Abstract
Objective:
This study investigated bilateral uterine artery chemo -embolization (BUACE) combined with dilation and curettage (D&C) in caesarean scar pregnancy (CSP).
Methods:
Nineteen women with CSP were referred for interventional radiology. In 13 patients, BUACE was performed before D&C, following a diagnosis of CSP. A further six patients received BUACE for massive vaginal bleeding after D&C for inevitable miscarriage; the diagnosis of CSP was subsequently confirmed ultrasonographically. BUACE of the uterine arteries was performed using gelfoam particles following intra-arterial infusion of 100 mg/m2 methotrexate.
Results:
BUACE was technically successful and immediate haemostasis was achieved in all patients. Blood loss was significantly greater during D&C undertaken before BUACE compared with D&C after BUACE, but this bleeding was controlled by BUACE. No patient required further surgical intervention and there were no severe complications. The gestational sac and placenta could no longer be detected ultrasonographically and the menstrual cycle returned to normal 2 - 3 months after treatment in all patients.
Conclusions:
BUACE followed by D&C seems to be a safe and effective treatment for CSP and should be considered as a treatment of choice.
Introduction
Caesarean scar pregnancy (CSP) is the rarest form of ectopic pregnancy and is associated with life-threatening complications such as massive haemorrhage and uterine rupture during the proceeding gestation. 1 CSP is an aggressive pathological condition in which there is early direct invasion of the myometrium by trophoblastic cells (Fig. 1A). 2 It is associated with spontaneous abortion and preterm delivery, 3 but the most significant complication is an abnormally adherent placenta (Fig. 1B), which may lead to life-threatening haemorrhage.4,5 A delay in either diagnosis or treatment can lead to uterine rupture, with the inevitable loss of fertility and possible significant long-term adverse effects on the woman's health and health-related quality of life, 6 or even maternal morbidity.1,7 Available data suggest that termination of the CSP soon after diagnosis is the treatment of choice in the first trimester, as expectant treatment has a poor prognosis because of the risk of uterine rupture. 8
Schematic diagram showing (A) the decidual defect at the site of the caesarean section scar and trophoblastic cells directly invading the myometrium, and (B) the gestational sac with placenta accreta located at the site of the caesarean section scar in the lower uterine segment
A variety of surgical and nonsurgical interventions have been proposed for terminating a CSP while preserving fertility. Suction curettage and excision of the CSP are associated with profuse bleeding. 9 Nonsurgical treatment, such as intralesional or systemic administration of methotrexate (MTX) which acts by inhibiting the metabolism of folic acid, is associated with longer hospitalization times and higher costs. 10 Interventions for CSP that result in massive haemorrhage may necessitate the use of bilateral uterine artery embolization. Currently, no treatment modality for the management of CSP appears to be entirely reliable and none can guarantee uterine integrity. 11 The present study aimed to present our experiences in managing CSP using bilateral uterine artery chemoembolization (BUACE) with biodegradable gelfoam particles and local administration of MTX.
Patients and methods
Patients
Chinese women with CSP referred to the Department of Interventional Radiology, Renji Hospital, Shanghai JiaoTong University Medical School, Shanghai, China, for superselective BUACE were recruited sequentially to the study between October 2009 and October 2011. For analysis, patients were divided into two groups: patients in group A were diagnosed with CSP and underwent BUACE before dilation and curettage (D&C), and those in group B underwent D&C for inevitable miscarriage and were then referred for BUACE due to massive vaginal bleeding. The diagnosis of CSP in patients in group B was subsequently confirmed by experienced ultra -sonographers. Patients who were allergic to contrast agent or who refused BUACE were excluded from the study.
The study protocol was approved by the Ethical Committee of the Shanghai JiaoTong University Medical School, Shanghai, China, and all patients provided written informed consent before beginning treatment.
Diagnosis of CSP
The clinical diagnosis of CSP was made on the basis of the following criteria: (i) a history of caesarean section; (ii) amenorrhoea with or without irregular vaginal bleeding; (iii) elevated serum β-human chorionic gonadotrophin (β-hCG) concentrations; (iv) ultrasonographic confirmation of CSP. Ultrasonographic confirmation included demonstration of an empty uterine cavity, development of the gestational sac in the anterior part of the isthmic portion, the absence of healthy myometrium between the bladder and the sac, 12 and high-velocity, low-impedance peritrophoblastic flow. 4
Buace and D&C Procedures
The BUACE procedure was performed on patients under local anaesthesia using 1% lidocaine injected into the groin area. Digital subtraction angiography (DSA) of both internal iliac arteries was performed by interventional radiologists (X.-B.Z. and J.-C.C.) using a right-sided femoral approach. A 4F cobra-shaped catheter (Cordis Corp., Miami Lakes, FL, USA) and 0.035-inch diameter hydrophilic polymer-coated guide wire (Radiofocus® Terumo Corp., Tokyo, Japan) were used for uterine artery catheterization. After the tip of the catheter had been placed into the uterine artery, DSA was performed using 10 ml of the contrast agent iopamidol (370 mg iodine/ml) at an inflow rate of 3 ml/s. MTX was then infused into each uterine artery, the total dose of 100 mg/m2 being divided equally between the two arteries. Absorbable gelfoam particles (Alicon Medical Co. Ltd, Hangzhou, China) 1000 – 1400 μm in diameter, mixed with iopamidol at a concentration of approximately 5 mg/ml, were then immediately injected into each uterine artery until the main uterine artery was blocked as demonstrated by stasis of the contrast agent. The presence of ovarian artery anastomoses was not a contraindication to the procedure. Repeat angiography of the internal iliac arteries was then performed to evaluate the effects of embolization. After the procedure, patients were admitted to the gynaecology ward for postprocedural care.
In group A, ultrasound-guided D&C was performed 2 days after BUACE by a senior attending physician (Y.-C.Z., A.-M.Z. or W. D.) . In group B, D&C had been performed before BUACE.
Follow-Up
A successful outcome was defined as complete recovery with no severe complications and the preservation of fertility. The success rate, postprocedural complications, volume of blood loss during D&C, time for β-hCG concentrations to fall to normal values (< 25 mIU/ml), time for recovery of the menstrual cycle, duration of hospital stay and hospital costs were recorded.
Serum β-hCG concentrations were measured in all patients every other day for 2 weeks and then at 7-day intervals until they decreased to normal values (< 25 mIU/ml). Routine ultrasonography screening was performed to detect pregnancy remnants implanted in the caesarean section scar within 2 weeks of BUACE treatment and again at 1 – 3-month intervals until the remnants could no longer be seen.
Statistical Analyses
The Mann–Whitney U-test was used to analyse differences in continuous variables between the two groups. A P-value of < 0.05 was considered to be statistically significant. All statistical analyses were performed using SPSS® software version 18.0 (IBM, Chicago, IL, USA).
Results
A total of 19 patients with CSP were recruited to the study. Their clinical and demographic characteristics are shown in Table 1. The patients' ages ranged between 23 and 41 years (mean ± SD 33.0 ± 4.9 years; 95% confidence interval [CI] 30.6, 35.4). The interval between caesarean section and hospital admission ranged between 11 and 159 months (mean ± SD 56.2 ± 40.4 months; 95% CI 36.1, 76.3). The thickness of the myometrium from the serosa to the gestational sac, as measured by ultrasonography, ranged between 0.7 and 9.5 mm (mean ± SD 3.7 ± 2.3 mm; 95% CI 2.6, 4.8).
Clinical and demographic characteristics of patients with caesarean scar pregnancy (n = 19) who underwent bilateral uterine artery chemoembolization (BUACE) followed by dilation and curettage (D&C) (group A) or D&C before BUACE (group B)
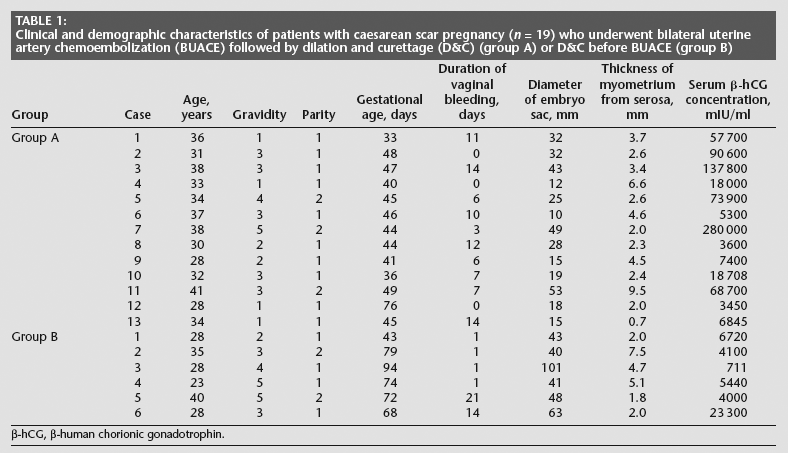
β-hCG, β-human chorionic gonadotropin.
Of the 19 patients recruited, 13 were in group A and six in group B. Ten patients in group A and six patients in group B received primary treatment for bleeding, including vaginal packing and intravenous administration of uterotonic drugs. The six patients in group B also received blood transfusion and fluid resuscitation, but the vaginal bleeding persisted. The other three patients in group A did not receive any primary management as no vaginal bleeding occurred; blood loss during D&C in these patients was minimal (5 – 150 ml).
Bilateral uterine artery chemo -embolization was successfully carried out in all patients and all vaginal bleeding was resolved. No patient required further surgical intervention and the success rate was 100%. The implanted gestational sac and residual placenta in the myometrium gradually shrank and could no longer be detected by ultrasound 2 – 3 months after treatment in all patients in both groups. During follow-up, the mean ± SD time to return of the menstrual cycle in all 19 patients was 1.94 ± 1.31 months. Blood loss during D&C and hospital costs were significantly lower in group A compared with group B (P < 0.002 and P < 0.018, resperctively; Table 2). There were no significant differences between the two groups in the time in hospital, time to return of the menstrual cycle or time for the serum β-hCG concentration to return to normal. In group B, emergency BUACE successfully controlled the profuse bleeding in all patients.
Clinical outcomes of patients with caesarean scar pregnancy (n = 19) who underwent bilateral uterine artery chemoembolization (BUACE) followed by dilation and curettage (D&C) (group A) or D&C before BUACE (group B)

Data presented as mean ± SD.
Statistical analyses were performed using the Mann-Whitney U-test.
β-hCG, β-human chorionic gonadotrophin; NS, no statistically significant between-group differences (P ≥ 0.05).
Digital subtraction angiography showed dilation and distortion of both uterine arteries and their branches in all patients (Figs 2 and 3). A strongly vascularized placental structure was also detected in the arterial phase in all 19 patients. The radiological manifestations of CSP on DSA, including the predominant feeding arteries of the gestational sac, ovarian branches of the uterine arteries (Fig. 3), contrast extravasation (Fig. 3) and supply by the bladder artery (Fig. 2), are summarized in Table 3. Embolization using gelfoam particles was seen mainly to block the uterine arteries from the peripheral branch to the main trunk.
Representative digital subtraction angiograms (A and C) before and (B and D) after embolization of the left and right uterine arteries. (A) Dilated and distorted left uterine artery (UA; white arrow) and gestational sac (GS; black arrow) before embolization; (B) left UA after embolization, showing disappearance of the vascularized placental structure; (C) dilated and distorted right uterine artery (UA; white arrow) and gestational sac (GS; short black arrow) before embolization; the right ovarian branch of the uterine artery is also demonstrated (long black arrow); (D) right UA after embolization, showing disappearance of the vascularized placental structure and the appearance of a slightly enlarged and distorted bladder artery (Bladder a; black arrow) Representative digital subtraction angiograms (A and C) before and (B and D) after embolization of the left and right uterine arteries in a patient diagnosed with a caesarean scar pregnancy (CSP); massive vaginal bleeding during dilation and curettage could not be controlled and ultrasonography demonstrated a CSP. (A) Dilated and distorted right uterine artery (UA; black arrow) before embolization, showing contrast extravasation (short white arrows); (B) right uterine artery after embolization, showing complete embolization of the artery; (C) dilated and distorted left uterine artery (UA; black arrow) with enlarged ovarian branch (long white arrow) before embolization, showing contrast extravasation (short white arrows); (D) left uterine artery after embolization, showing complete embolization of the artery

Digital subtraction angiography findings in patients with caesarean scar pregnancy (n = 19) who underwent bilateral uterine artery chemoembolization (BUACE) followed by dilation and curettage (D&C) (group A) or D&C and then BUACE (group B)

Data presented as number (%) of patients.
Mean serum β-hCG concentrations decreased significantly 5 days after treatment compared with before treatment (P < 0.05; Fig. 4). The time taken for serum β-hCG levels to fall to normal values was similar in both groups (Table 2); serum β-hCG concentrations returned to normal in all patients within 10 – 40 days.
Mean serum concentrations of β-human chorionic gonadotrophin (β-hCG) in patients (n = 19) with caesarean scar pregnancy following treatment with bilateral uterine artery chemoembolization combined with dilation and curettage. *P < 0.05
The most common postprocedural complication was postembolization syndrome, which included fever (19/19; 100%), mild or moderate lower abdominal pain (19/19; 100%), and nausea and vomiting (3/19; 16%). This lasted 1 – 3 days and the pain and fever were successfully controlled using tramadol and/or indomethacin suppositories. No severe complications, such as bladder perforation or massive pulmonary embolization, were observed in any of the patients. None of the patients died as a result of massive bleeding.
The mean ± SD follow-up period was 15.4 ± 5.4 months (range 6 – 24 months). Five women (26%) wanted to become pregnant in the future, but no pregnancies were reported during the follow-up period.
Discussion
Caesarean scar pregnancy is one of the rarest forms of ectopic pregnancy and little is known about its incidence and natural history.8,13 As a result of the rising incidence of caesarean sections worldwide, an increasing number of CSP cases are being diagnosed and reported.8,14 No reliable data are currently available on the risk of recurrence of CSP in future pregnancy or the role of the interval between the previous caesarean delivery and the occurrence of CSP. 8 In the present study, the interval between the previous caesarean section and CSP ranged between 11 and 159 months.
In CSP, the fertilized egg deviates from its normal position and implants in the scar of the anterior wall of the lower uterine segment. After implantation, placenta accreta is likely to develop because of the absence of a decidual interface between the placenta and myometrium. The thickness of the myometrium from the serosa to the gestational sac in the patients in the present study ranged between 0.7 and 9.5 mm, indicating that trophoblastic cells had penetrated the myometrium. The main clinical symptoms associated with CSP are vaginal bleeding, medical abortion without villi, an abnormal uterine wall mass or a continuous increase in serum β-hCG concentrations.
Pathologically, the embryo may penetrate into tiny fissures in the scar and implant within it. Caesarean section injures the endometrium, resulting in scarring and decidual hypoplasia or decidual defects. 15 Trophoblastic cells are then able to invade the myometrium directly. The villi adhere to and implant into the myometrium, and even penetrate through the uterine wall. 5 With the continuing growth of the trophoblastic tissue, the blood supply becomes very rich and irregular, and the bladder artery may become one of the supply arteries of the gestational sac. 16 In the present study, one patient demonstrated bladder artery staining after BUACE. In our opinion, DSA demonstrated in this case that the bladder artery had become a supply artery of the gestational sac, but there was no additional supportive evidence from other imaging modalities. Uterine arteriovenous malformations have been reported in CSP confirmed by the presence of trophoblastic cells in the resected area. 17 Although obvious arteriovenous malformations were not evident in the patients in the present study, the uterine drainage vein, often shown during the embolization procedure, could possibly induce existing minimal arteriovenous shunts. Contrast extravasation, which occurred in eight patients (42.1%) in the present study, may have been caused by villi penetration or damage from curettage.
A delay in diagnosis and/or treatment of CSP can lead to uterine rupture, major haemorrhage, hysterectomy and maternal morbidity. Early diagnosis can offer treatment options that prevent uterine rupture and haemorrhage, thereby preserving the uterus and future fertility. 8 In the present study, the diagnosis of CSP in the six patients in group B was confirmed after they had experienced massive vaginal bleeding causing haemorrhagic shock during D&C. Yang et al. 18 reported that, of 11 patients with CSP who were primarily treated with D&C, eight had severe haemorrhage during the procedure. Only three women were successfully treated without severe complications and with preservation of the uterus; the other eight women were treated with hysterectomy or wedge excision of the uterus. 18 In the present study, all the patients in group B were successfully treated with BUACE and did not require more invasive surgical procedures.
Available data suggest that termination of CSP is the treatment of choice. 8 Expectant treatment is associated with a poor prognosis because of the risk of uterine rupture requiring hysterectomy.8,13 MTX is one of the treatment options for terminating a CSP,19,20 but the disadvantages associated with MTX are lengthy treatment times 10 and slow recovery, together with failure to avoid completely the occurrence of bleeding after treatment because of insufficient involution of the placental mass at the site of the scar.19,21,22 In the present study, MTX was administered by intra-arterial infusion and further injections into the gestational sac were not required. The dose (100 mg/m2) was higher than that previously reported in the literature. 18 Use of a higher dose speeds up the necrosis of the gestational sac, enabling complete resolution to occur before the gelfoam particles are absorbed. The uterine artery was embolized immediately after MTX infusion using gelfoam particles to maintain a relatively high local MTX concentration. Polyvinyl alcohol particles are rarely used in this situation because they result in permanent embolization, which is not suitable for the management of CSP. Embolic agents such as biodegradable beads loaded with MTX may prove to be useful in the future but, to our knowledge, there have been no reports of their use to date.
Uterine artery chemoembolization is a minimally invasive procedure that is likely to reduce the vascularity of the placenta, causing a gradual involution of the pregnancy. 23 Uterine artery chemo -embolization followed by suction curettage may be a priority option for the management of CSP. 10 Persistent bleeding is a strong indication for embolization. In the present study, persistent bleeding in 16 patients with CSP was effectively controlled by BUACE and no-one died as a result of vaginal bleeding. The three patients who had no vaginal bleeding experienced minimal bleeding during D&C. Following treatment to terminate the CSP, serum β-hCG concentrations returned to normal within 10 – 40 days in all patients.
Absorbable gelfoam particles of diameter 1000 – 1400 μm were used as the embolic materials in the present study. The gelfoam particles mainly blocked the dilated bleeding uterine arteries from the peripheral branch to the main trunk, causing mechanical embolization, which can also lead to platelet aggregation and intravascular fibrin deposition, forming blood clots that aid haemostasis. The capillary network is not damaged by gelfoam particles of this diameter and the uterine blood supply can be maintained through other communicating arteries. Gelfoam particles begin to be absorbed within around 2 weeks and are completely absorbed by approximately 3 months later. 24 The uterine artery becomes revascularized and its function is unaffected. Although, in the present study, it was observed that the ovarian branches of the uterine arteries may become embolized by gelfoam particles, menstruation returned to normal in all patients within ∼3 months. No patient presented with secondary amenorrhoea during the follow-up period, an indication that uterine and ovarian functions were unaffected. These findings indicate the suitability of BUACE for the treatment of CSP. D&C following uterine artery embolization quickly removed most of the embryonic tissue and bleeding was not a concern. Serum β-hCG concentrations decreased after both procedures were undertaken because most of the villi had been removed. In our opinion, this therapeutic protocol appears to offer a safe and effective treatment for CSP.
Medical costs were significantly reduced in group A compared with group B, possibly because there was no need for blood transfusion or other medical treatment for haemorrhagic shock in group A. No significant difference was observed, however, in the time spent in hospital between the two groups.
The most common postprocedural complication after BUACE is postembolization syndrome, which usually presents as lower abdominal pain, fever, nausea and vomiting. In the present study, all of the patients experienced postembolization syndrome to some degree, the duration of which was approximately 1 – 3 days. Tramadol and/or indomethacin suppositories effectively controlled the pain and/or fever. Severe complications such as bladder necrosis, infection, soft tissue damage, buttock claudication and pulmonary embolization did not occur in the present study. The absorbable gelfoam particles applied during BUACE were temporary embolic agents that have been shown previously to be effective and nontoxic, and are associated with few complications and a good safety profile. 24
The present study had several limitations. First, the sample size was small because of the low incidence of CSP, which may have resulted in selection bias. Secondly, long-term data on maternal fertility were not available due to the relatively short follow-up period, although normal menstruation returned in all the patients. Thirdly, it was not possible to perform a randomized controlled clinical study of the management of CSP because of the potential danger of bleeding when D&C is performed alone.
In conclusion, BUACE followed by D&C seems to be a safe and effective treatment for CSP and, in our opinion, should be considered as the primary treatment of choice, particularly in the presence of massive uterine haemorrhage. Performing D&C before BUACE is relatively dangerous because of the risk of massive haemorrhage that can occur during uterine curettage. BUACE followed by D&C should help to terminate the CSP, preserve maternal fertility and prevent haemorrhage-related mortality.
Footnotes
Acknowledgement
This study was supported by a grant from the Shanghai Natural Science Foundation (No.10ZR1418900).
The authors had no conflicts of interest to declare in relation to this article.
