double-blind, randomized, placebo-controlled study was performed to assess the clinical efficacy and safety of an intra-operative intra-articular injection of a magnesium/ropivacaine mixture during TKA.
Patients and methods
Study Participants
Patients who underwent unilateral TKA for osteoarthritis between January and October 2008 in the Department of Orthopaedics, Changzheng Hospital, Shanghai, China, were eligible for enrolment in this study. The inclusion criteria included: age < 76 years; body weight 43 – 70 kg; and being scheduled to undergo routine total primary unilateral knee arthroplasty with a standardized regimen of spinal anaesthesia. Exclusion criteria included: known allergy to one of the study medications; major renal, hepatic, or cardiovascular dysfunction; prior treatment with opioids and calcium channel blocking drugs; and neuromuscular disease. This study was approved by the Institutional Ethics Committee of the Changzheng Hospital, Shanghai, China, and each patient provided written informed consent.
Study Design
Based on our pilot study in 15 patients (unpublished data), a sample size of 40 patients in each group was estimated in order to be able to detect a difference in the mean amount of analgesic consumption from 60 mg in the placebo group to 36 mg in the trial group with a standard deviation of ± 30 mg, a two-sided P-value of 0.05 and an approximate power of 80%.
A biostatistician, who was blinded to the process of the research, generated a random number sequence using a simple random number table. The numbers were written on cards and all the random number cards were sealed in envelopes. When a patient was enrolled, he/she was allocated to one of two groups, labelled A or B, by a research nurse based on whether the number was odd or even. The blinded codes were released after the data analysis stage. Patients were randomly assigned to either the trial or control group.
Interventions
Patients randomized to the trial group received an intra-operative intra-articular injection of magnesium sulphate 50 mg/kg and ropivacaine 190 mg, mixed with sterile normal saline solution to make up a combined volume of 100 ml, and patients randomized to the control group received an intra-operative intra-articular injection of the same volume of normal saline solution.
Surgery was performed under spinal anaesthesia through the medial parapatellar approach. In both groups the intra-articular injections were in the same areas. The first 30 ml of the analgesic injection mixture or normal saline was injected into the posterior aspect of the capsule and the medial lateral collateral ligaments, just prior to implantation of the component. Care was taken to avoid excessive infiltration in the area of the common peroneal nerve. Before closure of the incision, the quadriceps tendon, the patella tendon and peripatella tissue were infiltrated with another 30-ml injection. The remaining 40 ml was used to infiltrate the synovium, fat pad, articular capsule, retinacular tissues, periosteum, subcuticular tissues and the subcutaneous tissues around the incision. The surgical incision was pressure dressed without drainage. The duration of surgery for each patient was recorded. Both surgeons and patients were blinded to the injection administered.
All patients in both groups received PCA (25 mg/100 ml morphine: 0.5 mg bolus, 6 min lock-out, 5 mg/h maximum rate) for 48 h after surgery. If the pain remained intolerable, 5 – 10 mg morphine was injected intramuscularly. All patients were encouraged to perform active straight leg raises (SLR) after the anaesthetic stage, but a continuous passive motion machine was not used.
Outcome Measures and Follow-Up
An evaluator, who did not participate in the study but who was familiar with the assessment tools, was responsible for collecting the data. This evaluator and the patients were blinded to study treatment during the surgical procedure.
The patient's total morphine consumption was measured as the primary outcome, and pain measured using a visual analogue scale (VAS) was the secondary outcome measure. Morphine consumption was calculated at the different time points during the 48-h post-operative period by adding the amount administered by PCA to any intramuscular injection that was administered. A VAS, ranging from 0 mm (no pain) to 100 mm (severest pain imaginable), was used to assess pre- and post-operative pain at rest and during motion. Post-operative pain was assessed by the VAS at rest at 2, 4, 6, 10, 24 and 36 h and 2, 7 and 15 days post-operatively, and during motion at 24 and 36 h and 2, 7 and 15 days post-operatively.
The times to be able to undertake a SLR and to be able to reach a 90° knee flexion on post-operative days 1, 3, 7, 15 and 90 were recorded. Blood pressure, heart rate and adverse reactions including nausea and vomiting, urinary retention, respiratory depression and rash were also recorded. All patients were followed-up at post-operative months 1, 3, and 6 to determine if there was any delayed infection at the surgical site.
Statistical Analysis
Data analyses were performed using the SPSS® statistical package, version 10.0 (SPSS Inc., Chicago, IL, USA) for Windows®. An intention-to-treat analysis (ITT) was carried out using the last observation carried forward method for imputing missing values. The Student's t-test and the χ 2 test were used to compare continuous and categorical variables, respectively, between the two groups. Continuous variables are presented as mean ± SD and categorical variables are given as the frequency and percentage. A P-value < 0.05 was considered to be statistically significant. A post-hoc power analysis was performed with PASS® 2005 software (NCSS, Kaysville, UT, USA), based on the non-significant observed results for the parameter estimates.
Results
Out of the 169 prospective subjects who responded to the research notice, 143 were assessed for eligibility and 81 met the study entry criteria and began trial intervention with study drug or saline placebo control. Compliance was good as most outcome measures were short-term and good communication was maintained with all patients. Fig. 1 is a detailed flow chart summarizing the study recruitment process and flow of patients through the study. The two groups were followed-up for 6 – 8 months with a mean of 7 months. At the time of writing, except for one patient in the control group who was lost to follow-up at 6 months post-operation, the remaining patients are still being followed-up.
The Consolidated Standards of Reporting Trials (CONSORT) flowchart of subject screening, enrolment, randomization and progress through the clinical trial
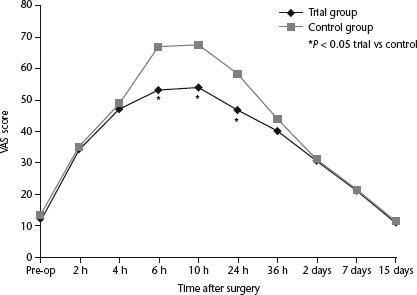
The patient characteristics in both groups were comparable with respect to age, gender ratio, body mass index, VAS and duration of surgery (Table 1). Overall, there was a 28% reduction in morphine consumption in the trial group over the entire 48-h post-operative period compared with the control group (P < 0.05) (Fig. 2). Consumption of morphine at post-operative times 0 – 12 h and 12 – 24 h was also significantly lower in the trial group than in the control group (P < 0.05) (Fig. 2). There was no significant difference in the consumption of morphine at post-operative times 24 – 36 h and 36 – 48 h between the two groups. The post-operative VAS scores at rest were significantly lower at 6, 10 and 24 h in the trial group compared with the control group (P < 0.05) (Fig. 3), although the differences between the two groups were not significant at 2, 4 and 36 h and on days 2, 7 and 15. The post-operative VAS score during motion was significantly lower in the trial group at 24 h than in the control group (P < 0.05) (Fig. 4), but all other differences between the two groups were not significant. The times to being able to perform SLR and to reach a 90° knee flexion were significantly shorter in the trial group than the control group (P < 0.0001 for both parameters) (Table 2).
Post-operative (0 – 48 h) consumption of morphine (mg) by patients who underwent total knee arthroplasty during which they received either an intra-articular injection of magnesium sulphate 50 mg/kg and ropivacaine 190 mg (trial group) or saline (control group) Pain scores at rest during the post-operative period as recorded on a 0 – 100 mm visual analogue scale (VAS) by patients who underwent total knee arthroplasty during which they received either an intra-articular injection of magnesium sulphate 50 mg/kg and ropivacaine 190 mg (trial group) or saline (control group) Pain scores during motion as recorded on a visual analogue scale (VAS) during the post-operative period by patients who underwent total knee arthroplasty during which they received either an intra-articular injection of magnesium sulphate 50 mg/kg and ropivacaine 190 mg (trial group) or saline (control group)

Demographic and duration of surgery data for patients who underwent total knee arthroplasty during which they either received either an intra-articular injection of magnesium sulphate 50 mg/kg and ropivacaine 190 mg (trial group) or saline (control group)
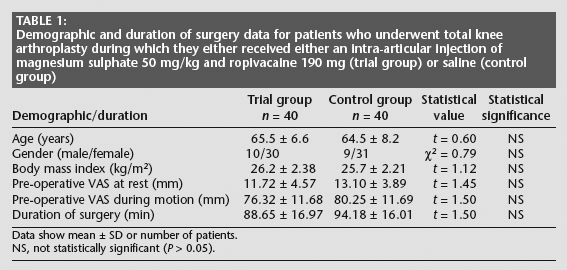
Data show mean ± SD or number of patients.
NS, not statistically significant (P > 0.05).
Clinical parameters after total knee arthroplasty during which patients received either an intra-articular injection of magnesium sulphate 50 mg/kg and ropivacaine 190 mg (trial group) or saline (control group)

Data show mean ± SD.
There were no significant differences in blood pressure, heart rate, urinary retention, respiratory depression, or rash between the two groups following TKA (Table 3). There were 12 patients (30%) in the trial group and 18 (45%) patients in the control group who experienced nausea and vomiting (not statistically significant). No incision infection from the local injection occurred in any patient and no signs of deep infection were observed at the latest follow-up. No sedation was reported in either the trial or control groups during the course of the study. No side-effects relating to local administration of magnesium sulphate and ropivacaine mixture were noted.
Vital signs and adverse effects after total knee arthroplasty during which patients received either an intra-articular injection of magnesium sulphate 50 mg/kg and ropivacaine 190 mg (trial group) or saline (control group)

Data show mean ± SD or number and percentage of patients.
NS, not statistically significant (P > 0.05).
Discussion
The results of the present study demonstrated that intra-operative, intra-articular infiltration with a mixture of magnesium sulphate and ropivacaine reduced the postoperative consumption of morphine compared with an equivalent injection of saline alone. Cumulative morphine consumption was significantly lower in the trial group for the post-operative period 0 – 24 h, leading to a 27% reduction over the entire 48-h post-operative period as compared with the control group (P < 0.05). The incidence of adverse effects, such as nausea, was low in the trial group and no sedation was reported. No side-effects relating to local administration of magnesium sulphate and ropivacaine mixture were noted.
The magnesium ion was first identified as an inhibitor of N-methyl-D-aspartate (NMDA) receptors in the spinal cord, and intravenous magnesium sulphate alters pain processing and reduces the induction and maintenance of central sensitization by non-competitively blocking the NMDA receptors in the spinal cord. 10 The mechanism of the peripheral anti-nociceptive effect of NMDA antagonism has not been precisely defined; it has been hypothesized to occur through an analgesic and anti-inflammatory effect. 11 NMDA antagonists reduce the excitability of nociceptive input terminals of C-fibres that play a role in the central processing of pain.12,13 Magnesium has been shown to reduce post-operative analgesic requirements14,15 and intra-operative intravenous magnesium sulphate at very high doses has been reported to reduce postoperative morphine consumption, but not post-operative pain scores.16,17 It was reported that intravenous magnesium (3.0 – 3.5 g) can be a useful adjuvant in intra-operative analgesia, 18 but the exact dose needs to be determined. Since the magnesium ion poorly crosses the blood– brain barrier in humans, 19 it is not clear whether the therapeutic effect is related to NMDA antagonism in the central nervous system (CNS). In addition to in vitro electrophysiological studies showing the NMDA antagonist properties of magnesium ion in the CNS, 20 intrathecal magnesium sulphate has been shown to potentiate morphine antinociception in a postoperative pain model. 14 It has also been shown that co-administration of magnesium sulphate with ropivacaine for post-operative infiltration analgesia after radical retropubic prostatectomy produces a significant reduction in tramadol requirement. 21 Elsharnouby et al. 22 reported that magnesium sulphate combined with bupivacaine produced a reduction in postoperative pain, when given intra-articularly, in comparison with either bupivacaine or magnesium alone, or with saline placebo.
Magnesium sulphate can provideanalgesia when administered intra-articularly for arthroscopic surgery. 22 In addition, NMDA receptors are present in the peripheral terminal of articular primary afferent fibres in the knee joint and on cellular elements within the joint, such as synoviocytes and immune cells, 11 and their activation has been found potentially to play a role in nociception. 23 Considering this evidence, the authors of the present study were interested to investigate whether magnesium sulphate had beneficial effects in TKA, one of the most painful surgical procedures in orthopaedics.
The analgesic effect of intra-articular magnesium sulphate and ropivacaine mixture was evident. It seems likely that a local effect was at least partially responsible for this as the side-effects usually seen after systemic administration of magnesium sulphate were not observed. Based on the present study, it was shown that low concentrations of intra-articular magnesium and ropivacaine provided efficient pain relief compared with intra-articular saline injections alone.
In conclusion, an intra-operative intra-articular injection of magnesium sulphate and ropivacaine during TKA can reduce the post-operative morphine analgesic requirements with no adverse events. These results demonstrate that a magnesium sulphate and ropivacaine mixture could be used as an adjuvant therapy for postoperative analgesic management.
Footnotes
The authors had no conflicts of interest to declare in relation to this article.
