Abstract
Objective:
Nasal congestion is a common symptom in allergic and nonallergic rhinitis, rhinosinusitis and nasal polyposis. The present study evaluated the clinical effectiveness of Narivent®, an osmotically-acting medical device with anti -oedematous and anti-inflammatory effects, in nasal congestion.
Methods:
A single-centre, prospective study with a pre- post design and consecutive patient enrolment was conducted in an Italian otolaryngology department. Patients with persistent nasal congestion were allocated to treatment groups as follows: group 1 (n = 36) treated for 7 days; group 2 (n = 56) treated for 30 days. In each group, patients received two puffs of Narivent® into each nostril twice daily. Symptom severity was assessed subjectively on a 0 - 10 visual analogue scale, and objectively by the presence/absence of signs and symptoms. Differences in subjective and objective severity measures before and after treatment were compared using Wilcoxon's signed rank test.
Results:
All symptoms and objective scores improved after treatment with Narivent® for 7 or 30 days and no adverse effects were reported by the patients in either group.
Conclusion:
Narivent® appears to be efficacious in treating nasal congestion over a 7- or a 30-day period.
Keywords
Introduction
Nasal congestion, a feeling of blockage, fullness or obstruction of the nasal cavity with consequent restricted airflow, is the major symptom of common upper respiratory tract disorders including allergic rhinitis, acute and chronic rhinosinusitis and nasal polyposis.1–4 Other primary symptoms of these conditions are reduction or loss of smell, rhinorrhoea, facial pain or pressure, and headache. Congestion can also be associated with other rhinological conditions, such as nonallergic rhinitis, viral or bacterial rhinitis and vasomotor rhinitis.1–4 Reversible nasal congestion is usually caused by mucosal inflammation and secretions, while obstruction (a term often used synonymously with congestion) usually refers to irreversible blockage and to fixed or relatively constant congestion. 2 Causes of nasal congestion include occlusion (e.g. nasal polyps), anatomical variation (e.g. septal deformity or turbinate hypertrophy) or, more rarely, neoplasm. 2 The condition has a complex pathophysiology that involves neural, vascular and inflammatory elements. 5 It is associated with inflammation of the nasal epithelium and the production of inflammatory mediators that cause dilatation of nasal blood vessels.6,7 Mucosal inflammation, therefore, underlies many of the specific and interrelated factors that contribute to nasal congestion and other symptoms of allergic rhinitis and rhinosinusitis. 8
The complaint of a blocked nose is often a complex clinical problem involving mucosal, structural and subjective or individual factors. 9 In clinical practice, it is frequently difficult to assess the relative importance of individual factors contributing to nasal obstruction and to decide on the most beneficial therapy for the restoration of satisfactory nasal breathing. Perception of nasal airflow is a subjective sensation and, by definition, difficult to quantify unless it is nearly complete. 10 Inconsistencies between subjective nasal obstruction and the appearance of the nasal cavities is common, and there is controversy about the relationship between subjective assessments of nasal obstruction and objective measurement of nasal airway obstruction using rhinomanometry. 11 Improvements in the objective measurement of nasal patency are continually being sought. 10
Physical examination by anterior rhinoscopy is of limited use in the evaluation of the entire nasal cavity, and complete and thorough examination using nasal endoscopy is therefore advocated in chronic rhinological diseases.12,13 The Lund–Mackay system of scoring nasal endoscopy findings is the only system based on mucosal thickening (oedema).14–16
Subjective changes in perceived congestion severity can be assessed using a visual analogue scale (VAS) or one of a number of symptom-scoring systems including ordinal scales and questionnaires such as the RhinoSinusitis Outcome Measurement, the SinoNasal Assessment Questionnaire, the Congestion Quantifier five-item test for nasal congestion and the SinoNasal Outcome Test-22. 9 Therapeutic intervention is aimed at relieving subjective complaints and, as such, measurement of subjective parameters is necessary. 14 The VAS offers a reproducible, quantifiable evaluation of patients' symptoms, which may provide more subtle information than simply asking whether the patient is better, the same, or worse.17,18
Although various pharmacological options exist, no agent has been proven to be completely efficacious, and there is a paucity of data supporting commonly used symptomatic therapies. 1 Narivent® (DMG Italia Srl, Pomezia, Italy) is a medical device dispensing a nasal lubricant containing eucalyptol, glycyrrhizin and mannitol that acts osmotically and has antioedematous and anti-inflammatory actions.19,20
The present study was conducted in order to evaluate the short- (7 days) and longer-term (30 days) safety and clinical effectiveness of Narivent® on subjective and objective symptoms in patients with persistent nasal congestion. Measurement of symptom severity from the patient's perspective was by VAS. 21
Patients and methods
Study Population
Male and female patients with persistent nasal congestion caused by allergic or nonallergic rhinitis, turbinate hypertrophy, sinus nonocclusive polyposis or vasomotor rhinopathy presenting at the Ear, Nose and Throat (ENT) Department of the San Giovanni Addolorata Hospital, Rome, Italy were consecutively enrolled in the present study between January and June 2011. Patients were excluded if they had: a diagnosis of cystic fibrosis; the presence of asthma episodes in the 30 days preceding the study; any acute upper respiratory tract infections; the presence of massive occlusive polyps in the sinus; septal deviations; used nasal or oral corticosteroids or decongestants during the 4 weeks preceding the study; or used antileukotrienes or antihistamines during the previous week.
At enrolment, patients provided verbal and written informed consent to participate in the study. The study was conducted in compliance with the ethics requirements of the San Giovanni Addolorata Hospital Institutional Review Board.
Study Design
This was a single-centre, prospective study with a pre–post design. Patients were divided into two groups depending on whether treatment was acute or chronic. Patients in group 1 were assessed by the investigators (V.D., A.C. and C.V.) at enrolment (day 0) and after 7 days' treatment and those in group 2 were assessed by the investigators at enrolment (day 0) and after 30 days' treatment. In both groups, patients self-administered two puffs of Narivent® into each nostril twice daily for the duration of the treatment period.
Study Assessments
A physical examination was conducted at each visit by complete ENT endoscopy. Turbinate hypertrophy was classified according to the investigator's personal experience as: absent, good (turbinates obstructing one-third of the nasal fossae), fair (turbinates obstructing two-thirds of the nasal fossae), or poor (turbinates completely obstructing the nasal fossae). Septal deviation (assessed at pretreatment only) was classified according to the investigator's personal experience as: absent, good (septum slightly deviated from baseline), fair (septum markedly deviated from baseline), or poor (obstructing septum). The Lund– Mackay system was used to classify nasal polyps (assessed at pretreatment only).15,16 Adenoid hyper trophy was classified as: absent, good (slightly enlarged adenoids), fair (adenoids enlarged but not beyond tubal ostium), or poor (adenoids enlarged beyond tubal ostium).22,23 The nasal mucosa was described as either normal, hyperaemic, pallid/livid or atrophic, and nasal secretions were described as either absent, haematic/purulent, pallid/serous, or mucous.
At each visit a VAS was used to quantify subjective feelings of nasal obstruction, itching and dryness. 18 The subjective symptom score was obtained using a modified VAS. 24 Patients rated the perceived degree of obstruction on a scale of 0 (complete patency) to 10 (complete stenosis). A subjective evaluation was also performed for other symptoms including cephalea, rhinorrhoea, rhinopharyngeal exudates, pain and hyposmia. In addition, treatment compliance and palatability of Narivent® according to patient opinion were measured. Patient-reported adverse events were recorded.
Statistical Analyses
The primary outcomes in group 1 (short-term, 7-days' treatment) and group 2 (long-term, 30 days' treatment) were symptom resolution (improvement in each symptom between enrolment and day 7 or enrolment and day 30) and improvement in overall symptom burden (as measured by the overall VAS score). Sample size was computed on the basis of a type I error of 0.05 and a power of 0.80. At this error level, 34 and 51 patients were needed in groups 1 and 2, respectively, to detect a significant post-treatment change in the VAS score of 2 points (± SD 3 in group 1 and ± SD 4.5 in group 2). Assuming a drop-out rate of 5% with short-term treatment and 10% with long-term treatment, 36 patients and 56 patients were estimated to be required in groups 1 and 2, respectively.
All continuous variables were expressed as median and interquartile range and categorical variables as percentages and absolute numbers. Differences between symptoms experienced before and after treatment with Narivent® were compared using the paired-sample Wilcoxon's signed rank test. Tests were performed using the R system. 25 A P-value of < 0.05 was considered to be statistically significant.
Results
A total of 36 patients were enrolled in group 1 (7 day follow-up) and 56 patients were enrolled in group 2 (30 day follow-up). The baseline demographic and disease characteristics of patients in both treatment groups are shown in Table 1. One patient in each treatment group dropped out from the study due, in each case, to paradoxical nasal congestion. Analyses were, therefore, performed on 35 patients in group 1 and 55 patients in group 2.
Pretreatment (baseline) demographic and disease characteristics of the patients with nasal congestion who were allocated to receive short-term (7 days) or long-term (30 days) treatment with Narivent®
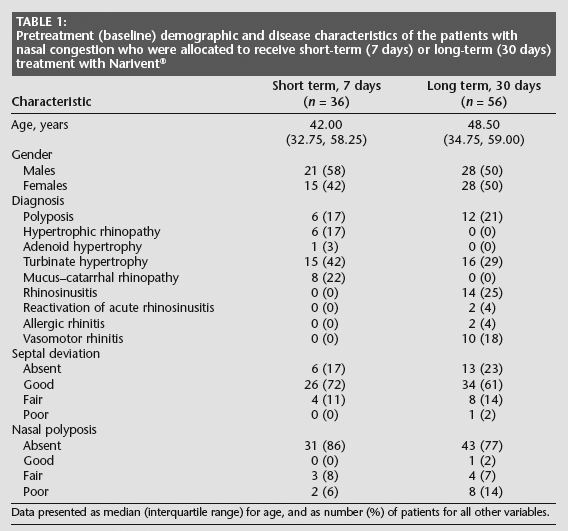
Data presented as median (interquartile range) for age, and as number (%) of patients for all other variables.
Table 2 shows the subjective evaluation of the symptoms of nasal congestion before and after 7 days' treatment with Narivent®. All symptom evaluations were improved following treatment although the effect was statistically significant only for nasal congestion and cephalea (P < 0.001 for both symptoms). The overall symptom burden, as indicated by the VAS score, showed significant improvement after treatment (P < 0.001, Table 2). Physical examination revealed significant improvements in turbinate hypertrophy (P < 0.001), and in normal and hyperaemic mucosal status (P < 0.001 for both) after 7 days of treatment with Narivent® (Table 3). Significantly more patients were also observed with an absence of nasal secretions (P = 0.006). At the end of the 7-day treatment period, the palatability of Narivent® was considered good in 83% (29/35) of patients, fair in 11% (4/35) and poor in 6% (2/35). No patient reported unsatisfactory palatability. Compliance was high in 83% (29/35) of patients, fair in 11% (4/35) and poor in 6% (2/35). No adverse events were reported by any patients after 7 days of treatment.
Visual analogue scale (VAS) scores and subjective symptom evaluation in patients with nasal congestion before and after 7 days' treatment with Narivent®

Data presented as median (interquartile range) for VAS score and as number (%) of patients for all other variables.
One patient dropped out due to paradoxical nasal congestion and so was not included in the after Narivent® analysis.
Between-group comparison using Wilcoxon's signed rank test.
Patients rated the perceived degree of obstruction on a VAS of 0 (complete patency) to 10 (complete stenosis).
NS, not statistically significant (P > 0.05).
Objective observations on physical examination of patients with nasal congestion before and after 7 days' treatment with Narivent®
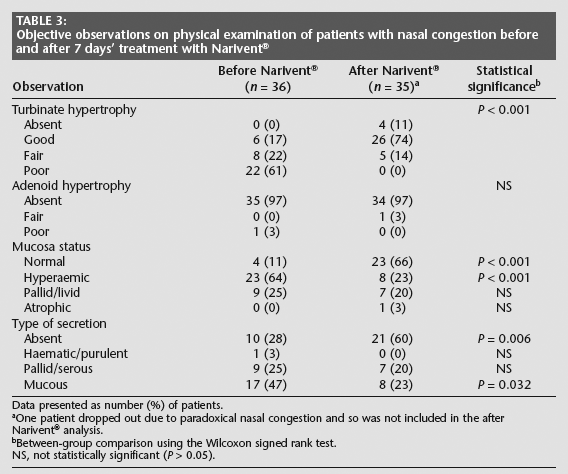
Data presented as number (%) of patients.
One patient dropped out due to paradoxical nasal congestion and so was not included in the after Narivent® analysis.
Between-group comparison using the Wilcoxon signed rank test.
NS, not statistically significant (P > 0.05).
Table 4 shows the subjective evaluation of the symptoms of nasal congestion before and at the end of longer-term (30 days) Narivent® treatment. Nasal congestion, cephalea and rhinorrhoea were all significantly decreased (P < 0.001) after treatment, and a significant improvement in hyposmia was also observed (P < 0.001). The overall symptom burden, as indicated by the VAS score, was also significantly improved at the end of the 30 days' treatment (P < 0.001, Table 4). There were significant differences in several physical examination findings following treatment with Narivent® for 30 days (Table 5): turbinate hypertrophy and the overall condition of the mucosa were improved (P < 0.001), and a decrease in nasal secretions was also recorded (P < 0.001). The palatability of Narivent® following 30 days of treatment was considered good by 73% (40/55) of patients and fair by 27% (15/55). No patient reported poor or unsatisfactory palatability. Compliance was high in 75% (41/55) of patients, fair in 20% (11/55) and poor in 5% (3/55). No adverse effects were reported by patients receiving the longer-term (30-day) Narivent® treatment.
Visual analogue scale (VAS) scores and subjective symptom evaluation in patients with nasal congestion before and after 30 days' treatment with Narivent®

Data presented as median (interquartile range) for overall VAS and as number (%) of patients for all other variables.
One patient dropped out due to paradoxical nasal congestion and so was not included in the after Narivent® analysis.
Between-group comparison using Wilcoxon's signed rank test.
Patients rated the perceived degree of obstruction on a VAS of 0 (complete patency) to 10 (complete stenosis).
NS, not statistically significant (P > 0.05).
Objective observations on physical examination of patients with nasal congestion before and after 30 days' treatment with Narivent®
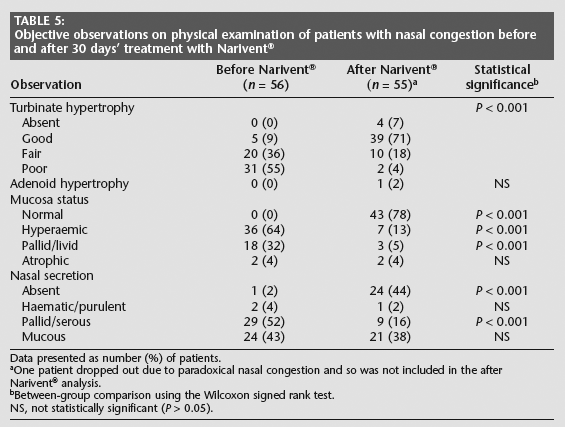
Data presented as number (%) of patients.
One patient dropped out due to paradoxical nasal congestion and so was not included in the after Narivent® analysis.
Between-group comparison using the Wilcoxon signed rank test.
NS, not statistically significant (P > 0.05).
Discussion
Nasal congestion is one of the most common complaints dealt with in otorhino -laryngology. Of the pathologies responsible for general complete and continuous or occasional nasal obstruction, specific and nonspecific vasomotor rhinitis are those with the greatest epidemiological impact. 26 The pervasiveness of allergic rhinitis and rhinosinusitis has caused congestion to become a highly prevalent problem; however, it is important to note that the perception of congestion in chronic rhinosinusitis can also be caused by polyps extruding into the nasal airway, producing a physical obstruction in the nostril. 8 Patients diagnosed with these upper respiratory conditions identify congestion as the most common and typically the most troublesome symptom. 2
The negative effects of nasal congestion are far-reaching and can affect the physical and emotional functioning of patients. Nasal congestion can affect quality of life, work/school productivity and the ability to perform daily activities. 27 Furthermore, chronic nasal congestion can cause sleep-disordered breathing, promoting daytime sleepiness and fatigue2,28,29 which leads to decreased alertness, increased accident rates, reduced work efficiency, and irritability and depression.30–32 Taking into account the high prevalence of nasal congestion and the associated social and economic burden, relief of this symptom should be a key consideration in the treatment of patients with rhinological disease. 2
Mucosal inflammation is the central pathophysiological mechanism leading to nasal congestion in common upper respiratory diseases. A wide range of cell types and biologically active agents (e.g. histamine, tumour necrosis factor-α, interleukins and cell adhesion molecules) contribute to inflammation, which can manifest as venous engorgement, increased nasal secretions and tissue swelling/oedema that, ultimately, lead to impaired airflow and the sensation of nasal congestion.7,8 Treatments for the relief of nasal congestion include environmental control measures, where possible, and medical therapy and surgical intervention for severe or persistent cases of nasal obstruction. 1 The development of pharmacological therapies for congestion in upper respiratory diseases has been guided by the need to oppose vasodilation thus reducing nasal airway resistance and facilitating nasal breathing.8,26 Standard conservative treatment for chronic conditions, such as chronic rhinosinusitis, with or without nasal polyposis, is based on the use of short- or long-term antibiotics and topical steroids with the addition of decongestants – mostly in a short-term regimen and for the acute attack itself. 3 The most extensively evaluated therapies for nasal congestion include antihistamines, decongestants, leukotriene receptor antagonists and intranasal corticosteroids. 1
Intranasal corticosteroids have potent and broad anti-inflammatory activities. In controlled clinical trials, they have been shown to be more effective than other classes of agents for the relief of congestion. They do not, however, reduce mean nasal congestion scores to normal levels or effectively reduce congestion in every patient. 1 Decongestants are sympathomimetic drugs, employed as systemic or topical products, which act by constricting capacitance vessels in the turbinates to produce a decrease in subjective symptoms and nasal airway resistance.6,26 Frequently reported decongestant side effects in patients with chronic nasal congestion include systemic effects such as elevated blood pressure, tachycardia, palpitations, restlessness, insomnia, anxiety, tremors and hypersensitivity reactions; side effects of topical agents include burning, stinging, sneezing and local irritation.1,6,26,33,34 The side-effect profile of topical and oral decongestants limits their usefulness in allergic rhinitis, and there is limited evidence to support their utility for the relief of congestion associated with nonallergic/ vasomotor rhinitis, rhinosinusitis or nasal polyposis. 1
Numerous preparations have been investigated for the treatment of symptoms associated with rhinitis, rhinosinusitis or nasal polyposis, but substantial evidence for their benefit is lacking. These medications include isotonic/hypertonic saline as a nasal douche, antihistamines (in allergic conditions), antimycotics, mucolytic agents/ phytomedical preparations, immuno -modulators/immunostimulants and bacterial lysate preparations. 3
As a consequence of the many adverse effects related to standard therapies and long-term treatments and the paucity of evidence for the efficacy of symptomatic therapy, there is a growing need for alternative or coadjuvant treatments. Narivent® is a medical device dispensing a nasal lubricant that has antioedematous and anti-inflammatory actions due to the components glycyrrhizin and mannitol.19,20 The present study was conducted in order to verify whether treatment with Narivent® was effective in reducing nasal obstruction and other symptoms associated with chronic rhinosinusitis, allergic rhinitis, turbinate hypertrophy, sinus nonocclusive polyposis or vasomotor rhinopathy. The patients' perception of nasal symptoms and objective features of nasal obstruction were both assessed.
The results of the present study demonstrated significant improvement in the symptoms of nasal congestion after treatment with Narivent® for 7 or 30 days. Furthermore, the data indicated that the action of Narivent® was not limited to promotion of a subjective sensation of increased nasal air flow, but corresponded to an objective reduction in nasal resistance. Narivent® given for 7 or 30 days reduced the main subjective symptoms, including the sensation of nasal congestion and cephalea. Treatment for 30 days also reduced rhinorrhoea and improved impaired sense of smell (hyposmia). The patients' overall subjective assessment of the sensation of nasal obstruction, recorded using a VAS score, also showed a significant reduction after the 7- or 30-day treatment periods. Physical examination of patients treated with Narivent® demonstrated improvement in the patients' condition with the most positive post-treatment changes observed in mucosa status, turbinate hypertrophy and reduction of nasal secretion. No adverse events were reported by patients during the 7- or 30-day treatment periods and compliance with the use of the product was generally assessed as high.
In conclusion, the present study has provided evidence that Narivent® is effective in improving subjective and objective nasal symptoms, both in the short (7 days) and longer term (30 days), in patients with nasal turbinate hypertrophy, allergic rhinitis or nonspecific vasomotor rhinitis.
Footnotes
Acknowledgement
We thank DMG Italia Srl for providing Narivent® for use in this study.
AS Scirè and G Morpurgo have ongoing contracts with DMG Italia Srl for conducting clinical studies. The other authors had no conflicts of interest in relation to this article.
