Abstract
Objective:
Telomere length was used as a biomarker of cell senescence to explore the role of telomere shortening in photoageing induced by ultraviolet A (UVA) light.
Methods:
Real-time polymerase chain reaction was used to determine telomere length in cultured human fibroblasts of different generations and after exposure to UVA at doses up to 10 000 mJ/cm2. Twoway analysis of variance was used to determine whether passaging or UVA was the main factor contributing to telomere shortening.
Results:
In nonirradiated cells, telomere length was inversely related to cell generation number. In fibroblasts exposed to UVA at a dose of 1000 or 10 000 mJ/cm2, telomere length was significantly shorter than that of nonirradiated controls and was negatively related to UVA dose.
Conclusions:
Telomere length and subsequent cell viability may be affected by UVA irradiation. DNA damage caused by UVA irradiation may initiate the photoageing process and telomeres may be a useful new target for attempts to prevent photoageing.
Keywords
Introduction
Ageing of the skin is regarded as resulting from intrinsic factors related to cell replication and aerobic metabolism, and also to extrinsic factors, one of the most important of which is sunlight.1,2 Sunlight includes radiation in the infrared, visible and ultraviolet regions of the spectrum. 3 Ultraviolet radiation can be subdivided into UVA, UVB and UVC wavelengths. 3 UVA waves range from 320 to 400 nm in length, and have many biological effects on living organisms. UVA induces the production of matrix metalloproteinases that degrade the extracellular matrix; 4 it also induces the production of singlet oxygen, 5 which causes deletions or point mutations in mitochondrial DNA (considered to be a fundamental cause of ageing), 6 and contributes to DNA damage, which activates the DNA damage response system, 7 eventually leading to cell senescence.
Telomeres are specialized structures occurring at the ends of chromosomes. They are several thousand bases (kb) in length and consist of the tandemly repeated DNA sequence TTAGGG. Telomeres are approximately 15 kb long at birth but may be shorter than 5 kb in older persons and persons with chronic diseases. 8 The telomere acts as a protective cap, preventing the ends of chromosomes from being recognized as broken or damaged DNA. 8 Telomeres shorten when the cell divides, and all human tissues except brain and myocardium show telomere shortening with age.9–12 This property allows telomeres to be used as biomarkers of cellular senescence. Telomere length can be reduced by stresses, such as psychological stress and obesity,13,14 and there is evidence that telomeres are hypersensitive to UV-induced DNA damage and are refractory to repair. 15
The aim of the present research study was to investigate the effect of UVA irradiation on telomere shortening and attrition.
Materials and methods
Sampling and Cell Culture
Primary human skin fibroblasts were derived from foreskins of 6- to 10 year-old boys after circumcision, with the approval of the Air Force General Hospital Ethics Committee. The samples were washed with a solution containing 75% alcohol, 10 mM phosphate-buffered saline (PBS; pH 7.4) and 1% antibiotic (100 IU/ml penicillin/streptomycin; PAA Laboratories, Pasching, Austria). After removing the epidermis, the dermis was cut into small pieces and digested in a solution of 0.03% collagenase type I (Worthington Biochemical Corp., Lakewood, NJ, USA) at 37 °C for 2 – 3 h. The cells were then rinsed with 10 mM PBS, pH 7.4, and cultured in Dulbecco's modified Eagle's medium (DMEM; PAA Laboratories) supplemented with 10% (v/v) fetal bovine serum (PAA Laboratories) at 37 °C in 5% carbon dioxide in a humidified incubator. After 5 – 6 days, the adherent, confluent diploid fibroblasts were trypsinized and passaged. These cells were considered as generation 1. The fibroblasts were subcultured until 80 – 90% confluence and serial passaging was then performed by trypsinization. For the experiments, cells were used at generations 4 – 12.
UVA Irradiation
The cells were seeded at a density of 2 × 105 cells per 35 mm culture dish. Before UVA irradiation, they were washed and covered with 10 mM PBS, pH 7.4. The lids of the culture dishes were removed and the cells were then exposed to UVA irradiation, with the source at a fixed distance of 5 cm from the surface of the cell culture. The source was a solar simulator (Aohua, Beijing, China) emitting UVA light mainly between 320 and 400 nm. Each dose of radiation was replicated with three dishes. The intensity of irradiation, measured with a UV meter (made at Beijing Normal University, Beijing, China), was 30 mW/cm2. Control cells were incubated under the same conditions as experimental cells but were not exposed to UVA.
Effect of UVA on Cell Viability
To examine phototoxicity, experimental cells were exposed to 0, 2000, 4000, 6000, 8000 or 10000 mJ/cm2 of UVA. The culture medium was replaced 24 h after irradiation with fresh DMEM containing 10% water-soluble tetrazolium salts (WST-8; Dojindo Molecular Technologies, Tokyo, Japan). Optical density was measured after 2 h with a microplate reader (RT-6000; Rayto Life and Analytical Sciences, Shenzhen, China).
Telomere Erosion After UVA Irradiation
Fibroblasts were exposed to a single dose of 500, 1000 or 10000 mJ/cm2 of UVA, or were not exposed to UVA (control). The cells were kept for a further 24 h in the same medium without passaging and telomere length was then determined as described below.
Telomere Length and Effects of UVA Irradiation and Cell Passaging
Fibroblasts were exposed to 1000 mJ/cm2 of UVA irradiation. The cells were then aliquoted 1 : 1; one aliquot was stored at -80°C; the other was plated in a 35 mm culture dish and cultured for a further two cell generations. DNA was then extracted from both groups of cells and telomere length was determined as described below. Similarly, two aliquots of nonirradiated control cells were cultured for two generations or stored at -80 °C without passaging until extraction of DNA and determination of telomere length.
DNA Extraction and Determination of Telomere Length
DNA was extracted with a TIANamp Genomic DNA kit (Tiangen Biotech, Beijing, China) according to the manufacturer's protocol. Telomere length was determined as described previously. 16 Briefly, to amplify the telomeres, two sets of real time polymerase chain reactions (PCRs) were performed for each sample: the first amplified telomeric DNA (T) and the second amplified a single-copy control gene (S), 36B4. These were both performed in triplicate. The ratio of telomere repeat copy number to single-copy gene copy number (T/S) is proportional to the average telomere length in a cell. The T/S ratio of one individual relative to the T/S ratio of another should correspond to the relative telomere lengths of their DNAs. 16
Specific primers for telomere and 36B4 amplification were: telomere, 5′-GGTTT TTGAGGGTGAGGGTGAGGGTGAGGGTGAG GGT-3′ (forward); 5′-TCCCGACTATCCCT ATCCCTATCCCTATCCCTATCCCTA-3′ (reverse); 36B4, 5′-CAGCAAGTGGGAAGG TGTAATCC-3′ (forward); 5′-CCCATTCTA TCATCAACGGGTACAA-3′ (reverse).
All PCRs were performed using the ABI Prism® 7700 Sequence Detection System (Applied Biosytems, Foster City, CA, USA), using SYBR® Green mixture (Invitrogen, Carlsbad, CA, USA) and with 36B4 as internal control. The reaction mixtures were adjusted to final volumes of 30 μl/well using 1.5 μl of genomic DNA (17.5 ng/μl), 3 μl of both primers (telomere 1, 2.70 mM; telomere 2, 9 mM; 36B4 1, 3 mM; 36B4 2, 5 mM), 7.5 μl of sterile water and 15 μl of Master Mix (Real Master Mix-SYBR® Green; Tiangen Biotech, Beijing, China).
The PCRs for amplification of the telomeres, performed in triplicate, started at 95 °C for 15 min, followed by 35 cycles of 95 °C for 15 s, 58 °C for 30 s and 72 °C for 30 s. Those for 36B4, also performed in triplicate, started at 95 °C for 15 min, followed by 35 cycles of 94 °C for 30 s, 60 °C for 30 s and 72 °C for 30 s. Melting curves were examined for problems associated with genomic DNA contamination, primer– dimers or multiple products. The telomere PCR yielded the expected product, in the range 76 – 500 bp, mainly 76 bp. The product yielded by 36B4 PCR was of about 74 bp. 16
Statistical Analyses
The mean ± SD were calculated and all statistical analyses were performed with the SPSS® statistical package, version 11.0 for Windows® (SPSS Inc., Chicago, IL, USA). The effects of cell generation number and UVA irradiation on telomere length were assessed by one-way analysis of variance (ANOVA); when the analysis indicated a significant difference, the means were compared by Student–Newman–Keuls analysis. Correlations between the UVA dose and telomere length, and between cell generation number and telomere length were calculated using linear regression analysis. Two-way ANOVA was used to determine whether passaging or UVA was the main factor associated with telomere shortening. A P-value < 0.05 was considered statistically significant.
Results
Effect of UVA on cell Viability
Viability of the human fibroblast cells, measured 24 h after receiving UVA irradiation, decreased as the UVA dose increased from 2000 to 10000 mJ/cm2 (Fig. 1). The median inhibitory UVA dose was 4000 mJ/cm2.
Cytotoxicity analysis of human fibroblast cells 24 h after receiving various doses of ultraviolet A (UVA) irradiation showing survival relative to the proportion of cells surviving after receiving no UVA irradiation; mean ± SD of three independent experiments
Effect of Cell Passaging on Telomere Length
To confirm the reliability of the experimental system and the change in telomere length with intrinsic ageing of human fibroblasts, the telomere lengths of generations 8, 10 and 12 were compared. There was a trend for relative telomere length (T/S) to decrease as generation number increased (R2 = 0.636, P = 0.01), from a mean ± SD of 1 ± 0.02 in generation 8 to 0.82 ± 0.05 and 0.76 ± 0.14 in generations 10 and 12, respectively. Relative telomere length was significantly shorter in generations 10 and 12 than in generation 8 (P = 0.042 and P = 0.013, respectively).
Telomere Erosion After UVA Irradiation
Irradiation with UVA significantly shortened the telomeres (P = 0.038). Telomere length in fibroblasts exposed to 1000 and 10 000 mJ/cm2 UVA was significantly shorter than in control fibroblasts (P = 0.027 and P = 0.009, respectively). Moreover, telomere length was significantly correlated with UVA irradiation dose (P = 0.044, R2 = 0.346; Fig. 2); it declined from 0.79 ± 0.09 at a dose of 500 mJ/cm2 to 0.59 ± 0.21 and 0.49 ± 0.29 at doses of 1000 and 10 000 mJ/cm2, respectively.
Telomere length in cultured human fibroblasts exposed to different doses of ultraviolet A (UVA) irradiation relative to the length in cells that received no UVA irradiation (linear regression analysis demonstrated a significant inverse relationship between telomere length and UVA dose [R2 = 0.346, P = 0.044]; *P < 0.05, **P < 0.01 compared with nonirradiated cells (Student–Newman–Keuls analysis)
Telomere Length and Effects of UVA Irradiation and Cell Passaging
Telomere length showed no significant difference between generations 0 and 2, however UVA irradiation did show a statistically significantly effect on telomere length (P = 0.044) (Fig. 3). The interaction effect was not statistically significant (two-way ANOVA). Thus, UVA was more closely related to telomere shortening than cell passaging.
Telomere length in generations 0 and 2 of cultured human fibroblasts relative to the length in generation 0 cells that received no irradiation following 0 mJ/cm2 (control) or 1000 mJ/cm2 UVA irradiation (no significant difference between generations 0 and 2; P = 0.044 for effect of UVA irradiation), two-way analysis of variance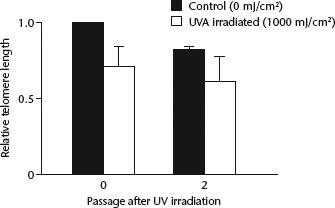
Discussion
This study found a shortening of telomere length as cell generation increased and with increasing dose of UVA irradiation, and confirmed that UVA irradiation is more closely related than cell generation to telomere shortening. The results provide further evidence that telomere shortening may be part of the process of replicative senescence, 17 and that telomere shortening may be one of the mechanisms of UV-induced cellular senescence.
The progressive shortening of the telomere is the ‘mitotic clock' that regulates the onset of replicative senescence in normal somatic cells. 18 In normal proliferating somatic cells, telomeres shorten until they reach a critical length (the Hayflick limit), at which point the cells stop proliferating and enter a permanently nondividing state (senescence). 19 The telomere cap is composed of a DNA–multiprotein complex with specific structure and functions. 20 The classical view of telomere biology described above remains consistent with this updated view: the telomere is able to strengthen and maintain the stability of the telomere cap if the telomere is long enough. 21
In the present study, UVA induced and accelerated telomere shortening. When telomeres reach a critical length, i.e. when they are too short to provide a basis for the telomere cap structure, they activate a signalling pathway that leads to senescence. The protein p53 is a downstream effector of telomere damage signalling and is activated by the exogenous DNA damage arising from post-translational modifications caused by UV exposure. 22 It finally induces apoptotic cell death and cellular senescence via the ATM (ataxia telangiectasia mutated) and ATR (ATM-Rad3-related) signalling pathways, each of which functions as a tumour suppressor mechanism and has a role in ageing. 13
In conclusion, telomeres were shown to be adversely affected by UVA irradiation and the damage caused was related to cell ageing. These findings add weight to recommendations by dermatologists that the skin should be protected from UV damage. Telomeres may be a useful new target for attempts to prevent photoageing.
Footnotes
Acknowledgements
We thank Dr Qing Cai, who is Director of the Centre Laboratory of the Air Force General, for sharing his PCR equipment.
The authors had no conflicts of interest to declare in relation to this article.
